Omidria: Package Insert / Prescribing Info
Package insert / product label
Generic name: phenylephrine and ketorolac
Dosage form: injection, solution, concentrate
Drug class: Ophthalmic surgical agents
J Code (medical billing code): J1097 (1 mL, ophth irrigation solution)
Medically reviewed by Drugs.com. Last updated on Jul 2, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
OMIDRIA ® (phenylephrine and ketorolac intraocular solution) 1% / 0.3%, for addition to ocular irrigating solution
Initial U.S. Approval: 2014
Indications and Usage for Omidria
OMIDRIA is an alpha 1-adrenergic receptor agonist and nonselective cyclooxygenase inhibitor indicated for:
OMIDRIA is added to an ocular irrigating solution used during cataract surgery or intraocular lens replacement.
Omidria Dosage and Administration
- Each vial of OMIDRIA must be diluted prior to use for administration to a single patient undergoing cataract surgery or intraocular lens replacement.
- Dilute 4 mL of OMIDRIA in 500 mL of ocular irrigating solution. Irrigation solution is to be used as needed for the surgical procedure. ( 2)
Dosage Forms and Strengths
Intraocular solution containing phenylephrine 10.16 mg/mL (1%) and ketorolac 2.88 mg/mL (0.3%) for use in a single patient. ( 3)
Contraindications
Hypersensitivity to any component of this product ( 4)
Warnings and Precautions
Systemic exposure to phenylephrine can cause elevations in blood pressure ( 5.1).
Adverse Reactions/Side Effects
The most common reported adverse reactions (≥2%) are eye irritation, posterior capsule opacification, increased intraocular pressure, and anterior chamber inflammation. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Rayner Surgical Inc. at 1-877-0MIDRIA or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2023
Full Prescribing Information
1. Indications and Usage for Omidria
Omidria ® is added to an ocular irrigating solution used during cataract surgery or intraocular lens replacement and is indicated for maintaining pupil size by preventing intraoperative miosis and reducing postoperative ocular pain.
2. Omidria Dosage and Administration
Omidria must be diluted prior to intraocular use. For administration to patients undergoing cataract surgery or intraocular lens replacement, 4 mL of Omidria is diluted in 500 mL of ocular irrigating solution. Irrigation solution is to be used as needed for the surgical procedure for a single patient.
The storage period for the diluted product is not more than 4 hours at room temperature or 24 hours under refrigerated conditions.
Do not use if the solution is cloudy or if it contains particulate matter.
3. Dosage Forms and Strengths
Omidria is an intraocular solution containing 10.16 mg/mL (1% w/v) of phenylephrine and 2.88 mg/mL (0.3% w/v) of ketorolac for use in a single patient.
4. Contraindications
Omidria is contraindicated in patients with a known hypersensitivity to any of its ingredients.
5. Warnings and Precautions
5.1 Elevated Blood Pressure
Systemic exposure to phenylephrine can cause elevations in blood pressure.
5.2 Cross-Sensitivity or Hypersensitivity
There is the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other non-steroidal anti- inflammatory drugs (NSAIDs). There have been reports of bronchospasm or exacerbation of asthma associated with the use of ketorolac in patien ts who either have a known hypersensitivity to aspirin/NSAIDs or a past medical history of asthma. Therefore, use Omidria with caution in individuals who have previously exhibited sensitivities to these drugs.
6. Adverse Reactions/Side Effects
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Table 1 shows frequently reported ocular adverse reactions with an incidence of ≥ 2% of adult patients as seen in the combined clinical trial results from three randomized, placebo-controlled studies [see Clinical Studies ( 14)] .
| MedDRA Preferred Term | Placebo
(N=462) | Omidria
(N=459) |
| n (%) | n (%) | |
| Ocular Events | ||
| Anterior Chamber Inflammation | 102 (22%) | 111 (24%) |
| Intraocular Pressure Increased | 15 (3%) | 20 (4%) |
| Posterior Capsule Opacification | 16 (4%) | 18 (4%) |
| Eye Irritation | 6 (1%) | 9 (2%) |
| Foreign Body Sensation in Eyes | 11 (2%) | 8 (2%) |
In a safety study that enrolled 72 pediatric patients up to 3 years old, no overall difference in safety was observed between pediatric and adult patients.
