GG 431 Pill - red round, 8mm
Pill with imprint GG 431 is Red, Round and has been identified as Amitriptyline Hydrochloride 50 mg. It is supplied by Sandoz Pharmaceuticals Inc.
Amitriptyline is used in the treatment of Chronic Pain; Depression; Headache; Migraine and belongs to the drug class tricyclic antidepressants. Risk cannot be ruled out during pregnancy. Amitriptyline 50 mg is not a controlled substance under the Controlled Substances Act (CSA).
Images for GG 431






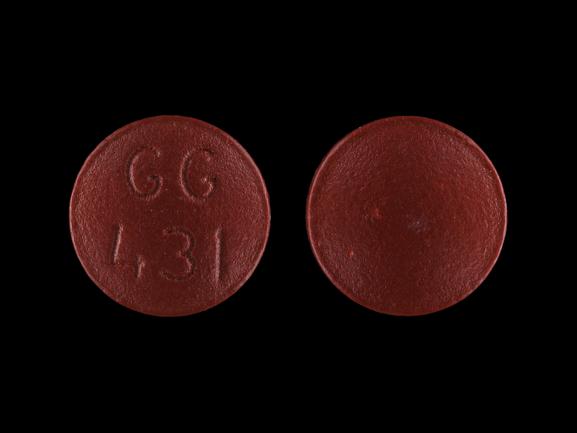

Amitriptyline Hydrochloride
- Imprint
- GG 431
- Strength
- 50 mg
- Color
- Red
- Size
- 8.00 mm
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Tricyclic antidepressants
- Pregnancy Category
- C - Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Sandoz Pharmaceuticals Inc.
- Inactive Ingredients
-
microcrystalline cellulose,
D&C Yellow No. 10,
FD&C Blue No. 2,
FD&C Red No. 40,
hydroxypropyl cellulose,
hypromelloses,
lactose monohydrate,
magnesium stearate,
polyethylene glycol,
silicon dioxide,
corn starch,
titanium dioxide
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 00781-1488 | Sandoz Inc. |
| 00904-0202 (Discontinued) | Major Pharmaceuticals |
| 16714-0448 | NorthStar Rx LLC |
| 33261-0358 | Aidarex Pharmacuticals, LLC (repackager) |
| 54569-1519 (Discontinued) | A-S Medication Solutions, LLC (repackager) |
| 63874-0359 (Discontinued) | Altura Pharmaceuticals Inc. (repackager) |
See also:
Rexulti
Rexulti is an antipsychotic used to treat major depressive disorder, schizophrenia, or agitation ...
Ubrelvy
Ubrelvy (ubrogepant) tablets are used for the acute treatment of migraine. Includes Ubrelvy side ...
Qulipta
Qulipta is used to help prevent episodic or chronic migraine headaches in adults. Qulipta is an ...
Nurtec ODT
Nurtec ODT (rimegepant) is used to treat acute migraines and prevent episodic migraines, by ...
Paracetamol
Paracetamol (Panadol, Calpol, Alvedon) is a widely used over-the-counter painkiller and fever ...
Tylenol
Tylenol is a pain reliever and a fever reducer used to treat many conditions such as headaches ...
Cymbalta
Cymbalta (duloxetine) is used to treat major depressive disorder, general anxiety disorder and ...
Naproxen
Naproxen is a nonsteroidal anti-inflammatory drug used to treat pain or inflammation caused by ...
Oxycodone
Oxycodone is an opioid analgesic used to treat moderate to severe pain; it has a high potential for ...
Diclofenac
Diclofenac is a nonsteroidal anti-inflammatory drug used to treat pain and inflammation associated ...
More about amitriptyline
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1,812)
- Drug images
- Side effects
- Dosage information
- Patient tips
- During pregnancy
- Support group
- Drug class: tricyclic antidepressants
- Breastfeeding
- En español
Patient resources
Other brands
Professional resources
Other brands
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
