Helidac: Package Insert / Prescribing Info
Package insert / product label
Generic name: bismuth subsalicylate, metronidazole, tetracycline hydrochloride
Dosage form: kit, tablets, capsules
Drug class: H. pylori eradication agents
Medically reviewed by Drugs.com. Last updated on Jun 16, 2025.
On This Page
THESE PRODUCTS ARE INTENDED ONLY FOR USE AS DESCRIBED. The individual products contained in this package should not be used alone or in combination for other purposes. The information described in this labeling concerns only the use of these products as indicated in this combination package. For information on use of the individual components when dispensed as individual medications outside this combined use for treating Helicobacter pylori, please see the package inserts for each individual product.
WARNING: POTENTIAL FOR CARCINOGENICITY
Metronidazole has been shown to be carcinogenic in mice and rats. It is unknown whether metronidazole is associated with carcinogenicity in humans (see WARNINGS).
Helidac Description
HELIDAC Therapy consists of 112 bismuth subsalicylate 262.4-mg chewable tablets, 56 metronidazole 250-mg tablets, USP, and 56 tetracycline hydrochloride 500-mg capsules, USP, for oral administration.
Bismuth subsalicylate chewable tablets: Each pink round tablet contains 262.4 mg bismuth subsalicylate (102 mg salicylate) for oral administration.
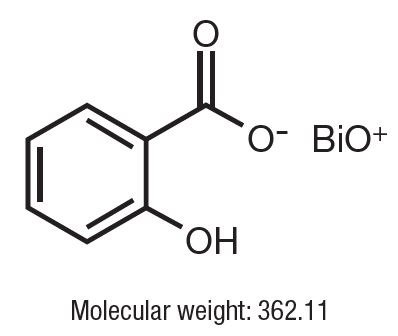
Bismuth subsalicylate is a fine, white, odorless, and tasteless powder that is stable and non-hygroscopic. It is a highly insoluble salt of trivalent bismuth and salicylic acid.
Bismuth subsalicylate is 2-Hydroxybenzoic acid bismuth (3+) salt with the following structural formula:
Inactive Ingredients: Each bismuth subsalicylate chewable tablet contains calcium carbonate, D&C Red No. 27 aluminum lake, magnesium stearate, mannitol, povidone, saccharin sodium, talc, and wintergreen flavor.
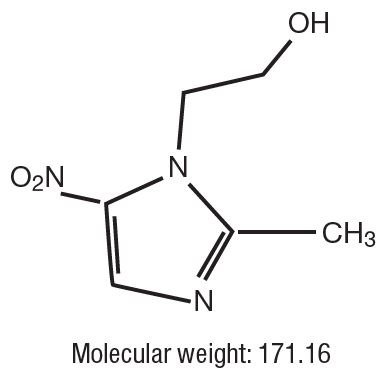
Metronidazole tablets, USP: Each round, white tablet contains 250 mg metronidazole. Metronidazole is 2-Methyl-5-nitroimidazole-1-ethanol, with the following structural formula:
Inactive Ingredients: Each metronidazole tablet contains colloidal silicon dioxide, hydroxypropyl cellulose, lactose (anhydrous), sodium starch glycolate, stearic acid, and microcrystalline cellulose.
Tetracycline hydrochloride capsules, USP: Each black and yellow capsule contains 500 mg tetracycline hydrochloride. Tetracycline is a yellow, odorless, crystalline powder. Tetracycline is stable in air but exposure to strong sunlight causes it to darken. Its potency is affected in solutions of pH below 2 and is rapidly destroyed by alkali hydroxide solutions. Tetracycline is very slightly soluble in water, freely soluble in dilute acid and in alkali hydroxide solutions, sparingly soluble in alcohol, and practically insoluble in chloroform and ether.
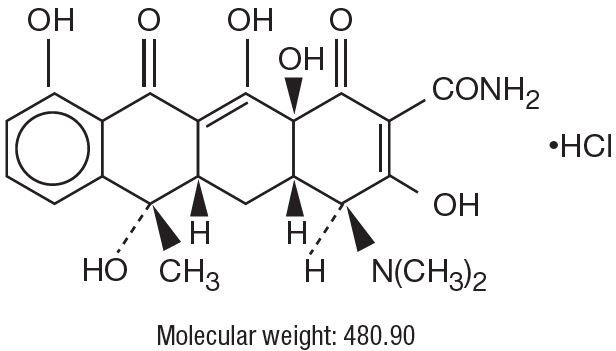
Tetracycline hydrochloride is (4S,4aS,5aS,6S,12aS)-4-(Dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,6,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide monohydrochloride, with the following structural formula:
Inactive Ingredients: Each tetracycline hydrochloride capsule contains lactose, magnesium stearate and sodium lauryl sulfate; D & C yellow no.10, FD & C blue No 1, FD & C red no. 40, gelatin and titanium dioxide. It may also contain benzyl alcohol, butylparaben, edetate calcium disodium, FD & C yellow no. 6, methylparaben, propylparaben, silicon dioxide and sodium propionate.
The imprinting ink for tetracycline hydrochloride capsule contains titanium dioxide. It may also contain dimethyl polysiloxane, distilled water, ethyl alcohol, ethylene glycol monoethyl ether, pharmaceutical glaze, pharmaceutical shellac, and soya lecithin.
Helidac - Clinical Pharmacology
Pharmacokinetics: Pharmacokinetics for HELIDAC Therapy components (bismuth subsalicylate chewable tablets, metronidazole tablets, and tetracycline hydrochloride capsules) when coadministered has not been studied. There is no information about the gastric mucosal concentrations of bismuth, metronidazole, and tetracycline after administration of these agents concomitantly or in combination with an acid suppressive agent. The systemic pharmacokinetic information presented below is based on studies in which each product was administered alone.
Bismuth Subsalicylate: Upon oral administration, bismuth subsalicylate is almost completely hydrolyzed in the gastrointestinal tract to bismuth and salicylic acid. Thus, the pharmacokinetics of bismuth subsalicylate following oral administration can be described by the individual pharmacokinetics of bismuth and salicylic acid.
Bismuth: Less than 1% of bismuth from oral doses of bismuth subsalicylate is absorbed from the gastrointestinal tract into the systemic circulation. Absorbed bismuth is distributed throughout the body. Bismuth is highly bound to plasma proteins (> 90%). Bismuth has multiple disposition half-lives with an intermediate half-life of 5 to 11 days and a terminal half-life of 21 to 72 days. Elimination of bismuth is primarily through urinary and biliary routes with a renal clearance of 50 ± 18 mL/min. The mean trough blood bismuth concentration after 2 weeks oral administration of 787 mg bismuth subsalicylate (3 chewable tablets) four times daily under fasted condition was 5.1 ± 3.1 ng/mL. In another study, the mean trough blood bismuth concentration after 2 weeks oral administration of 525 mg bismuth subsalicylate (as PEPTO-BISMOL® liquid suspension) four times daily was 5 ng/mL with the highest value being 32 ng/mL.
Salicylic Acid: More than 80% of the salicylic acid is absorbed from oral doses of bismuth subsalicylate chewable tablets. Salicylic acid is about 90% plasma protein bound. The volume of distribution is about 170 mL/kg of body weight. Salicylic acid is extensively metabolized and about 10% is excreted unchanged in the urine. The metabolic clearance of salicylic acid is saturable; accordingly, nonlinear pharmacokinetics is observed at bismuth subsalicylate doses above 525 mg. Salicylic acid metabolic clearance is lower in females than in males. The terminal half-life of salicylic acid upon a single oral dose of 525 mg bismuth subsalicylate is between 2 to 5 hours. After a single oral dose of 525 mg bismuth subsalicylate (2 chewable tablets), the mean peak plasma salicylic acid concentration was 13.1 ± 3.4 mcg/mL under fasted condition. The mean steady-state serum total salicylate concentration after 2 weeks oral administration of 525 mg bismuth subsalicylate (as PEPTO-BISMOL liquid suspension) four times daily was 24 mcg/mL with the highest value being 70 mcg/mL.
