Betasept Antiseptic Surgical Scrub: Package Insert / Prescribing Info
Package insert / product label
Generic name: chlorhexidine gluconate
Dosage form: topical solution
Drug classes: Antiseptic and germicides, Mouth and throat products
Medically reviewed by Drugs.com. Last updated on May 9, 2025.
Indications and Usage for Betasept Antiseptic Surgical Scrub
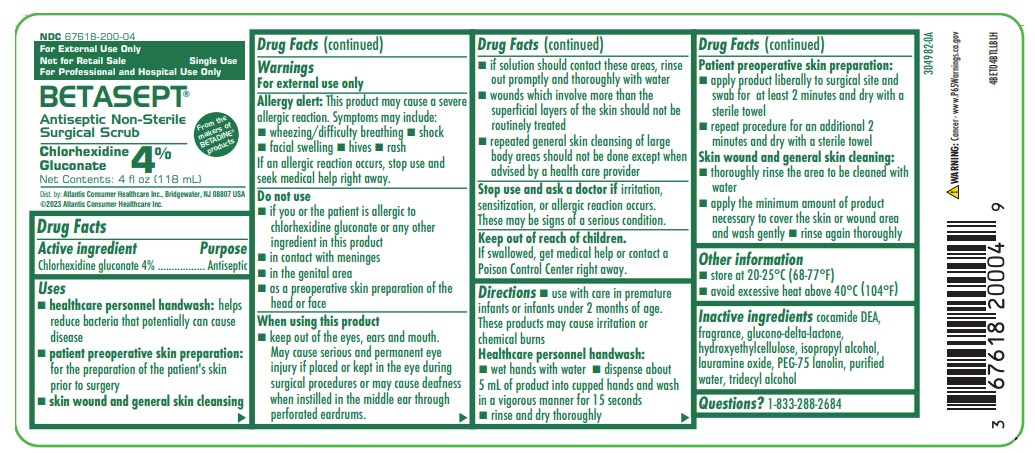
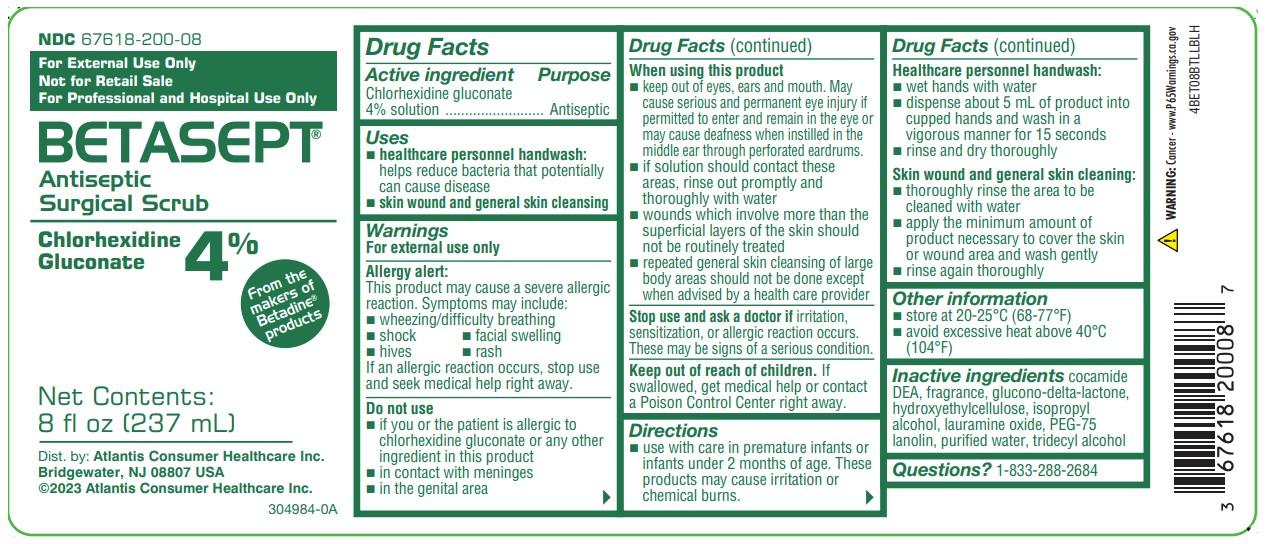
- surgical hand scrub: significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care
- healthcare personnel handwash: helps reduce bacteria that potentially can cause disease
- patient preoperative skin preparation: preparation of the patient's skin prior to surgery
- skin wound and general skin cleansing
Warnings
Warnings
For external use only
Allergy alert:
This product may cause a severe allergic reaction. Symptoms may include:
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
If an allergic reaction, stop use and seek medical help right away.
