Auranofin
Medically reviewed by Drugs.com. Last updated on Jun 18, 2025.
Pronunciation
(au RANE oh fin)
Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Capsule, Oral:
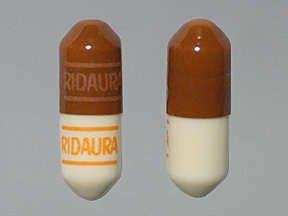
Ridaura: 3 mg [contains brilliant blue fcf (fd&c blue #1), fd&c red #40, fd&c yellow #6 (sunset yellow)]
Brand Names: U.S.
- Ridaura
Pharmacologic Category
- Gold Compound
Pharmacology
The exact mechanism of action of gold is unknown; gold salts decrease cellular proliferation, reduce antibody and cytokine release, and inhibit collagenase action (Doan 2005).
Absorption
Oral: ~25% gold in dose is absorbed
Metabolism
Rapid; intact auranofin not detectable in the blood
Excretion
Urine (~60% of absorbed gold); remainder in feces
Onset of Action
Delayed; therapeutic response may not be seen for 3-4 months after start of therapy, as long as 6 months in some patients
Time to Peak
Serum: ~2 hours (Blocka 1986)
Duration of Action
Prolonged
Half-Life Elimination
Single or multiple dosing dependent: 21-31 days
Protein Binding
60%
Use: Labeled Indications
Rheumatoid arthritis: Management of adult patients with active stage classic or definite rheumatoid arthritis in patients who do not respond to or tolerate an adequate trial of full doses of one or more nonsteroidal anti-inflammatory drugs (NSAIDs)
Contraindications
History of gold-induced disorders, including anaphylactic reactions, bone marrow aplasia, severe hematologic disorders, exfoliative dermatitis, necrotizing enterocolitis, or pulmonary fibrosis.
Dosing: Adult
Rheumatoid arthritis: Oral: Initial: 6 mg/day in 1-2 divided doses; after 6 months may be increased to 9 mg/day in 3 divided doses; discontinue therapy if no response after 3 months at 9 mg/day
Note: Signs of clinical improvement may not be evident until after 3 months of therapy.
Dosing: Geriatric
Refer to adult dosing.
Dosing: Pediatric
Juvenile idiopathic arthritis: Limited data available: Note: Reserve for patients intolerant to or unresponsive to other therapies. Children ≥18 months and Adolescents ≤17 years: Oral: Initial: 0.1 to 0.15 mg/kg/day in 1 to 2 divided doses; usual maintenance: 0.15 mg/kg/day in 1 to 2 divided doses; maximum daily dose: 0.2 mg/kg/day or 9 mg/day. Note: With current market availability, may not be able to replicate the exact dosing used in trials; in trials, doses were rounded to nearest whole mg using 1 mg tablet (no longer available) (Brewer 1983; Giannini 1990; Giannini 1991; Marcolongo 1988).
Storage
Store at 15°C to 30°C (59°F to 86°F). Protect from light.
Drug Interactions
There are no known significant interactions.
Adverse Reactions
The following adverse drug reactions and incidences are derived from product labeling unless otherwise specified.
>10%:
Dermatologic: Skin rash (24%), pruritus (17%)
Gastrointestinal: Diarrhea (≤47%), loose stools (≤47%), abdominal pain (14%), stomatitis (13%)
1% to 10%:
Dermatologic: Alopecia (1% to 3%), urticaria (1% to 3%)
Gastrointestinal: Nausea (10%), vomiting (10%), anorexia (3% to 9%), dyspepsia (3% to 9%), flatulence (3% to 9%), constipation (1% to 3%), dysgeusia (1% to 3%), glossitis (1% to 3%)
Genitourinary: Proteinuria (3% to 9%), hematuria (1% to 3%)
Hematologic and Oncologic: Anemia (1% to 3%), eosinophilia (1% to 3%), leukopenia (1% to 3%), thrombocytopenia (1% to 3%)
Hepatic: Increased serum transaminases (1% to 3%)
Ophthalmic: Conjunctivitis (3% to 9%)
<1%, postmarketing, and/or case reports: Agranulocytosis, aplastic anemia, angioedema, bronchitis (gold), corneal deposits, dysphagia, exfoliative dermatitis (other gold compounds), fever, gastrointestinal hemorrhage, gingivitis, hepatotoxicity, interstitial pneumonitis, jaundice, metallic taste, melena, neutropenia, pancytopenia, peripheral neuropathy, pure red cell aplasia, ulcerative enterocolitis
Related/similar drugs
ALERT: U.S. Boxed Warning
Gold toxicities:Auranofin contains gold and, like other gold-containing drugs, can cause gold toxicity, signs of which include: Fall in hemoglobin, leukopenia <4,000 WBC/mm3, granulocytes <1,500/mm3, decrease in platelets <150,000/mm3, proteinuria, hematuria, pruritus, rash, stomatitis or persistent diarrhea.
Appropriate use:Like other gold preparations, auranofin is only indicated for use in selected patients with active rheumatoid arthritis.
Experienced physician:Physicians planning to use auranofin should be experienced with chrysotherapy and should thoroughly familiarize themselves with the toxicity and benefits of auranofin.
Monitoring:The results of recommended laboratory work should be reviewed before writing each auranofin prescription. In addition, the following precautions should be routinely employed: The possibility of adverse reactions should be explained to patients before starting therapy and patients should be advised to report promptly any symptoms suggesting toxicity.