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are no available data on Omidria use in pregnant women or animals to inform
any drug-associated risks. Oral administration of ketorolac to rats during late
gestation produced dystocia and increased pup mortality at a dose 740-times the
plasma exposure at the recommended human ophthalmic dose (RHOD). Since
human systemic exposure to Omidria following a lens replacement procedure is low
(see Clinical Pharmacology (12.3)], the applicability of animal findings to the risk
of Omidria in humans during pregnancy is unclear. Omidria should be used during
pregnancy only if the potential benefit justifies the potential risk to the fetus.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature closure of the ductus arteriosus in the fetus has occurred with third
trimester use of oral and injectable NSAIDs. Ketorolac plasma concentrations are
detectable following ocular Omidria administration [see Clinical Pharmacology (12.3)].
The use of Omidria during late pregnancy should be avoided.
.Q.a1a
Animal Data
No well-controlled animal reproduction studies have been conducted with Omidria
or phenylephrine.
Ketorolac, administered during organogenesis, did not cause embryofetal abnormalities
or mortalities in rabbits or rats at oral doses of 3.6 mg/kg/day and 10 mg/kg/day,
respectively. These doses produced systemic exposure that is 1150 times and
4960 times the plasma exposure (based on Cma,c) at the RHOD, respectively. When
administered to rats during late gestation (after Day 17 of gestation) at oral doses
up to 1.5 mg/kg/day (740 times the plasma exposure at the RHOD), ketorolac
produced dystocia and increased pup mortality.
Risk Summary
There are no available data on Omidria use in pregnant women or animals to inform any drug-associated risks. Oral administration of ketorolac to rats during late gestation produced dystocia and increased pup mortality at a dose 740-times the plasma exposure at the recommended human ophthalmic dose (RHOD). Since human systemic exposure to Omidria following a lens replacement procedure is low [ see Clinical Pharmacology ( 12.3) ], the applicability of animal findings to the risk of Omidria in humans during pregnancy is unclear. Omidria should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature closure of the ductus arteriosus in the fetus has occurred with third trimester use of oral and injectable NSAIDs. Ketorolac plasma concentrations are detectable following ocular Omidria administration [see Clinical Pharmacology ( 12.3)] . The use of Omidria during late pregnancy should be avoided.
Data
Animal Data
No well-controlled animal reproduction studies have been conducted with Omidria or phenylephrine.
Ketorolac, administered during organogenesis, did not cause embryofetal abnormalities or mortalities in rabbits or rats at oral doses of 3.6 mg/kg/day and 10 mg/kg/day, respectively. These doses produced systemic exposure that is 1150 times and 4960 times the plasma exposure (based on C max) at the RHOD, respectively. When administered to rats during late gestation (after Day 17 of gestation) at oral doses up to 1.5 mg/kg/day (740 times the plasma exposure at the RHOD) , ketorolac produced dystocia and increased pup mortality.
8.2 Lactation
Risk Summary
There are no data on the presence of Omidria in human milk, the effects on the breastfed infant, or the effects on milk production. Howerver, systemic exposure to Omidria, following a lens replacement procedure is low [see Clinical Pharmacology ( 12.3)] . The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Omidria and any potential adverse effects on the breastfed child from Omidria.
8.4 Pediatric Use
The safety and effectiveness of Omidria have been established in the pediatric population from neonates to adolescents (birth to younger than 17 years). Use of Omidria in this population is supported by evidence from adequate and well-controlled studies of Omidria in adults with additional data from a single active-controlled safety study in pediatric patients up to 3 years old [see Clinical Studies ( 14)] .
No overall differences in safety were observed between pediatric and adult patients.
10. Overdosage
Systemic overdosage of phenylephrine may cause a rise in blood pressure. It may also cause headache, anxiety, nausea, vomiting, and ventricular arrhythmias. Supportive care is recommended.
11. Omidria Description
Omidria is a sterile aqueous solution, containing the α 1-adrenergic receptor agonist phenylephrine HCl and the nonsteroidal anti-inflammatory ketorolac tromethamine, for addition to ocular irrigating solution.
