Glofil-125: Package Insert / Prescribing Info
Package insert / product label
Generic name: iothalamate sodium I-125
Dosage form: injection, solution
Drug class: Ionic iodinated contrast media
Medically reviewed by Drugs.com. Last updated on Mar 3, 2025.
On This Page
Glofil-125 Description
General
GLOFIL-125 (Sodium Iothalamate I-125 Injection) is a sterile, nonpyrogenic aqueous injection containing approximately 1 mg sodium iothalamate per mL, and 0.9 percent benzyl alcohol as a preservative. The radioactive concentration of the material is 250-300 µCi/mL as of the calibration date. Sodium bicarbonate and hydrochloric acid are present for pH adjustment.
Physical Characteristics
Iodine-125 decays by electron capture with a physical half-life of 60.14 days.
Photons that are useful for detection are listed in Table 1.
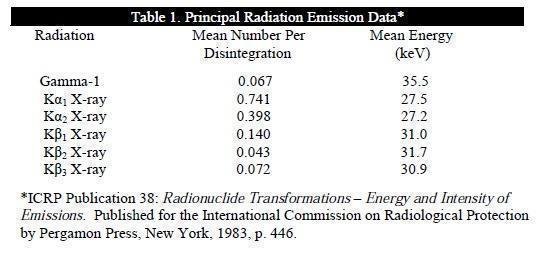
The specific gamma ray constant for I-125 is 1.43 R/mCi-hr at 1 cm. The first half value thickness of lead (Pb) for I-125 is 0.017 mm. A range of values for the relative attenuation of the radiation emitted by this radionuclide resulting from interposition of various thicknesses of Pb is shown in Table 2. For example, the use of 0.28 mm of Pb will decrease the external radiation exposure by a factor of 10,000.

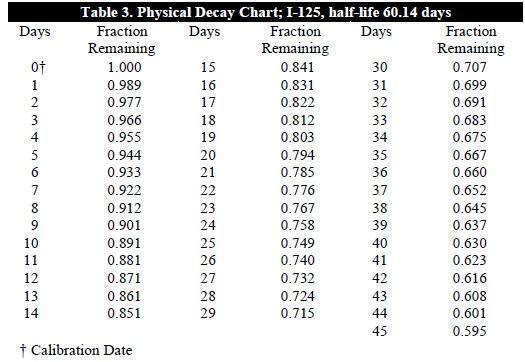
To correct for physical decay of this radionuclide, the fractions that remain at selected time intervals after the date of calibration are shown in Table 3.
Glofil-125 - Clinical Pharmacology
The renal clearance of sodium iothalamate in man closely approximates that of inulin. The compound is cleared by glomerular filtration without tubular secretion or reabsorption. Following infusion administration of I-125 iothalamate, the effective half-life is about 0.07 days.
Indications and Usage for Glofil-125
GLOFIL-125 (Sodium Iothalamate I-125 Injection) is indicated for evaluation of glomerular filtration in the diagnosis or monitoring of patients with renal disease.
Precautions
General
As in the use of any radioactive material, care should be taken to minimize radiation exposure to the patient, consistent with proper patient management, and to insure minimum radiation exposure to occupational workers. Radiopharmaceuticals should be used only by physicians who are qualified by training and experience in the safe use and handling of radionuclides. Rapid or bolus-like injections should be avoided.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic potential, mutagenic potential, or whether this drug affects fertility in males or females.
Pregnancy Category C
Animal reproduction studies have not been conducted with GLOFIL-125. It is also not known whether GLOFIL-125 can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. GLOFIL-125 should be given to a pregnant woman only if clearly needed.
Nursing Mothers
Radioiodine is excreted in human milk during lactation. It is not known whether GLOFIL-125 is excreted in human milk. Therefore, formula feedings should be substituted for breast feedings.
Pediatric Use
Safety and effectiveness in children have not been established.
Related/similar drugs
Glofil-125 Dosage and Administration
The suggested dose range employed in the average patient (70 kg) is as follows:
Continuous intravenous infusion: 20 to 100 μCi (0.74-3.7 megabecquerels) (Sigman, et al (1) method).