Metronidazole: Following oral administration, metronidazole is well absorbed, with peak plasma concentrations occurring between 1 and 2 hours after administration. Plasma concentrations of metronidazole are proportional to the administered dose, with oral administration of 250 mg producing a peak plasma concentration of 6 mcg/mL. Studies reveal no significant bioavailability differences between males and females; however because of weight differences, the resulting plasma levels in males are generally lower.
Metronidazole is the major component appearing in the plasma, with lesser quantities of the 2-hydroxymethyl metabolite also being present. Less than 20% of the circulating metronidazole is bound to plasma proteins. Metronidazole also appears in cerebrospinal fluid, saliva, and human milk in concentrations similar to those found in plasma.
The average elimination half-life in normal volunteers is 8 hours. The major route of elimination of metronidazole and its metabolites is via the urine (60% to 80% of the dose), with fecal excretion accounting for 6% to 15% of the dose. The metabolites that appear in the urine result primarily from side-chain oxidation [1-(ß-hydroxyethyl)-2-hydroxymethyl-5-nitroimidazole and 2-methyl-5-nitroimidazole-1-yl-acetic acid] and glucuronide conjugation, with unchanged metronidazole accounting for approximately 20% of the total. Renal clearance of metronidazole is approximately 10 mL/min/1.73 m2.
Decreased renal function does not alter the single-dose pharmacokinetics of metronidazole. In patients with decreased liver function, plasma clearance of metronidazole is decreased.
Tetracycline Hydrochloride: Tetracycline hydrochloride is absorbed (60%-90%) in the stomach and upper small intestine. The presence of food, milk or cations may significantly decrease the extent of absorption. In the plasma, tetracycline is bound to plasma proteins in varying degrees. It is concentrated by the liver in the bile and excreted in the urine and feces at high concentrations in biologically active form. Tetracycline hydrochloride is distributed into most body tissues and fluids. It is distributed into the bile and undergoes varying degrees of enterohepatic recirculation. Tetracycline hydrochloride tends to localize in tumors, necrotic or ischemic tissue, liver and spleen and form tetracycline-calcium orthophosphate complexes at sites of new bone formation or tooth development. Tetracycline readily crosses the placenta and is excreted in high amounts in breast milk (see WARNINGSand PRECAUTIONS, Nursing Mothers). Tetracyclines are readily absorbed and are bound to plasma proteins in varying degrees. They are concentrated by the liver in the bile and excreted in the urine and feces at high concentrations in a biologically active form.
The relative contribution of systemic versus local antimicrobial activity against H. pylori for agents used in eradication therapy has not been established.
Microbiology:
Mechanism of Action
HELIDAC Therapy is a combination of antibacterial agents (metronidazole and tetracycline hydrochloride) and bismuth subcitrate potassium. Tetracycline hydrochloride interacts with the 30S subunit of the bacterial ribosome and inhibits protein synthesis. Metronidazole's antibacterial mechanism of action in an anaerobic environment is not fully understood but a possible mechanism includes reduction by intracellular electron transport proteins after entry into the organism. Because of this alteration to the metronidazole molecule, a concentration gradient is created and maintained which promotes the drug's intracellular transport. Presumably, free radicals are formed which, in turn, react with cellular components resulting in death of bacteria. The antibacterial action of bismuth salts is not well understood.
Activity In Vitro and In Vivo
HELIDAC Therapy has been shown to be active against most isolates of Helicobacter pylori both in vitro, and in clinical infections as described in the CLINICAL STUDIES and INDICATIONS AND USAGEsections.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
Indications and Usage for Helidac
The components of HELIDAC Therapy (bismuth subsalicylate, metronidazole, and tetracycline hydrochloride), in combination with an H2 antagonist, are indicated for the eradication of H. pylori for treatment of patients with H. pylori infection and duodenal ulcer disease (active or a history of duodenal ulcer). The eradication of H. pylori has been demonstrated to reduce the risk of duodenal ulcer recurrence in patients with active duodenal ulcer disease (see CLINICAL STUDIESand DOSAGE AND ADMINISTRATION).
To reduce the development of drug-resistant bacteria and maintain the effectiveness HELIDAC Therapy and other antibacterial drugs, HELIDAC Therapy should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Contraindications
Methoxyflurane
Do not administer methoxyflurane to patients HELIDAC Therapy. The concurrent use of tetracycline hydrochloride, a component of HELIDAC Therapy with methoxyflurane has been reported to result in fatal renal toxicity (see DRUG INTERACTIONS).
Disulfiram
HELIDAC Therapy is contraindicated in patients who have taken disulfiram within the last two weeks. Psychotic reactions have been reported in alcoholic patients who are using metronidazole, a component of HELIDAC Therapy and disulfiram concurrently (see DRUG INTERACTIONS).
Alcohol
Alcoholic beverages or other products containing propylene glycol should not be consumed during and for at least 3 days after usage of HELIDAC Therapy. A disulfiram-like reaction (abdominal cramps, nausea, vomiting, headaches, and flushing) may occur due to the interaction between alcohol or propylene glycol and metronidazole, a component of HELIDAC Therapy (see DRUG INTERACTIONS).
Renal Impairment
HELIDAC Therapy is contraindicated in patients with severe renal impairment. The antianabolic action of the tetracyclines may cause an increase in blood urea nitrogen (BUN) (see ADVERSE REACTIONS). In patients with significantly impaired renal function, higher serum concentrations of tetracyclines may lead to azotemia, hyperphosphatemia, and acidosis.
Hypersensitivity Reactions
HELIDAC Therapy is contraindicated in patients with known hypersensitivity (e.g. urticaria, erythematous rash, flushing, and fever) to bismuth subsalicylate, metronidazole or other nitroimidazole derivatives, or tetracycline (see ADVERSE REACTIONS).
Allergy to Aspirin or other Salicylates
HELIDAC Therapy should not be administered to those patients who have a known allergy to aspirin or salicylates.
Warnings
Potential for Carcinogenicity
Metronidazole has been shown to be carcinogenic in mice and rats. Tumors affecting the liver, lungs, mammary and lymphatic tissues have been detected in several studies of metronidazole in rats and mice, but not hamsters (see PRECAUTIONS, Carcinogenesis, Mutagenesis, Impairment of Fertility). It is unknown whether metronidazole is associated with carcinogenicity in humans.
Fetal Toxicity
There are no adequate and well-controlled studies of HELIDAC Therapy in pregnant women. However, tetracycline can cause fetal harm when administered to a pregnant woman. The use of drugs of the tetracycline class during the second and third trimester of pregnancy can also cause permanent discoloration of the teeth (yellow-gray brown) and possibly inhibit bone development (See WARNINGS). Administration of oral tetracycline to pregnant rats at various doses resulted in yellow fluorescence in teeth and bones in the newborn animals. Metronidazole usage in pregnancy has been evaluated in numerous studies. In one of these studies, an increased risk of cleft lip, with or without cleft palate, was noted in infants exposed to metronidazole in–utero; however these findings were not confirmed. If HELIDAC Therapy is used during pregnancy, or if the patient becomes pregnant while taking HELIDAC Therapy, the patient should be apprised of the potential hazard to the fetus (see PRECAUTIONS, Pregnancy).
Maternal Toxicity
Tetracycline administered during pregnancy at high doses (> 2 g IV) was associated with rare but serious cases of maternal hepatotoxicity. This syndrome may result in stillborn or premature birth due to maternal pathology (see PRECAUTIONS).
Tooth Enamel Discoloration and Hypoplasia
The use of drugs of the tetracycline class during tooth development (last half of pregnancy, infancy, and childhood to the age of 8 years) may cause permanent discoloration of the teeth (yellow-gray-brown). This adverse reaction is more common during long-term use of the drug, but has been observed following repeated short-term courses. Enamel hypoplasia has also been reported. HELIDAC Therapy, therefore, should not be used in this age group unless other drugs are not likely to be effective or are contraindicated (see PRECAUTIONS).
Central and Peripheral Nervous System Effects
Metronidazole: Cases of convulsive seizures, encephalopathy and peripheral neuropathy (including optic neuropathy) have been reported with metronidazole. Encephalopathy has been reported in association with cerebellar toxicity characterized by ataxia, dizziness, and dysarthria. CNS lesions seen on MRI have been described in reports of encephalopathy. CNS symptoms are generally reversible within days to weeks upon discontinuation of metronidazole. CNS lesions seen on MRI have also been described as reversible. Peripheral neuropathy, mainly of sensory type has been reported and is characterized by numbness or paresthesia of an extremity. Cases of aseptic meningitis have been reported with metronidazole. Symptoms can occur within hours of dose administration and generally resolve after metronidazole therapy is discontinued.