The descriptions and structural formulae are:
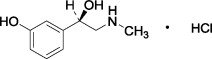
Phenylephrine Hydrochloride Drug Substance:
| Common Name: | phenylephrine hydrochloride |
| Chemical Name: | (-)- m-Hydroxy-α-[(methylamino)methyl]benzyl alcohol hydrochloride |
| Molecular Formula: | C 9H 13NO 2 · HCl |
| Molecular Weight: | 203.67 g/mole |
Figure 1: Chemical Structure for Phenylephrine HCl
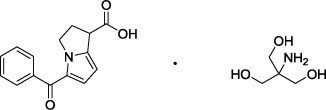
Ketorolac Tromethamine Drug Substance:
| Common Name: | ketorolac tromethamine |
| Chemical Name: | (±)-5-Benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid : 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1) |
| Molecular Formula: | C 15H 13NO 3 · C 4H 11NO 3 |
| Molecular Weight: | 376.40 g/mole |
Figure 2: Chemical Structure for Ketorolac Tromethamine
Omidria is a clear, colorless to slightly yellow, sterile solution concentrate with a pH of approximately 6.3.
Each vial of Omidria contains:
Actives: phenylephrine hydrochloride 12.4 mg/mL equivalent to 10.16 mg/mL of phenylephrine and ketorolac tromethamine 4.24 mg/mL equivalent to 2.88 mg/mL of ketorolac.
Inactives: citric acid monohydrate; sodium citrate dihydrate; water for injection; may include sodium hydroxide and/or hydrochloric acid for pH adjustment.
12. Omidria - Clinical Pharmacology
12.1 Mechanism of Action
The two active pharmaceutical ingredients (API) in Omidria, phenylephrine and ketorolac, act to maintain pupil size by preventing intraoperative miosis, and reducing postoperative pain.
Phenylephrine is an α 1-adrenergic receptor agonist and, in the eye, acts as a mydriatic agent by contracting the radial muscle of the iris. Ketorolac is a nonsteroidal anti-inflammatory that inhibits both cyclooxygenase enzymes (COX-1 and COX-2), resulting in a decrease in tissue concentrations of prostaglandins to reduce pain due to surgical trauma. Ketorolac, by inhibiting prostaglandin synthesis secondary to ocular surgical insult or direct mechanical stimulation of the iris, also prevents surgically induced miosis.
12.3 Pharmacokinetics
In a pharmacokinetic study evaluating Omidria, systemic exposure to both phenylephrine and ketorolac was low or undetectable.
A single-dose of Omidria as part of the irrigation solution was administered in 14 patients during lens replacement surgery. The volume of irrigation solution used during surgery ranged between 150 mL to 300 mL (median 212.5 mL). Detectable phenylephrine plasma concentrations were observed in one of 14 patients (range 1.2 to 1.4 ng/mL) during the first 2 hours after the initiation of Omidria administration. The observed phenylephrine plasma concentrations could not be distinguished from the preoperative administration of phenylephrine 2.5% ophthalmic solution prior to exposure to Omidria.
Ketorolac plasma concentrations were detected in 10 of 14 patients (range 1.0 to 4.2 ng/mL) during the first 8 hours after the initiation of Omidria administration. The maximum ketorolac concentration was 15 ng/mL at 24 hours after the initiation of Omidria administration, which may have been due to application of postoperative ketorolac ophthalmic solution.
14. Clinical Studies
Studies in Adults
The efficacy and safety of Omidria were evaluated in two Phase 3, randomized, multicenter, double-masked, placebo-controlled clinical trials in 808 adult patients undergoing cataract surgery or intraocular lens replacement.
Patients were randomized to either Omidria or placebo. Patients were treated with preoperative topical mydriatic and anesthetic agents. Pupil diameter was measured throughout the surgical procedure. Postoperative pain was evaluated by self-administered 0-100 mm visual analog scales (VAS).
Mydriasis was maintained in the Omidria-treated groups while the placebo-treated groups experienced progressive constriction.