Warnings/Precautions
Concerns related to adverse effects:
• Dermatologic reactions: [US Boxed Warning]: Signs of gold toxicity include pruritus, rash, and stomatitis. Stomatitis and dermatitis are common reactions and may be serious (including exfoliative dermatitis). Pruritus and metallic taste may occur before dermatitis and oral mucous membrane reactions, respectively, and should be considered warning signs. Stomatitis may be manifested by shallow ulcers on the buccal membranes, on border of tongue, and on palate or in the pharynx; sometimes diffuse glossitis or gingivitis develops. Gold dermatitis may be aggravated by sunlight exposure or an actinic rash may develop. Use with caution in patients with skin rash; may increase risk and/or symptoms of gold toxicity and toxicity may be more difficult to detect.
• Gastrointestinal effects: [US Boxed Warning]: Signs of gold toxicity include persistent diarrhea/loose stools, as well as nausea, vomiting, anorexia, abdominal cramps, and ulcerative enterocolitis (rare). Diarrhea may be managed with a dose reduction. Use with caution in patients with inflammatory bowel disease; may increase risk and/or symptoms of gold toxicity and toxicity may be more difficult to detect. Monitor patients for GI bleeding.
• Hematologic effects: [US Boxed Warning]: Signs of gold toxicity include hematologic depression (decreased hemoglobin, leukocytes (WBC <4000/mm3), granulocytes (<1500/mm3), or decreased platelets (<150,000/mm3). Use with caution in patients with bone marrow depression; may increase risk and/or symptoms of gold toxicity and toxicity may be more difficult to detect. Therapy should be discontinued if signs and symptoms (eg, purpura, ecchymoses, petechiae) of thrombocytopenia develop or if platelet count falls to <100,000/mm3; therapy should not be reinitiated until thrombocytopenia resolves and if thrombocytopenia was not caused by the gold therapy.
• Hepatic effects: May be associated with the development of cholestatic jaundice (rare). Use with caution in patients with hepatic impairment; may increase risk and/or symptoms of gold toxicity and toxicity may be more difficult to detect.
• Pulmonary toxicity: May be associated with rare reactions, including gold bronchitis, interstitial pneumonitis and fibrosis; monitor closely.
• Renal effects: [US Boxed Warning]: Signs of gold toxicity include proteinuria and hematuria. Renal toxicity ranges from mild proteinuria to nephrotic syndrome or glomerulitis with proteinuria and hematuria. Use with caution in patients with renal impairment; may increase risk and/or symptoms of gold toxicity and toxicity may be more difficult to detect. Therapy should be discontinued promptly if proteinuria or microscopic hematuria develops.
Dosage form specific issues:
• Benzyl alcohol and derivatives: Some dosage forms may contain benzyl alcohol; large amounts of benzyl alcohol (≥99 mg/kg/day) have been associated with a potentially fatal toxicity (“gasping syndrome”) in neonates; the “gasping syndrome” consists of metabolic acidosis, respiratory distress, gasping respirations, CNS dysfunction (including convulsions, intracranial hemorrhage), hypotension and cardiovascular collapse (AAP ["Inactive" 1997]; CDC 1982); some data suggests that benzoate displaces bilirubin from protein binding sites (Ahlfors 2001); avoid or use dosage forms containing benzyl alcohol with caution in neonates. See manufacturer’s labeling.
Other warnings/precautions:
• Appropriate use: [US Boxed Warning]: Use is only indicated in select patients with active rheumatoid arthritis.
• Experienced physician: [US Boxed Warning]: Physicians should be experienced with chrysotherapy and should be familiar with the toxicity and benefits of therapy.
• Monitoring: [US Boxed Warning]: Laboratory monitoring should be reviewed prior to each new prescription. The possibility of adverse reactions should be discussed with patients prior to starting therapy and patients should be advised to promptly report any symptoms suggesting toxicity. Disease states should be stable prior to initiating therapy.
Monitoring Parameters
Patients should have baseline CBC with differential, platelet count, renal function tests, liver function tests, and urinalysis; CBC with differential, platelet count and urinalysis should also be monitored monthly during therapy. Skin and oral mucosa should be inspected for skin rash, bruising or oral ulceration/stomatitis. Specific questioning for symptoms such as pruritus, rash, stomatitis or metallic taste should be included.
Pregnancy Risk Factor C Pregnancy Considerations
Adverse events were observed in animal reproduction studies.
Patient Education
What is this drug used for?
• It is used to treat rheumatoid arthritis.
All drugs may cause side effects. However, many people have no side effects or only have minor side effects. Call your doctor or get medical help if any of these side effects or any other side effects bother you or do not go away:
• Lack of appetite
• Nausea
• Vomiting
• Abdominal cramps
• Passing gas
• Mouth irritation
WARNING/CAUTION: Even though it may be rare, some people may have very bad and sometimes deadly side effects when taking a drug. Tell your doctor or get medical help right away if you have any of the following signs or symptoms that may be related to a very bad side effect:
• Kidney problems like unable to pass urine, blood in the urine, change in amount of urine passed, or weight gain.
• Severe abdominal pain
• Skin irritation
• Severe loss of strength and energy
• Bruising
• Bleeding
• Blood in the urine
• Itching
• Rash
• Severe diarrhea
• Infection
• Black, tarry, or bloody stools
• Vomiting blood
• Vision changes
• Eye pain
• Severe eye irritation
• Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing, swallowing, or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a limited summary of general information about the medicine's uses from the patient education leaflet and is not intended to be comprehensive. This limited summary does NOT include all information available about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not intended to provide medical advice, diagnosis or treatment and does not replace information you receive from the healthcare provider. For a more detailed summary of information about the risks and benefits of using this medicine, please speak with your healthcare provider and review the entire patient education leaflet.
More about auranofin
- Check interactions
- Compare alternatives
- Reviews (2)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: antirheumatics
- Breastfeeding
- En español
Patient resources
Other brands
Professional resources
Other brands
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.