Tetracycline: Intracranial hypertension (IH, pseudotumor cerebri) has been associated with the use of tetracyclines. Clinical manifestations of IH include headache, blurred vision, diplopia, and vision loss; papilledema can be found on fundoscopy. Women of childbearing age who are overweight or have a history of IH are at greater risk for developing tetracycline associated IH. Concomitant use of isotretinoin should be avoided because isotretinoin is also known to cause IH.
Although IH typically resolves after discontinuation of treatment, the possibility for permanent visual loss exists. If visual disturbance occurs during treatment, prompt ophthalmologic evaluation is warranted. Since intracranial pressure can remain elevated for weeks after drug cessation, patients should be monitored until they stabilize.
Bismuth-containing products: Cases of neurotoxicity associated with excessive doses of various bismuth-containing products, including bismuth subsalicylate have been reported. Effects have been reversible with discontinuation of therapy.
The appearance of abnormal neurologic signs and symptoms demands the prompt evaluation of the benefit/risk ratio of the continuation of therapy (see ADVERSE REACTIONS).
Risk of Reye’s syndrome
Use of HELIDAC Therapy is not recommended in children and teenagers who have or who are recovering from varicella (chicken pox) or influenza due to the risk of Reye’s syndrome, a rare but serious illness. If HELIDAC Therapy is used in this population and nausea or vomiting is present, patients are advised to consult a doctor because this could be an early sign of Reye’s syndrome.
Cutaneous Reactions
Skin and subcutaneous disorders including Stevens-Johnson syndrome, toxic epidermal necrolysis and DRESS syndrome (drug rash with eosinophilia and systemic symptoms) have been reported. Discontinue treatment at the first evidence of a cutaneous reaction (see ADVERSE REACTIONS).
Precautions
Development of Superinfections
Known or previously unrecognized candidiasis may present more prominent symptoms during therapy with metronidazole and requires treatment with an antifungal agent.
As with other antibiotics, use of tetracycline hydrochloride may result in overgrowth of nonsusceptible organisms, including fungi. If superinfection occurs discontinue HELIDAC Therapy and institute appropriate therapy.
Photosensitivity
Photosensitivity manifested by an exaggerated sunburn reaction has been observed in some individuals taking tetracyclines (see ADVERSE REACTIONS). Patients apt to be exposed to direct sunlight or ultraviolet light should be advised that this reaction can occur with tetracycline drugs. Instruct patients taking HELIDAC Therapy to avoid exposure to the sun or sun lamps. Treatment should be discontinued at the first evidence of skin erythema.
Darkening of the Tongue and/or Black Stool
Bismuth subsalicylate may cause a temporary and harmless darkening of the tongue and/or black stool, generally reversible within several days after treatment is stopped (see ADVERSE REACTIONS). Stool darkening should not be confused with melena.
Use in Patients with Blood Dyscrasias
Metronidazole is a nitroimidazole and should be used with caution in patients with evidence of, or history of, blood dyscrasia. A mild leukopenia has been observed during its administration; however, no persistent hematologic abnormalities attributable to metronidazole have been observed in clinical studies. Total and differential leukocyte counts are recommended before and after therapy (see ADVERSE REACTIONS).
Increased Plasma Concentrations in Patients with Hepatic Impairment
Patients with hepatic impairment metabolize metronidazole slowly, with resultant accumulation of metronidazole in the plasma. Use HELIDAC Therapy with caution in patients with mild to moderate hepatic impairment. HELIDAC Therapy may not be appropriate for patients with severe hepatic impairment (Child-Pugh C) (see DOSAGE AND ADMINISTRATIONand CLINICAL PHARMACOLOGY).
Laboratory Test Interactions
Bismuth absorbs x-rays and may interfere with x-ray diagnostic procedures of the gastrointestinal tract.
Bismuth subsalicylate may cause a temporary and harmless darkening of the stool. However, this change does not interfere with standard tests for occult blood.
Metronizadole may interfere with certain types of determinations of serum chemistry values, such as aspartate aminotransferase (AST, SGOT), alanine aminotransferase (ALT, SGPT), lactate dehydrogenase (LDH), triglycerides, and hexokinase glucose. Values of zero may be observed. All of the assays in which interference has been reported involve enzymatic coupling of the assay to oxidation-reduction of nicotinamide (NAD+ <=> NADH). Interference is due to the similarity in absorbance peaks of NADH (340 nm) and metronidazole (322 nm) at pH 7.
Development of Drug Resistant Bacteria
Prescribing HELIDAC Therapy in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
INFORMATION FOR PATIENTS:
Pregnant Women
Advise pregnant women that HELIDAC Therapy can cause fetal harm. Tetracycline use during the second and third trimesters of pregnancy can cause permanent discoloration of the teeth (yellow-gray brown) and may inhibit bone development and metronidazole has been shown in one study to increase the risk of cleft lip with or without cleft palate (see WARNINGS).
Human Milk Feeding
Advise women to avoid feeding their infants with their milk during HELIDAC Therapy. Women should either discontinue feeding or pump and discard their milk during treatment and for 24 hours after the last dose (see PRECAUTIONS, Nursing Mothers).
Hypersensitivity
Inform patients that HELIDAC Therapy may cause allergic reactions and to discontinue HELIDAC Therapy at the first sign of urticaria, erythematous rash, flushing, and fever or other symptoms of an allergic reaction (see CONTRAINDICATIONS).
Central Nervous System Effects
Inform patients of the risk of central and peripheral nervous system effects with HELIDAC Therapy and to discontinue HELIDAC Therapy and report immediately to their health-care provider if any neurologic symptoms occur (see WARNINGS).
Drug Interactions
Patients should be advised to report to their doctor the use of any other medications while taking HELIDAC Therapy. The administration of any of the following drugs with HELIDAC Therapy may result in clinically significant adverse reactions or insufficient drug efficacies (see CONTRAINDICATIONS, WARNINGS, PRECAUTIONS, and DRUG INTERACTIONS).
• Methoxyflurane
• Disulfiram
• Alcoholic Beverages, or Products Containing Propylene Glycol
• Oral Contraceptives
• Anticoagulants
• Lithium
• Antacids, Multivitamins, or Dairy Products
• Antidiabetic agents
• Aspirin
• Probenecid or Sulfinpyrazone
• Cimetidine
• Phenytoinor Phenobarbital
Darkening of the Tongue and/or Stool
HELIDAC Therapy may cause temporary and harmless darkening of the tongue and/or black stool and is generally reversible within several days after treatment is stopped. Stool darkening should not be confused with melena (blood in the stool) (see PRECAUTIONS).
Dosing and Administration Information
Each dose includes 4 pills: 2 pink round chewable tablets (bismuth subsalicylate), 1 white round tablet (metronidazole), and 1 black and yellow capsule (tetracycline hydrochloride). Each dose (all 4 pills) should be taken 4 times a day, at mealtimes and bedtime. Patients should be instructed to chew and swallow the pink round tablets (bismuth subsalicylate tablets) and to swallow the white round tablet (metronidazole tablet) and the black and yellow capsule (tetracycline hydrochloride capsule) whole with a full glass of water (8 ounces). Concomitantly prescribed H2 antagonist therapy should be taken as directed.
Missed doses can be made up by continuing the normal dosing schedule until the medication is gone. Patients should not take double doses. (If more than 4 doses are missed, the prescriber should be contacted (see DOSAGE AND ADMINISTRATION).
Salicylate Toxicity
HELIDAC Therapy contains salicylates. If taken with aspirin and ringing in the ears occurs, the prescriber should be consulted concerning discontinuation of the aspirin therapy until the HELIDAC Therapy is completed (see OVERDOSAGE, WARNINGS, and DRUG INTERACTIONS).
Antibacterial Resistance
Patients should be counseled that antibacterial drugs, including HELIDAC Therapy, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When HELIDAC Therapy is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by HELIDAC Therapy or other antibacterial drugs in the future.