Single intravenous injection: 10 to 30 µCi (0.37-1.11 megabecquerels) Cohen, et al (2) method)
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Technique
Continuous intravenous infusion
Sigman1 method
I. Preparation:
- Adequate diuresis (a urine flow exceeding 3 mL/min.) is established, preferably by an oral water load of 1,500 mL two hours prior to the beginning of the clearance study.
2. It is not necessary to withhold breakfast or admit the patient the night before.
II. Procedure:
- After the establishment of adequate diuresis, a number 14 or 16 French Foley catheter is aseptically inserted into the bladder.
- An intravenous infusion of Lactated Ringers (Hartmanns) solution is started in each arm, one to maintain a site for injection of the GLOFIL-125, the other to serve as a site for serial withdrawal of blood. A two-way stopcock connects the needle and intravenous tubing of each arm.
- The dose is equally divided into (1) an intravenous priming dose to be injected as is and (2) a sustaining dose to be diluted in 30 to 60 mL of isotonic sodium chloride, depending on how many collection periods are anticipated.
- The priming dose is slowly injected into one arm. This is immediately followed by infusion of the sustaining solution through the same site, usually at the rate of 0.5 mL/min., by means of an automatic pump. During this infusion, the Lactated Ringers solution in the same arm is discontinued, and 40 to 45 minutes are allowed for equilibration in order to reach a state of constant plasma concentration of radioactivity.
- After attaining equilibrium, consecutive 15 minute collection periods are started. From the arm opposite the injection site, 5 mL of blood (allowing duplicate plasma counting volumes) is drawn six minutes prior to the midpoint of each collection period, placed in heparinized tubes, mixed, and centrifuged. The blood samples may be obtained through the two-way stopcock after discarding the first 30 mL aspirated into the syringe. This 30 mL contains the contents of the tubing, including infusion fluid, and must be cleared in order to obtain an undiluted blood sample. If desired, this step may be eliminated and blood samples obtained by direct venipuncture.
- During each collection period, total urine must be accurately collected and the volume accurately measured. Three such consecutive collection periods are sufficient for most clinical studies.
III. Clearance Calculations:
1. Aliquots (1 mL each) of plasma and urine from each collection period are counted in a standard gamma-ray scintillation well detector.
2. All counts are corrected for background activity.
3. Glomerular filtration rate is calculated by the formula C=UV/P, in which:
C = glomerular filtration rate in mL/min
U= urinary concentration of radioactivity in net counts/min/mL
V= urinary flow rate in mL/min
P = plasma concentration of radioactivity in net counts/min/mL
4. Average glomerular filtration rate (GFR) is calculated from the rates for the individual collection periods. GFR can be expressed in terms of body
weight (mL/min/kg) or body surface area (mL/min/m2).
5. Unilateral glomerular filtration rates can be determined by the same technique by utilizing ureteral catheterization.
Single intravenous injection
Cohen2 method:
The method of Cohen, et al2 requires little preparation, few and small blood samples, no bladder catheterization, and no constant intravenous infusion. It is simple to perform, rapid, and utilizes equipment which is readily available in most modern laboratories.
I. Preparation:
1. Lugol's solution, 3 drops orally, three times a day, is administered for one or two days prior to the test.
No diet or water restriction is necessary.
2. Oral water load is begun one hour before starting the test. Start with 20 mL/kg and force any clear liquids (unless contraindicated) until the test is complete.
