Hepatitis C
Medically reviewed by Drugs.com. Last updated on Aug 4, 2025.
What is hepatitis C?
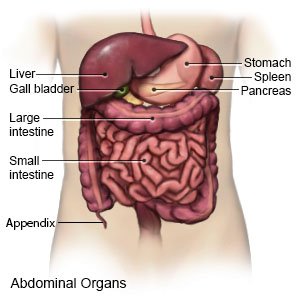
Hepatitis C is inflammation of the liver caused by hepatitis C virus (HCV) infection.
 |
How is HCV spread?
HCV is carried in the blood and other body fluids, such as semen or vaginal fluids. The following are some ways HCV is spread:
- A stick from an infected needle, including for illegal drugs and for procedures such as tattooing
- An object with infected blood or body fluids on it touches your wound
- Sharing personal items, such as razors, toothbrushes, or nail clippers with someone who has hepatitis C
- Travel to areas in the world where HCV is common
- Unprotected sex with someone who has hepatitis C, sex with more than one partner, or you are a man who has sex with men
- Rarely, a blood, organ, or tissue transplant from an infected donor, or long-term kidney dialysis
What are the signs and symptoms of hepatitis C?
You may not have any signs or symptoms at first. Any of the following may develop if HCV damages your liver:
- Fatigue
- Dark urine or pale bowel movements
- Fever
- Jaundice (yellow skin or eyes) and itchy skin
- Joint pain, body aches, or weakness
- Loss of appetite, nausea, or vomiting
- Pain in the upper right side of your abdomen
What do I need to know about hepatitis C screening?
Screening means you are tested for hepatitis C before you have signs or symptoms. This helps healthcare providers find and treat hepatitis C early. Screening is usually recommended 1 time for all adults who are 18 to 79 years of age. Screening may also be recommended during pregnancy to lower the risk for HCV being passed from mother to baby. Screening may start before age 18 or after 79 if your risk is high and continue regularly if your risk remains high.
How is hepatitis C diagnosed?
Your healthcare provider will ask if you have symptoms of hepatitis C. Tell your provider any health problems you have, and if you have other infections, such as HIV or hepatitis B. Tell your provider if you drink alcohol or use any illegal drugs. Also tell your provider if you have a tattoo, and when you got it. A tattoo applied with a needle increases the risk for hepatitis C. Your provider may also ask about your sex partners. You may need any of the following tests:
- Blood tests are used to check for HCV antibodies made by your body to fight the infection. The tests can show the type and amount of HCV you have. This will help your provider make a treatment plan.
- A liver biopsy is used to take a sample of your liver. Tests on the sample will show the type of HCV you have and if the infection is severe.
- An endoscopy or a CT scan may be needed to check for liver disease and to check your liver function.
How is hepatitis C treated?
Your body may be able to fight an HCV infection on its own. An infection that continues longer than 6 months will need treatment. Treatment helps prevent health problems hepatitis C can cause, such as liver failure or cirrhosis. You may need any of the following:
- Antiviral medicines help control HCV. These medicines will not get rid of the virus, but they can make it inactive in your body. Antivirals can also shorten the amount of time you have symptoms or make them less severe. You will need to take a combination of antivirals for at least 8 to 12 weeks.
- Surgery may be needed to remove part of your liver. A liver transplant may be done if your liver stops working. Your diseased liver is removed and replaced with a healthy, donated liver.
Treatment options
The following list of medications are related to or used in the treatment of this condition.
What can I do to manage hepatitis C?
- Do not drink alcohol or use illegal drugs. Alcohol and drugs cause or increase your risk for liver damage. Ask your healthcare provider for more information if you need help quitting.
- Do not use any medicines without talking to your provider. This includes medicine that has been ordered for you and over-the-counter medicine. Ask before you use acetaminophen, vitamins, herbs, herbal teas, laxatives, or food supplements. Any of these could harm your liver.
- Do not smoke. Nicotine can damage blood vessels and make it more difficult to manage hepatitis C. Do not use e-cigarettes or smokeless tobacco in place of cigarettes or to help you quit. They still contain nicotine. Ask your provider for information if you currently smoke and need help quitting.
- Eat a variety of healthy foods. Healthy foods include fruits, vegetables, low-fat dairy products, beans, and lean meats and fish. Ask if you need to be on a special diet.

- Get more rest. Slowly return to your normal activities when you feel better.
- Talk to your provider about vaccines. You may need to get vaccines to protect you from hepatitis A or B. You may also need a pneumonia vaccine. Get the flu vaccine as soon as recommended each year, usually in September or October. Get all recommended COVID-19 vaccine doses and boosters. Ask your provider about other vaccines you need.
How can I prevent the spread of HCV?
No vaccine is available to prevent hepatitis C. The following can help prevent HCV from spreading to others:
- Wash your hands often. Use soap and water. Wash your hands several times each day. Wash after you use the bathroom, change a child's diaper, and before you prepare or eat food. Wash for at least 20 seconds. Rinse with warm, running water for several seconds. Then dry your hands with a clean towel or paper towel. Use hand sanitizer that contains alcohol if soap and water are not available. Do not touch your eyes, nose, or mouth without washing your hands first.
- Cover any open cuts or scratches. If blood from your wound gets on a surface, clean the surface with bleach right away. Put on gloves before you clean. Throw away any items with blood or body fluids on them, as directed by your healthcare provider.
- Do not share personal items. These items include toothbrushes, nail clippers, and razors. Do not share needles.
- Tell household members and sex partners that you have HCV. They should be tested for HCV. Do not have sex, including oral and anal sex, until your provider tells you it is okay. If you have sex, make sure the male partner wears a latex condom.
- Protect your baby. If you are currently pregnant, your provider will give you more information on protecting your baby from HCV. Medicines used to treat hepatitis C are not routinely used during pregnancy. Your provider may recommend antivirals if the benefits to you and your baby outweigh the risks. Your provider will talk to you about the benefits and risks. It is okay to breastfeed your baby unless your nipples are cracked or bleeding.
- Do not donate blood, body organs, semen, or other tissues. Donations are checked for HCV, but it is best not to donate.
 |
When should I seek immediate care?
- You have severe abdominal pain.
- You are too dizzy to stand up.
- You vomit blood or material that looks like coffee grounds.
- You feel confused or are very sleepy.
- Your bowel movements are red or black, and sticky.
When should I call my doctor?
- You have a fever.
- You are vomiting and cannot keep food or liquids down.
- Your abdomen or legs have a rash or are swollen.
- You are bruising easily.
- You have questions or concerns about your condition or care.
Care Agreement
You have the right to help plan your care. Learn about your health condition and how it may be treated. Discuss treatment options with your healthcare providers to decide what care you want to receive. You always have the right to refuse treatment. The above information is an educational aid only. It is not intended as medical advice for individual conditions or treatments. Talk to your doctor, nurse or pharmacist before following any medical regimen to see if it is safe and effective for you.© Copyright Merative 2025 Information is for End User's use only and may not be sold, redistributed or otherwise used for commercial purposes.
Learn more about Hepatitis C
Treatment options
- Medications for Hepatitis C
- Medications for Infectious Hepatitis
- Medications for Infectious Hepatitis with Coma
Care guides
Symptoms and treatments
Medicine.com guides (external)
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