DRUG INTERACTIONS:
Methoxyflurane
Do not administer methoxyflurane to patients taking HELIDAC Therapy. The concurrent use of tetracycline hydrochloride, a component of HELIDAC Therapy, with methoxyflurane has been reported to result in fatal renal toxicity (see CONTRAINDICATIONS).
?Disulfiram
Psychotic reactions have been reported in alcoholic patients who are using metronidazole and disulfiram concurrently. HELIDAC Therapy should not be given to patients who have taken disulfiram within the last 2 weeks (see CONTRAINDICATIONS).
Alcohol
Consumption of alcoholic beverages or administration of other products containing propylene glycol during treatment with HELIDAC Therapy and for at least 3 days afterwards may cause a disulfiram-like reaction (abdominal cramps, nausea, vomiting, headaches, and flushing) due to the interaction between alcohol or propylene glycol and metronidazole, a component of HELIDAC Therapy. Discontinue alcoholic beverage or other products containing propylene glycol during and for at least 3 days after therapy with HELIDAC Therapy (see CONTRAINDICATIONS).
Oral Contraceptives
Concurrent use of HELIDAC Therapy with oral contraceptive may make oral contraceptives less effective due to an interaction with the tetracycline component of HELIDAC Therapy. Breakthrough bleeding has been reported. Women of child-bearing potential should use a different or additional form of contraception while taking HELIDAC Therapy.
Anticoagulants
HELIDAC Therapy may alter the anticoagulant effects of warfarin and other oral coumarin anticoagulants. Salicylates may cause an increased risk of bleeding when administered with anticoagulant therapy. Metronidazole has been reported to potentiate the anticoagulant effect of warfarin, and other oral coumarin anticoagulants, resulting in a prolongation of prothrombin time. Tetracycline has been shown to depress plasma prothrombin activity. Prothrombin time, International Normalized Ratio (INR), or other suitable anticoagulation tests should be closely monitored if HELIDAC Therapy is administered concomitantly with warfarin. Patients should also be monitored for evidence of bleeding.
Lithium
In patients stabilized on relatively high doses of lithium, short-term use of HELIDAC Therapy may cause elevation of serum lithium concentrations and signs of lithium toxicity due to the interaction between metronidazole and lithium. Serum lithium and serum creatinine concentrations should be monitored several days after beginning treatment with HELIDAC Therapy to detect any increase that may precede clinical symptoms of lithium toxicity.
Antacids, Multivitamins, or Dairy Products
The absorption of HELIDAC Therapy may be reduced if administered with antacids containing aluminium, calcium, or magnesium; preparations containing iron, zinc, or sodium bicarbonate; or milk or dairy products due to the interaction between these products and tetracycline. These products should not be consumed concomitantly with HELIDAC Therapy. However, the clinical significance of reduced tetracycline systemic exposure is unknown as the relative contribution of systemic versus local antimicrobial activity against Helicobacter pylori has not been established.
Antidiabetic agents, Aspirin, Probenecid, or Sulfinpyrazone
Caution is advised in the administration of bismuth subsalicylate to patients taking medication for diabetes (possible enhanced hypoglycemic effect when given with salicylates) or patients taking aspirin (may increase the risk of salicylate toxicity), probenecid, or sulfinpyrazone (uricosuric effects of probenecid or sulfinpyrazone may be decreased) (see CONTRAINDICATIONS, WARNINGS).
Busulfan
Metronidazole has been reported to increase plasma concentrations of busulfan, which can result in an increased risk for serious busulfan toxicity. HELIDAC Therapy should not be administered concomitantly with busulfan unless the benefit outweighs the risk. If no therapeutic alternatives to HELIDAC Therapy are available, and concomitant administration with busulfan is medically needed, frequent monitoring of busulfan plasma concentration should be performed and the busulfan dose should be adjusted accordingly.
Inhibitors of CYP450 liver enzymes
The simultaneous administration of HELIDAC Therapy and drugs that inhibit microsomal liver enzymes, such as cimetidine, may result in a prolonged half-life and decreased plasma clearance of metronidazole.
Inducers of CYP450 liver enzymes
The simultaneous administration of HELIDAC Therapy and drugs that induce microsomal liver enzymes, such as phenytoin or phenobarbital, may accelerate the elimination of metronidazole, resulting in reduced plasma concentrations of metronidazole. Impaired clearance of phenytoin has also been reported in this situation. Monitor phenytoin concentrations during treatment with HELIDAC Therapy.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
No long-term studies have been performed to evaluate the effect of HELIDAC Therapy on carcinogenesis, mutagenesis, or impairment of fertility.
Bismuth subsalicylate
No long-term toxicity studies have been conducted with bismuth subsalicylate. Bismuth did not show mutagenic potential in the NTP salmonella plate assay. No reproductive toxicity studies have been conducted with bismuth subsalicylate.
Metronidazole
Metronidazole has shown evidence of carcinogenic activity in a number of studies involving chronic, oral administration in mice and rats. Prominent among the effects in the mouse was an increased incidence of pulmonary tumorigenesis. This has been observed in all six reported studies in that species, including one study in which the animals were dosed on an intermittent schedule (administration during every fourth week only). At the highest dose levels, (approximately 500 mg/kg/day, which is approximately 1.6 times the indicated human dose for a 60 kg adult based on body surface area) there was a statistically significant increase in the incidence of malignant liver tumors in male mice. Also, the published results of one of the mouse studies indicate an increase in the incidence of malignant lymphomas as well as pulmonary neoplasms associated with lifetime feeding of the drug. All these effects are statistically significant. Long-term, oral-dosing studies in the rat showed statistically significant increases in the incidence of various neoplasms, particularly in mammary and hepatic tumors, among female rats administered metronidazole over those noted in the concurrent female control groups. Two lifetime tumorigenicity studies in hamsters have been performed and reported to be negative. Although metronidazole has shown mutagenic activity in a number of in vitro assay systems, studies in mammals (in vivo) have failed to demonstrate a potential for genetic damage.
Fertility studies have been conducted with male rats and mice with divergent results. Metronidazole, at doses up to 400 mg/kg/day (approximately 3 times the indicated human dose based on mg/m2) for 28 days, failed to produce any adverse effects on fertility and testicular function in male rats. In another study where rats were treated with up to 400 mg/kg/day for 8 weeks, there was severe degeneration of the seminiferous epithelium in the testes which was associated with a marked decrease in testicular spermatid counts and epididymal sperm counts and a marked decrease in fecundity. These effects were partially reversible.
Fertility studies have been performed in male mice at doses up to six times the maximum recommended human dose based upon mg/m2 and have revealed no evidence of impaired fertility. Another fertility study was performed in male mice at oral doses of 500 mg/kg/day (approximately 2 times the indicated human dose based on mg/m²) for 14 days. Metronidazole significantly decreased testes and epididymides weight, decreased sperm viability, and increased the incidence of abnormal sperm. The viability of sperm was normal by 2 months after the start of the treatment.
Tumors affecting the liver, lungs, mammary, and lymphatic tissues have been detected in several studies of metronidazole in rats and mice, but not hamsters.
Pulmonary tumors have been observed in all six reported studies in the mouse, including one study in which the animals were dosed on an intermittent schedule (administration during every fourth week only). Malignant liver tumors were increased in male mice treated at approximately 1500 mg/m2 (similar to the maximum recommended daily dose, based on body surface area comparisons). Malignant lymphomas and pulmonary neoplasms were also increased with lifetime feeding of the drug to mice. Mammary and hepatic tumors were increased among female rats administered oral metronidazole compared to concurrent controls. Two lifetime tumorigenicity studies in hamsters have been performed and reported to be negative.
Metronidazole has shown mutagenic activity in in vitro assay systems including the Ames test. Studies in mammals in vivo have failed to demonstrate a potential for genetic damage.
Metronidazole failed to produce any adverse effects on fertility or testicular function in male rats at doses up at 400 mg/kg/day (similar to the maximum recommended clinical dose, based on body surface area comparisons) for 28 days. However, rats treated at the same dose for 6 weeks or longer were infertile and showed severe degeneration of the seminiferous epithelium in the testes as well as marked decreases in testicular spermatid counts and epididymal sperm counts. Fertility was restored in most rats after an eight week, drug-free recovery period.