Do not use
- if you or the patient is allergic to chlorhexidine gluconate or any other ingredient in this product
- in contact with meninges
- in the genital area
- as a preoperative skin preparation of the head or face
When using this product
- keep out of the eyes, ears, and mouth. May cause serious and permanent eye injury if placed or kept in the eye during surgical procedures or may cause deafness when instilled in the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- wounds which involve more than the superficial layers of the skin should not be routinely treated
- repeated general skin cleansing of large body areas should not be done except when advised by a health care provider
Related/similar drugs
Stop use and ask a doctor if
irritation, sensitization, or allergic reaction occurs. These may be signs of a serious condition.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Betasept Antiseptic Surgical Scrub Dosage and Administration
- use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand scrub
- wet hands and forearms with water
- scrub for 3 minutes with about 5 mL of product and a wet brush, paying close attention to the nails, cuticles, and interdigital spaces
- a seperate nail cleaner may be used
- rinse thoroughly
- wash for an additional 3 minutes with 5 mL of product and rinse under running water
- dry thoroughly
Healthcare personnel handwash
- wet hands with water
- dispense about 5 mL of priduct into cupped hands and wash in a vigorous manner for 15 seconds
- rinse and dry thoroughly
Patient preoperative skin preparation
- apply product liberally to surgical site and swab for at least 2 minutes and dry with a sterile towel
- repeat procedure for an additional 2 minutes and dry with a sterile towel
Skin wound and general skin cleansing
- thoroughly rinse the area to be cleaned with water
- apply the minimum amount of product necessary to cover the skin or wound area and wash gently
- rinse again thoroughly
Inactive ingredients
cocamide DEA, fragrance, glucono-delta-lactone, hydroxyethylcellulose, isopropyl alcohol, lauramine oxide, PEG-75 lanolin, purified water, tridecyl alcohol
NDC 67618-200-04
From the Makers of BETADINE® products
For External Use Only
Not for Retail Sale
For Professional and Hospital Use Only
Single Use
BETASEPT®
Antiseptic Non-Sterile Surgical Scrub
Chlorhexidine Gluconate 4%
Net Contents: 4 fl oz (118 mL)
Dist. by: Atlantis Consumer Healthcare Inc. Bridgewater, NJ 08807 USA
©2023 Atlantis Consumer Healthcare Inc.
4BET04BTLLBLH
NDC 67618-200-08
From the Makers of BETADINE® products
For External Use Only
Not for Retail Sale
For Professional and Hospital Use Only
BETASEPT®
Antiseptic Surgical Scrub
Chlorhexidine Gluconate 4%
Net Contents: 8 fl oz (237 mL)
Dist. by:Atlantis Consumer Healthcare Inc. Bridgewater, NJ 08807 USA
©2023 Atlantis Consumer Healthcare Inc.
4BET08BTLLBLH
NDC 67618-200-16
From the Makers of BETADINE® products
For External Use Only
Not for Retail Sale
For Professional and Hospital Use Only
BETASEPT®
Antiseptic Surgical Scrub
Chlorhexidine Gluconate 4%
Net Contents: 16 fl oz (473 mL)
Dist. by: Atlantis Consumer Healthcare Inc. Bridgewater, NJ 08807 USA
©2023 Atlantis Consumer Healthcare Inc.
4BET16BTLLBLC

NDC 67618-200-32
From the Makers of BETADINE® products
For External Use Only
Not for Retail Sale
For Professional and Hospital Use Only
BETASEPT®
Antiseptic Surgical Scrub
Chlorhexidine Gluconate 4%
Net Contents: 32 fl oz (946 mL)
Dist. by: Atlantis Consumer Healthcare Inc. Bridgewater, NJ 08807 USA
©2023 Atlantis Consumer Healthcare Inc.
4BET32PBTLLBLC

| BETASEPT
antiseptic surgical scrub solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Xttrium Laboratories, Inc. (007470579) |
More about Betasept (chlorhexidine topical)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Latest FDA alerts (3)
- Side effects
- Dosage information
- During pregnancy
- Drug class: antiseptic and germicides
- Breastfeeding
- En español
Patient resources
Professional resources
Other brands
Bactoshield CHG, ChloraPrep One-Step, CliniClean, Acclean