Figure 3: Intraoperative Pupil Diameter (mm) Change-from-Baseline
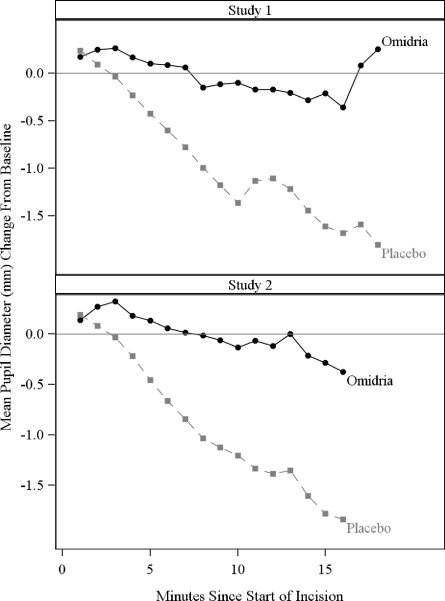
At the end of cortical clean-up, 23% of placebo-treated patients and 4% of Omidria-treated patients had a pupil diameter less than 6 mm (p < 0.01).
Pain during the initial 10-12 hours postoperatively was statistically significantly less in the Omidria-treated groups than in the placebo-treated groups.
Figure 4: Postoperative Mean Visual Analog Scale (VAS) Scores for Pain
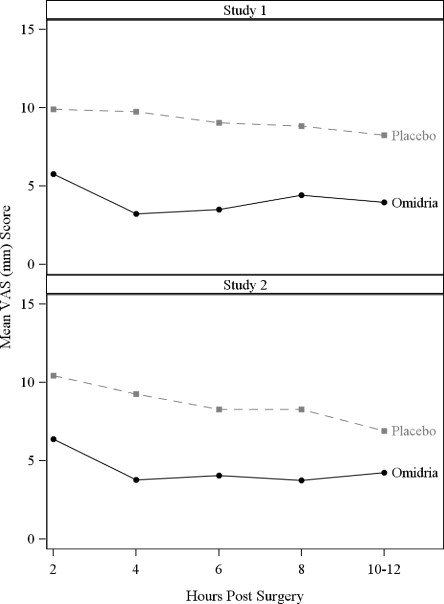
During the 10-12 hours postoperatively, 26% of Omidria-treated patients reported no pain (VAS = 0 at all timepoints) while 17% of placebo-treated patients reported no pain (p < 0.01).
Study in Pediatric Patients
The safety of Omidria was evaluated in a single, randomized, multicenter, double-masked, active-controlled clinical study in 72 pediatric patients up to 3 years old undergoing cataract surgery with or without intraocular lens replacement.
Patients were randomized to either Omidria or phenylephrine. Patients were treated with preoperative topical mydriatic and anesthetic agents. As in the adult studies, mydriasis was maintained in the Omidria-treated group. No overall differences in safety were observed between pediatric and adult patients.
16. How is Omidria supplied
Omidria (phenylephrine and ketorolac intraocular solution) 1%/0.3% is supplied in a clear, 5-mL glass, single-patient-use vial containing 4 mL of sterile solution, for addition to ocular irrigating solution.
Omidria is supplied in a multi-pack containing:
4 vials : NDC 82604-600-04 or
1 vials: NDC 82604-600-00
17. Patient Counseling Information
Inform patients that they may experience sensitivity to light.
Rayner Surgical Inc.
Suite 102, 14335 NE 24 Street
Bellevue, WA 98007
© Rayner 2023
Patented product; see www.rayner.com/patents for further details.
OMIDRIA®and the OMIDRIA® Logo are registered trademarks of Rayner Surgical Inc.