Tetracycline hydrochloride
There has been no evidence of carcinogenicity for tetracycline hydrochloride in studies conducted with rats and mice. Some related antibiotics (oxytetracycline, minocycline) have shown evidence of oncogenic activity in rats.
There was evidence of mutagenicity by tetracycline hydrochloride in two in vitro mammalian cell assay systems (L51784y mouse lymphoma and Chinese hamster lung cells).
Tetracycline hydrochloride had no effect on fertility when administered in the diet to male and female rats at a daily intake of 25 times the human dose.
Pregnancy
Pregnancy Category D
HELIDAC Therapy contains bismuth subsalicylate, metronidazole, and tetracycline hydrochloride. There are no adequate and well-controlled studies of HELIDAC Therapy in pregnant women. However, tetracycline can cause fetal harm when administered to a pregnant woman. The use of tetracycline during the second and third trimester of pregnancy can also cause permanent discoloration of the teeth (yellow gray brown) and possibly inhibit bone development. Administration of oral tetracycline to pregnant rats at various doses resulted in yellow fluorescence in teeth and bones in the newborn animals. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus (see WARNINGS).
The available human and animal data are summarized below for each of HELIDAC Therapy's active ingredients:
Tetracycline
Published case reports have described the yellowing of bones and teeth in human infants exposed to tetracycline during the second and third trimester of pregnancy. The yellowing is caused by the direct deposition of tetracycline during the mineralization process. This discoloration is more common during long-term use of the drug but has also been observed following repeated short-term courses. All tetracyclines form a stable calcium complex in any bone forming tissue. A decrease in fibula growth rate was observed in premature infants given oral tetracycline in doses of 25 mg/kg every six hours. This effect resolved when the drug was discontinued. One long-term follow-up study in children exposed to tetracycline in-utero showed no adverse effects on bone growth and development.
Tetracycline administered during pregnancy at high doses (greater than 2 g IV) was associated with rare but serious cases of maternal hepatotoxicity. This syndrome may result in stillborn or premature birth due to maternal pathology (see WARNINGS).
Results of animal studies indicate that tetracycline crosses the placenta, is found in fetal tissues, and can have toxic effects on the developing fetus (often related to reversible retardation of skeletal development). Evidence of embryotoxicity has also been noted in animals treated early in pregnancy. Multiple studies of limited design were conducted with pregnant and lactating female rats that resulted in fetuses and neonates with yellow discoloration of bones and teeth.
Metronidazole
There are published data from case-control studies, cohort studies, and 2 meta-analyses that include more than 5000 pregnant women who used metronidazole during pregnancy. Many studies included first trimester exposures. One study showed an increased risk of cleft lip, with or without cleft palate, in infants exposed to metronidazole in-utero; however, these findings were not confirmed. In addition, more than ten randomized, placebo-controlled clinical trials enrolled more than 5000 pregnant women to assess the use of antibiotic treatment (including metronidazole) for bacterial vaginosis on the incidence of preterm delivery. Most studies did not show an increased risk for congenital anomalies or other adverse fetal outcomes following metronidazole exposure during pregnancy. Three studies conducted to assess the risk of infant cancer following metronidazole exposure during pregnancy did not show an increased risk; however, the ability of these studies to detect such a signal was limited.
Metronidazole crosses the placental barrier and its effects on the human fetal organogenesis are not known. No fetotoxicity was observed when metronidazole was administered orally to pregnant mice at 10 mg/kg/day, approximately 5 percent of the indicated human dose (1500 mg/day) based on body surface area; however in a single small study where the drug was administered intraperitoneally, some intrauterine deaths were observed. The relationship of these findings to the drug is unknown.
Bismuth subsalicylate
Animal reproductive studies have not been conducted with bismuth subsalicylate. It is also not known whether bismuth subsalicylate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
Labor and Delivery: The effect of this therapy on labor and delivery is unknown.
Nursing Mothers: HELIDAC Therapy contains bismuth subsalicylate, metronidazole, and tetracycline hydrochloride. Metronidazole is present in human milk at concentrations similar to maternal serum levels, and infant serum levels can be close to or comparable to infant therapeutic levels. Because of the potential for tumorigenicity shown for metronidazole in animal studies, a decision should be made whether to discontinue nursing or to discontinue metronidazole, taking into account the importance of the therapy to the mother. Alternatively, a nursing mother may choose to pump and discard human milk for the duration of HELIDAC Therapy, and for 24 hours after therapy ends and feed her infant stored human milk or formula.
Tetracycline is present in human milk at concentrations similar to maternal serum levels; however, it binds with calcium in human milk. Data indicate that oral absorption of tetracycline in infants is low due to the calcium binding in human milk. It is not known whether bismuth subsalicyate is excreted in human milk.
Pediatric Use: Safety and effectiveness of HELIDAC Therapy in pediatric patients infected with H. pylori have not been established.
Tetracycline use in children may cause permanent discoloration of the teeth. Enamel hypoplasia has also been reported. HELIDAC Therapy should not be used in children up to 8 years of age (see WARNINGS).
Geriatric Use: Clinical studies of HELIDAC Therapy did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, elderly patients may have a greater frequency of decreased hepatic, renal, or cardiac function, and concomitant diseases or other drug therapies. Bismuth subsalicylate, a component of HELIDAC Therapy, is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, additional monitoring may be required (see CONTRAINDICATIONS).
Renal Impairment: The antianabolic action of the tetracyclines may cause an increase in blood urea nitrogen (BUN). While this is not a problem in those with normal renal function, in patients with significantly impaired renal function, higher serum levels of tetracycline may lead to azotemia, hyperphosphatemia, and acidosis (see CONTRAINDICATIONS).
Hepatic Impairment: Patients with severe hepatic disease metabolize metronidazole slowly, with resultant accumulation of metronidazole and its metabolites in plasma. Use HELIDAC Therapy with caution in patients with hepatic impairment (see PRECAUTIONS).
Adverse Reactions/Side Effects
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions (≥ 1%) reported in clinical trials when all three components of this therapy were given concomitantly are listed in Table 1 below. The majority of the adverse reactions were related to the gastrointestinal tract, were reversible, and infrequently led to discontinuation of therapy.
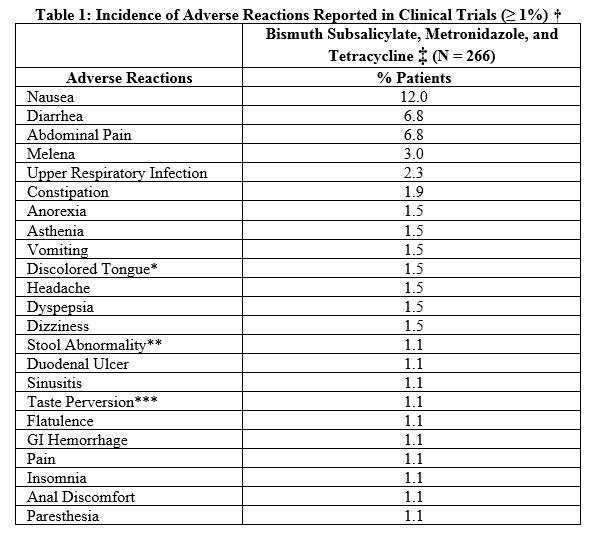
* darkening of the tongue
** black or dark stools
*** metallic taste
† Includes reactions reported at ≥1% in patients taking bismuth subsalicylate, metronidazole, and tetracycline in Studies 1, 2, 3, and 4 (see CLINICAL STUDIES).
‡ In Studies 1, 2, and 3(N = 197), most patients were on concomitant acid suppression therapy.