II. Procedure: Record actual times for the collection of the blood and urine samples.
1. Empty the bladder and label the urine Urine control.
2. Inject 10-30 µCi GLOFIL-125 intravenously; wait 30 to 60 minutes.
3. Collect the entire urine and label Urine discard.
4. Draw 4 to 5 mL of blood into a heparinized syringe. Label Plasma #1.
5. After another 30 to 60 minutes, collect the entire urine and label Urine #1.
6. Immediately draw another blood specimen. Label Plasma #2.
7. After final 30 to 60 minute wait, collect the urine. Label Urine #2.
8. Draw the last blood specimen immediately. Label Plasma #3.
III. Clearance Calculations:
1. Radioactivity of one mL aliquots of both urine and plasma are determined using a well-scintillation detector with a single channel pulse-height analyzer. Sufficiently reproducible counts are usually obtained with time settings of 2 minutes for urine samples and 20 minutes for the plasma samples. Calculations of the clearance rates are made by using the formula:(1)
C = C = UV/P + 1.73/SA where
C = glomerular filtration rate in mL/min/1.73 m2
U = urine radioactivity in counts/min/mL
V = urine flow rate in mL/min
P= mean plasma radioactivity in counts/min/mL
SA= body surface area in m2
Radiation Dosimetry
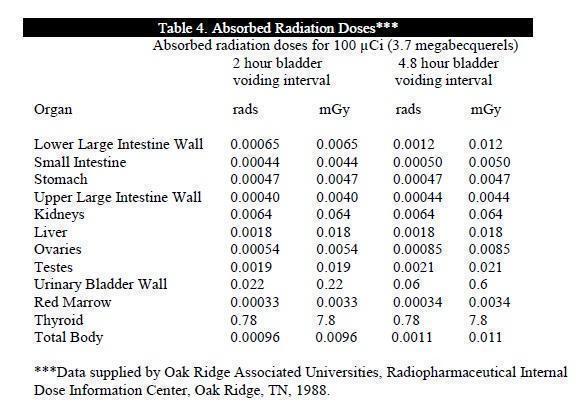
The estimated absorbed radiation doses to an average (70 kg) patient from an intravenous dose of 100 µCi (3.7 megabecquerels) of GLOFIL-125 are shown in Table 4. Calculations assume that there is 1% free iodide in the preparation and that the thyroid uptake of the iodine is 25%.
Visual Inspection
Parental drug products should be inspected visually for particulate matter and discoloration prior to admnistration, whenever solution and container permit.
How is Glofil-125 supplied
Identity
No. 1000, GLOFIL-125 is a clear, colorless, sterile, and nonpyrogenic solution available as a 4 mL vial. It is supplied in a concentration of approximately 1 mg/mL sodium iothalamate (range is 0.5–2.0 mg sodium iothalamate per mL), with a radioactivity concentration of 250 to 300 μCi/mL at the time of calibration. Benzyl alcohol 0.9%, is added as a preservative. Sodium bicarbonate and hydrochloric acid are added for pH adjustment. The calibration and expiration dates are shown on the label.
Dose Volume Calculation
Table 3 provides the required factors for the determination of activity per mL post calibration date for GLOFIL-125 sterile solution.
To determine the dose volume, locate the decay factor (fraction remaining) which corresponds to the day that the dose is to be administered. The following equation is then utilized to determine the dose volume:
activity of desired dose = dose
decay factor x amount of activity/mL on calibration day volume (mL)
(information on label)
References
1. Sigman EM, Elmwood CM, Reagan ME, Morris AM, Calanzaro A. The renal clearance of 131 I labeled sodium iothalamate in man. Invest Urol 1965; 2:432.
2. Cohen ML, Smith FG Jr., Mindell RS, Vernier RL. A simple reliable method of measuring glomerular filtration rate using single low dose sodium iothalamate 131I. Pediatrics 1969; 43:407
ADDITIONAL REFERENCES
3. Maher FT, Nolan NG, Elveback LR. Comparisons of simultaneous clearances of 125I labeled sodium iothalamate (Glofil) and of Inulin. Mayo Clin Proc 1971; 46: 690-691.
4. Skov PE. Glomerular filtration rate in patients with severe and very severe renal insufficiency. Acta Med Scand 1970; 187:419-428.
| GLOFIL-125
sodium iothalamate i-125 injection injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Iso-Tex Diagnostics, Inc. (181202995) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Iso-Tex Diagnostics, Inc. | 181202995 | manufacture(50914-7729) | |
More about iothalamate
- Check interactions
- Compare alternatives
- Latest FDA alerts (2)
- Side effects
- Dosage information
- During pregnancy
- Drug class: ionic iodinated contrast media
- Breastfeeding