640069
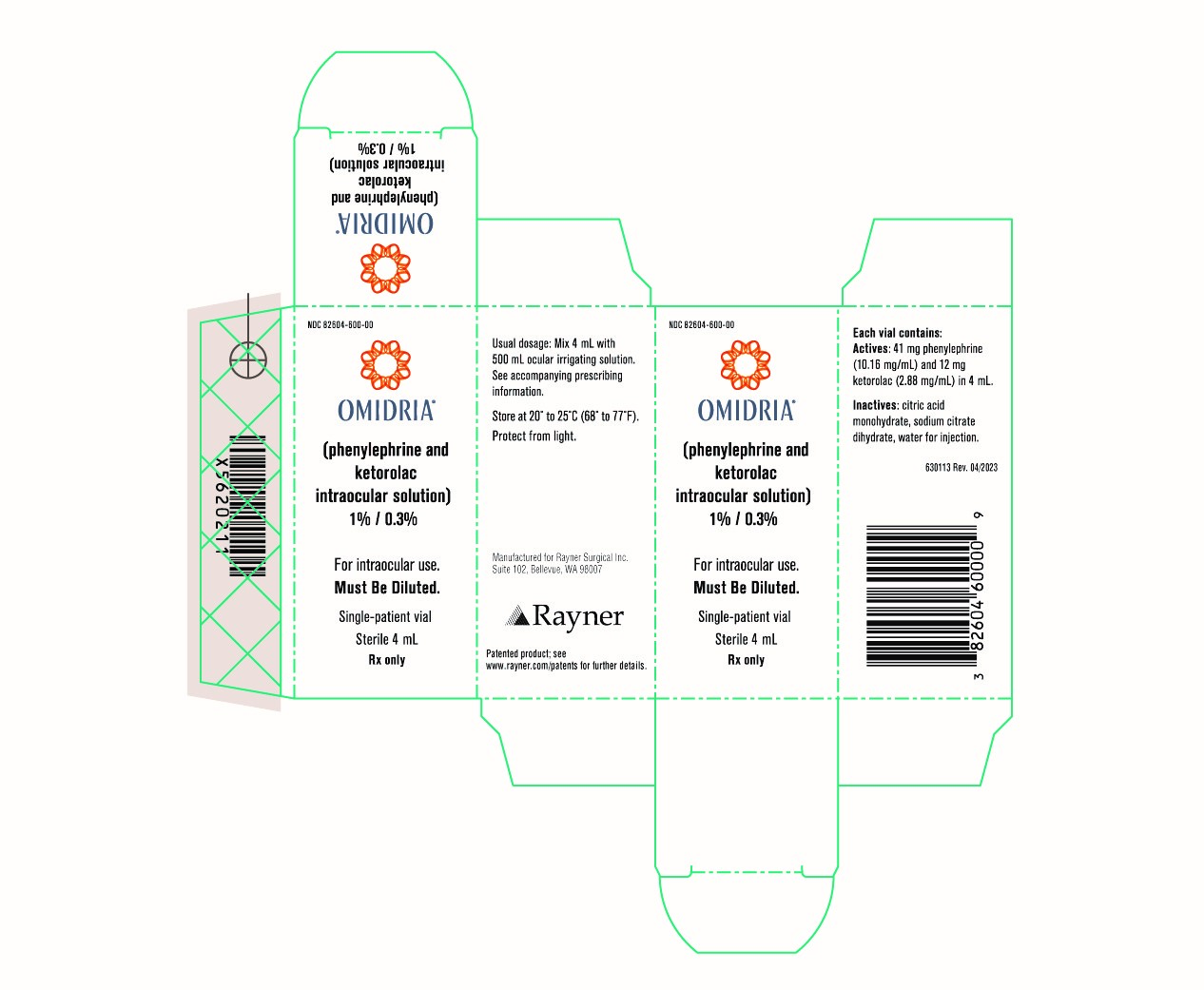
PRINCIPAL DISPLAY PANEL
NDC 82604-600-04
OMIDRIA®
(phenylephrine and ketorolac intraocular solution)
1% / 0.3%
For Intraocular use.
Must Be Diluted.
Single-Patient vial
Sterile 4 mL
Quantity: 4
Rx Only

NDC 82604-600-00
OMIDRIA ®
(phenylephrine and ketorolac intraocular solution)
1% / 0.3%
For Intraocular use.
Must Be Diluted.
Single-Patient vial
Sterile 4 mL
Quantity: 1
Rx Only
| OMIDRIA
phenylephrine and ketorolac injection, solution, concentrate |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Rayner Surgical Inc. (118502278) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Patheon Manufacturing Services LLC | 079415560 | manufacture(82604-600) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Hospira | 093132819 | manufacture(82604-600) | |
More about Omidria (ketorolac / phenylephrine ophthalmic)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: ophthalmic surgical agents