The additional adverse reactions (< 1%) reported in clinical trials when all three components of this therapy were given concomitantly are listed below and divided by body system:
Gastrointestinal: dry mouth, dysphagia, eructation, GI monilia, glossitis, intestinal obstruction, rectal hemorrhage, stomatitis
Skin: acne, ecchymosis, photosensitivity reaction (see PRECAUTIONS), pruritus, rash
Cardiovascular: cerebral ischemia, chest pain, hypertension, myocardial infarction
CNS: nervousness, somnolence
Musculoskeletal: arthritis, rheumatoid arthritis, tendonitis
Metabolic: SGOT increase, SGPT increase
Urogenital: urinary tract infection
Other: conjunctivitis, flu syndrome, infection, malaise, neoplasm, rhinitis, syncope, tooth disorder
Other Important Adverse Reactions from Labeling for the Individual Components of HELIDAC Therapy
Metronidazole
Blood and Lymphatic system disorders: Reversible neutropenia (leucopenia) in cases of prolonged treatment; rarely reversible thrombocytopenia however no persistent hematological abnormalities attributable to metronidazole have been observed (see PRECAUTIONS).
Cardiac disorders: Flattening of the T-wave may be seen in electrocardiographic tracings.
Gastrointestinal disorders: Nausea, vomiting, diarrhea, abdominal pain, constipation, anorexia, metallic taste, furry tongue, glossitis, stomatitis and candida overgrowth (see PRECAUTIONS).
Hypersensitivity/Immune system disorders: Urticaria, erythematous rash, Stevens-Johnson syndrome, toxic epidermal necrolysis, flushing, nasal congestion, dryness of the mouth (or vagina or vulva), and fever (see CONTRAINDICATIONS).
Metabolism and nutrition disorders: Pancreatitis.
Nervous system disorders: Convulsive seizures, encephalopathy, aseptic meningitis, optic and peripheral neuropathy, headache, syncope, dizziness, vertigo, incoordination, ataxia, confusion, dysarthria, irritability, depression, weakness, and insomnia (see WARNINGS).
Dermatologic disorders: Erythematous rash and pruritus.
Renal and urinary disorders: Dysuria, cystitis, polyuria, incontinence, darkened urine, and a sense of pelvic pressure.
Other: Dyspareunia, decrease of libido, proctitis, joint pains.
Tetracycline Hydrochloride
Blood and lymphatic system disorders: Hemolytic anemia, thrombocytopenia, thrombocytopenic purpura, neutropenia, and eosinophilia.
Gastrointestinal disorders: Nausea, vomiting, diarrhea, anorexia, glossitis, black hairy tongue, dysphagia, enterocolitis, inflammatory lesions (with Candida overgrowth) in the anogenital region, esophagitis and esophageal ulceration.
Nervous system disorders: Intracranial hypertension including pseudotumor cerebri, tinnitus, and myasthenic syndrome.
Renal and urinary disorders: Increased BUN.
Skin and subcutaneous tissue disorders: Maculopapular and erythematous rashes, DRESS syndrome (drug rash with eosinophilia and systemic symptoms), Steven-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, onycholysis, discoloration of the nails, exfoliative dermatitis and photosensitivity have been rarely reported (see WARNINGS).
Liver: Hepatotoxicity and liver failure.
Hypersensitivity reactions: Urticaria, angioedema, anaphylaxis, Henoch-Schonlein purpura, pericarditis, exacerbation of systemic lupus erythematosus, and serum sickness-like reactions.
To report SUSPECTED ADVERSE REACTIONS, contact Casper Pharma LLC. at 1-844-5-CASPER (1-844-522-7737) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Related/similar drugs
Overdosage
In case of an overdose, patients should contact a physician, poison control center, or emergency room.
Bismuth Subsalicylate: The main concern of an acute bismuth subsalicylate (BSS) overdose focuses on the salicylate burden and not on bismuth, since less than 1% of the bismuth is normally absorbed. Each 262.4-mg tablet of BSS contains an amount of salicylate comparable to approximately 130 mg aspirin. Acute ingestion of less than 150 mg/kg of aspirin (i.e., less than one tablet of bismuth subsalicylate per kilogram of body weight) is not expected to lead to toxicity. Mild to moderate toxicity may result from the ingestion of 150 to 300 mg/kg, while severe toxicity may occur from ingestions over 300 mg/kg. Salicylate intoxication presents a complex clinical picture. Multiple respiratory and metabolic effects result in fluid, electrolyte, glucose, and acid-base disturbances. Initial symptoms of salicylate toxicity include hyperpnea, nausea, vomiting, tinnitus, hyperpyrexia, lethargy, tachycardia, and confusion. In severe cases, these symptoms may progress to severe hyperpnea, convulsions, pulmonary or cerebral edema, respiratory failure, cardiovascular collapse, coma, and death.
Treatment: There is no specific antidote for salicylate poisoning. If there are no contraindications, vomiting should be induced as soon as possible with syrup of ipecac, or gastric lavage should be instituted, provided that no more than one hour has elapsed since ingestion. Activated charcoal and a cathartic may be administered as primary decontamination therapy in those cases where greater than one hour has elapsed since ingestion, or to further decontaminate the gastrointestinal tract in those who have already received ipecac or gastric lavage. Plasma salicylate levels may be useful; a common nomogram can be used to help predict the severity of intoxication. Supportive and symptomatic treatment should be provided, with emphasis on correcting fluid, electrolyte, blood glucose, and acid-base disturbances. Elimination may be enhanced by urinary alkalinization, hemodialysis, or hemoperfusion. Since hemodialysis aids in correcting acid-base disturbances, this method may be preferred over hemoperfusion.
Metronidazole: Symptoms of metronidazole toxicity include nausea, vomiting, and neurotoxic effects such as ataxia, seizures and peripheral neuropathy.
Treatment: There is no specific antidote for metronidazole overdose. Management of the patient should consist of symptomatic and supportive therapy. Metronidazole is dialyzable.
Tetracycline: Therapeutic and overdose quantities of tetracycline can cause gastrointestinal symptoms such as nausea, vomiting, and diarrhea.
Treatment: There is no specific antidote for tetracycline overdose. Management of the patient should consist of symptomatic and supportive therapy. Tetracycline is not dialyzable.
Helidac Dosage and Administration
Adults: The recommended dosages are: bismuth subsalicylate, 525 mg (two 262.4 mg-chewable tablets), metronidazole, 250 mg (one 250-mg tablet), and tetracycline hydrochloride, 500 mg (one 500-mg capsule) taken four times daily for 14 days plus an H2 antagonist approved for the treatment of acute duodenal ulcer. Patients should be instructed to take the medicines at mealtimes and at bedtime. The bismuth subsalicylate tablets should be chewed and swallowed. The metronidazole tablet and tetracycline hydrochloride capsule should be swallowed whole with a full glass of water (8 ounces). Concomitantly prescribed H2 antagonist therapy should be taken as directed.
Ingestion of adequate amounts of fluid, particularly with the bedtime dose of tetracycline hydrochloride, is recommended to reduce the risk of esophageal irritation and ulceration (see ADVERSE REACTIONS).
Missed doses can be made up by continuing the normal dosing schedule until the medication is gone. Patients should not take double doses. If more than 4 doses are missed, the prescriber should be contacted.
How is Helidac supplied
The HELIDAC Therapy is supplied in a carton containing patient instructions, patient reminders, and 14 blister cards, each card containing the following daily dosage:
8 bismuth subsalicylate 262.4-mg chewable tablets, each pink round tablet debossed “W10”.
4 metronidazole 250-mg tablets are round, white, debossed with “WPI” on one side and “3969” on the other.
4 tetracycline hydrochloride 500-mg capsules, each black/yellow capsule imprinted with “WPI” on cap and “2235” on the body.
NDC 70199-021-14 carton containing 14 days of therapy
Store at 20° to 25° C (68° to 77° F). [See USP Controlled Room Temperature.].
Clinical Studies
Eradication of H. pylori in Patients with Active Duodenal Ulcer Disease: Three clinical trials in the U.S. (Studies 1, 2 and 3) evaluated the effect of therapy on the eradication of H. pylori using bismuth subsalicylate, metronidazole, and tetracycline hydrochloride. The patient population in these studies consisted predominantly of duodenal ulcer patients with active disease. In addition to bismuth subsalicylate, metronidazole, tetracycline hydrochloride triple therapy, most patients were also prescribed antisecretory therapy at doses recommended for ulcer healing, with the majority receiving ranitidine. The efficacy in these studies was assessed using H. pylori eradication, or cure of infection. Use of cure of infection as a surrogate for reduced ulcer recurrence is based on an extensive review of the literature. Eradication rates are derived from results of Studies 1 (randomized, controlled study) and Study 3 (uncontrolled, nonrandomized) and are shown in Table 2. H. pylori eradication was defined as no positive test (culture, histology, rapid urease, or 13C breath test) at least 4 weeks following the end of treatment. In the analysis performed, dropouts and patients with missing H. pylori tests post-treatment were excluded. HELIDAC Therapy (bismuth subsalicylate, metronidazole, and tetracycline hydrochloride) was effective in eradicating H. pylori.
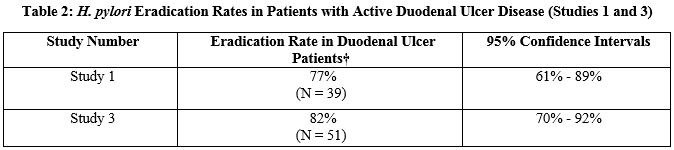
† Evaluable patients were defined as having a confirmed duodenal ulcer within 2 years prior to treatment and having taken 14 days of bismuth subsalicylate, metronidazole, and tetracycline (range 11 to 17 days). Eradication was defined as no evidence of H. pylori infection by culture, histology, rapid urease test and/or urea breath test from at least 4 weeks post-treatment up to 1 year posttreatment.
Study 2 evaluated the long-term outcome in patients treated for active duodenal ulcer by frequently monitoring for ulcer recurrence for up to 1 year after therapy. This study compared patients who received bismuth subsalicylate (BSS), metronidazole (MTZ), and tetracycline hydrochloride (TCN) for 2 weeks with ranitidine to those who received ranitidine alone. The ulcer recurrence rates at 6 months (Table 3) and one year (Table 4) regardless of post-treatment eradication status are summarized below for duodenal ulcer patients who were H. pylori positive at baseline.
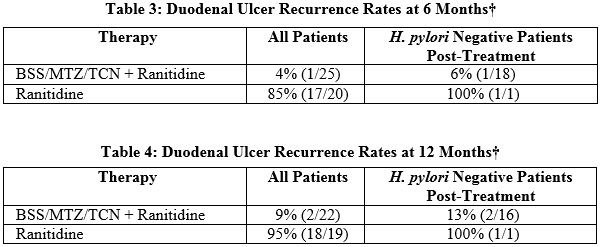
† Includes all patients randomized to therapy who were H. pylori positive at baseline (by culture, histology, and/or urea breath test) who had ulcer healing and 24 or 48 weeks of endoscopic follow-up data.
Eradication of H. pylori in Patients with a History of Duodenal Ulcer Disease: A controlled, multicenter trial (Study 4) in the U.S. compared the rates of eradication of H. pylori following 14 days of treatment with HELIDAC Therapy (bismuth subsalicylate, metronidazole, and tetracycline hydrochloride) or control (bismuth subsalicylate, metronidazole placebo, and tetracycline placebo) in 103 patients infected with H. pylori who had a history of duodenal ulcer disease. No H2-receptor antagonist was used. H. pylori eradication was assessed by rapid urease testing, histology, and culture at least 4 weeks after the last dose. HELIDAC Therapy was effective in eradicating H. pylori. The eradication rates are noted in the table below:
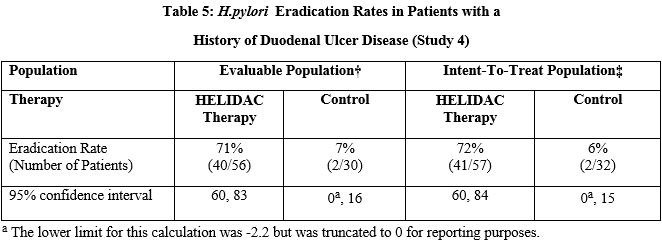
† Evaluable patients were defined as having a history of a confirmed duodenal ulcer prior to treatment and having taken ≥ 70% of each component of HELIDAC Therapy (bismuth subsalicylate, metronidazole, tetracycline hydrochloride) during the 14-day dosing period. Eradication was defined as no evidence of H. pylori infection by two or three diagnostic tests (culture, histology, and rapid urease test) from at least 4 weeks to 6 weeks post-treatment. Excluded from the evaluable population were patients who were noncompliant with medication, patients who were not infected with H. pylori at baseline, and patients without ulcer documentation. Three patients in the HELIDAC Therapy group and one patient in the Control group were included as eradication failures because they withdrew from the study due to treatment-related adverse events.
‡ Patients were included in the analysis if they had documented H. pylori infection at baseline and had confirmed duodenal ulcer disease. All dropouts were included as failures of therapy. Eradication was defined as no evidence of H. pylori infection by culture, histology, or rapid urease test.
Compliance with the triple therapy regimen was also evaluated in the clinical study. In the intent-to-treat population, 93% of the HELIDAC Therapy group took at least 75% of their medication.
HELIDAC is a registered trademark of Casper Pharma LLC.
PEPTO-BISMOL is the registered trademark of The Procter & Gamble Company.
Bismuth Subsalicylate Chewable Tablets, 262.4 mg are manufactured by:
WES Pharma Inc
Westminster, MD 21157 USA
Metronidazole Tablets USP, 250 mg are manufactured by:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722 INDIA
Tetracycline Hydrochloride Capsules USP, 500 mg are manufactured by:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722 INDIA
for
Casper Pharma LLC.
East Brunswick, NJ 08816
Issued: 08/2019
PIB02114-01
PHARMACIST INFORMATION AND COUNSELING AID
HELIDAC® Therapy
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Bismuth Subsalicylate Chewable Tablets/Metronidazole Tablets/Tetracycline Hydrochloride Capsules and other antibacterial drugs, Bismuth Subsalicylate Chewable Tablets/Metronidazole Tablets/Tetracycline Hydrochloride Capsules should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Casper Pharma LLC.
PHARMACIST INFORMATION AND COUNSELING AID
Not intended for distribution to Patient
• This Counseling Aid contains concise information for the Pharmacist and is meant to aid in counseling; please refer to the attached Package Insert for complete prescribing information.
• There is a detailed PATIENT INFORMATION BOOKLET contained in this package which should be read by the patient prior to initiating therapy.
• Instructions for opening the enclosed dosing blister cards can be found on the top of this package.
HELIDAC Therapy
bismuth subsalicylate
metronidazole tablet
tetracycline hydrochloride capsule
1. What is the most important information my patient should know about HELIDAC Therapy?
a. Scientists have discovered that most ulcers are caused by an infection from a specific germ called H. pylori. Helicobacter pylori (H. pylori) infection has been found to play a primary role in the pathogenesis of duodenal ulcers, and is considered the causative organism in 80% to 95% of patients with duodenal ulcers.
b. Each pill of every dose is important. Studies have shown that taking all this medicine is very important to eradicate all the ulcer-causing germs. If doses are skipped or treatment is stopped early, some of these germs may not be eradicated and another ulcer can develop.
2. What is HELIDAC Therapy?
a. The components of HELIDAC Therapy (bismuth subsalicylate, metronidazole, and tetracycline hydrochloride) are indicated for the treatment of patients with an active duodenal ulcer associated with H. pylori infection. The eradication of H. pylori has been demonstrated to reduce the risk of duodenal ulcer recurrence. Appropriate doses of an H2 antagonist indicated for the treatment of active duodenal ulcer should be prescribed for ulcer healing.
b. HELIDAC Therapy is a combination of three antimicrobial agents: bismuth subsalicylate, metronidazole, and tetracycline hydrochloride. Clinical trials using this combination have demonstrated H. pylori eradication in up to 82% of patients with a duodenal ulcer. Please note: bismuth subsalicylate is used in this combination for its antimicrobial properties and not for symptomatic relief of upset stomach.
c. Clinical data show that eradication of H. pylori is directly related to compliance. This therapy has been designed to enhance compliance and contains 14 blister cards, one for each day of the 14-day treatment plan, with each card divided into the 4 daily doses. The therapy also contains a patient-oriented information booklet and patient reminders.
3. Who should not take HELIDAC Therapy?
HELIDAC Therapy is contraindicated in the following patient populations:
a. Pregnant women. Advise pregnant women that HELIDAC Therapy can cause fetal harm due to the tetracycline it contains. Tetracycline use during the second and third trimesters of pregnancy can cause permanent discoloration of the teeth (yellow-gray brown) and may inhibit bone development.
b. Nursing women. Advise women to avoid feeding their infants with their milk during HELIDAC Therapy. Women should either discontinue feeding or pump and discard their milk during treatment and for 24 hours after the last dose.
c. Pediatric patients.
d. Patients with renal or hepatic impairment.
e. Patients with known hypersensitivity to bismuth subsalicylate, metronidazole or other nitroimidazole derivatives, or any of the tetracyclines; this product does not contain aspirin, but should not be administered to those patients who have a known allergy to aspirin or salicylates.
f. Patients with evidence of, or a history of, blood dyscrasias.
g. Administer with caution in patients with central nervous system diseases.
h. Administer with caution in elderly patients who may suffer from asymptomatic renal and hepatic dysfunction.
4. How should my patient take HELIDAC Therapy?
The medicines in this therapy have been demonstrated to treat the infection in the majority of cases when taken correctly. Treating the infection is important to reduce the risk of the ulcer coming back. (It may be useful to open the therapy and explain dosing while showing the patient one of the dosing blister cards.) Patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by HELIDAC Therapy or other antibacterial drugs in the future.
a. Dosing:
i. Each dose includes 4 pills: 2 pink round chewable tablets (bismuth subsalicylate), 1 white round tablet (metronidazole), and 1 black and yellow capsule (tetracycline hydrochloride).
ii. Take 1 dose (all 4 pills) 4 times a day – at mealtimes and bedtime.
iii. Chew and swallow the 2 pink tablets.
iv. Then swallow the white tablet and black and yellow capsule whole with a full glass of water (8 ounces).
v. Remember: Chew-Chew-Swallow-Swallow = 1 dose.
vi. All 4 daily doses are contained on an individual blister card.
vii. This therapy contains 14 blister cards, one card for each day of the 14-day treatment plan.
viii. If you miss a dose, do not take a double dose. Instead, make up any missed doses by continuing your normal dosing schedule until the medication is gone. (Please contact prescriber if more than 4 doses are missed.)
ix. Each dose should be taken with a full glass of water (8 ounces), especially the bedtime dose (to reduce risk of esophageal irritation and ulceration due to tetracycline).
x. Concomitantly prescribed H2 antagonist therapy should be taken as directed.
b. In case of an overdose, patients should contact a physician, poison control center, or emergency room.
5. What should my patients avoid while taking HELIDAC Therapy?
Patients should avoid:
a. Methoxyflurane (an anaesthesia). Concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
b. Disulfiram (treatment for alcoholism) within the last 2 weeks. Psychotic episodes have been reported in patients using metronidazole and disulfiram concurrently.
c. Alcohol. There is a potential for a disulfiram-like interaction with metronidazole. Patients should be warned not to drink alcohol or products containing propylene glycol during therapy and for at least 3 days after completion of all doses due to potential metronidazole effects.
d. Antacids containing aluminum, calcium, or magnesium; preparations containing iron, zinc, or sodium bicarbonate; and milk or dairy products. Absorption of tetracycline may be impaired when co-administered.
e. The sun (or sunlamps) because of possible photosensitivity due to tetracycline.
f. Other medications without first consulting with your pharmacist or prescriber.
6. The administration of any of the following drugs with HELIDAC Therapy may result in adverse reactions or insufficient drug efficacies:
a. Oral contraceptives. As with many antibacterial preparations, concomitant use of HELIDAC Therapy may reduce the effectiveness of estrogen-containing oral contraceptives; breakthrough bleeding may occur. Patients should be advised to use an additional or different form of contraception. Women who become pregnant while taking HELIDAC Therapy should be advised to notify their prescriber immediately.
b. Anticoagulants. Individual components of HELIDAC Therapy have the potential to interact with anticoagulants. Monitoring anticoagulant therapy with appropriate adjustment of the anticoagulant dosage may be warranted if concurrent therapy is instituted. (See Package Insert.)
c. Lithium. In patients stabilized on relatively high doses of lithium, short-term metronidazole therapy has been associated with elevation of serum lithium and, in a few cases, signs of lithium toxicity. Serum lithium and serum creatinine should be obtained several days after beginning metronidazole to detect any increase that may precede clinical symptoms of lithium toxicity.
d. Antidiabetic agents. There is a possible enhanced hypoglycemic effect when given with salicylates.
e. Cimetidine. The simultaneous administration of drugs that decrease microsomal liver enzyme activity, such as cimetidine, may prolong the half-life and decrease plasma clearance of metronidazole.
f. Others. See the Package Insert regarding the following possible, suspected, or confirmed interactions: aspirin, probenecid, sulfinpyrazone, methoxyflurane, disulfiram (within 2 weeks), lithium, microsomal liver enzyme inducers, such as phenytoin or phenobarbital, and microsomal liver enzyme inhibitors.
g. Although there is an anticipated reduction in tetracycline systemic absorption due to an interaction with bismuth and/or bismuth subsalicylate tablet excipients (calcium carbonate), the relative contribution of systemic versus local antimicrobial activity against H. pylori for these agents has not been established.
7. What are possible or reasonably likely side effects of HELIDAC Therapy?
Adverse reactions associated with the individual components of HELIDAC Therapy have been well described and are included in the Package Insert. The most common adverse reactions (≥1.5%) are nausea (12.0%), diarrhea (6.8%), abdominal pain (6.8%), melena (3.0%), upper respiratory infection (2.3%), constipation (1.9%), anorexia (1.5%), asthenia (1.5%), vomiting (1.5%), discolored tongue (1.5%), headache (1.5%), dyspepsia (1.5%), and dizziness (1.5%). The majority of the events were related to the gastrointestinal tract, were reversible, and usually did not lead to discontinuation of therapy. Some other possible adverse reactions include:
a. Bismuth subsalicylate may cause a temporary and harmless darkening of the tongue and/or black stool. This should not be confused with melena (blood in the stool).
b. Metallic taste in the mouth
c. Temporary darkening of the urine (rare)
d. Sore mouth
e. Yeast infections in women
f. Tooth enamel discoloration and hypoplasia (spots on teeth). Use of tetracycline during tooth development (second and third trimesters of pregnancy, infancy, and childhood to 8 years) may cause permanent discoloration of the teeth.
g. Superinfections. Use of antibiotics may result in overgrowth of nonsusceptible organisms, including fungi.
h. Central Nervous Effects. All of the components of HELIDAC Therapy have been associated with central nervous effects such as seizures, peripheral neuropathy, neurotoxicity and intracranial hypertension.
As with most medications, rare but serious side effects have been reported with the use of the components contained in HELIDAC Therapy. (See Package Insert.) Patients should be instructed to report any unusual symptoms to their pharmacist or prescriber. Patients may also report side effects to FDA at 1-800-FDA-1088 or Casper Pharma LLC. at 1-844-5-CASPER (1-844-522-7737).
8. General Information about the safe and effective use of HELIDAC Therapy Medicines are sometimes prescribed for purposes other than those listed. If your patients have any questions or concerns about HELIDAC Therapy, ask them to consult their doctor. Tell your patients not to use HELIDAC Therapy for a condition for which it was not prescribed. Tell them not to share their medicine with other people.
Bismuth Subsalicylate Chewable Tablets, 262.4 mg are manufactured by:
WES Pharma Inc
Westminster, MD 21157 USA
Metronidazole Tablets USP, 250 mg are manufactured by:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722 INDIA
Tetracycline Hydrochloride Capsules USP, 500 mg are manufactured by:
Watson Pharma Private Limited
Verna, Salcette Goa 403 722 INDIA
for
Casper Pharma LLC.
East Brunswick, NJ 08816
Issued: 08/2019
MGB02114-00
| HELIDAC THERAPY
bismuth subsalicylate chewable tablets/metronidazole tablets/tetracycline hydrochloride capsules kit |
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Casper Pharma LLC (080025838) |
More about Helidac (bismuth subsalicylate / metronidazole / tetracycline)
- Check interactions
- Compare alternatives
- Reviews (2)
- Side effects
- Dosage information
- During pregnancy
- Drug class: H. pylori eradication agents
- En español