Alprolix: Package Insert / Prescribing Info
Package insert / product label
Generic name: coagulation factor ix (recombinant), fc fusion protein
Dosage form: injection
Drug class: Miscellaneous coagulation modifiers
J Code (medical billing code): J7201 (Per intl units, injection)
Medically reviewed by Drugs.com. Last updated on May 26, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
ALPROLIX® [coagulation factor IX (recombinant), Fc fusion protein], lyophilized powder for solution for intravenous injection.
Initial U.S. Approval: 2014
Indications and Usage for Alprolix
ALPROLIX, Coagulation Factor IX (Recombinant), Fc Fusion Protein, is a recombinant DNA derived coagulation Factor IX concentrate indicated in adults and children with hemophilia B for:
- On-demand treatment and control of bleeding episodes,
- Perioperative management of bleeding,
- Routine prophylaxis to reduce the frequency of bleeding episodes.
Limitation of Use:
ALPROLIX is not indicated for induction of immune tolerance in patients with hemophilia B. (1)
Alprolix Dosage and Administration
For intravenous use after reconstitution only.
On-demand treatment and control of bleeding episodes:
- Each vial of ALPROLIX contains the labeled amount of coagulation Factor IX potency in international units (IU). (2.1)
- On average, one unit per kilogram body weight of ALPROLIX increased the circulating Factor IX level by approximately 1% (IU/dL) in adults and children ≥6 years of age and by 0.6% (IU/dL) in children under 6 years of age. (2.1)
| Initial Dose: Type of Bleeding | Target Circulating FIX (IU/dL) | Dosing Interval (hours) |
|---|---|---|
| Minor and Moderate | 30–60 | Repeat every 48 hours as needed if there is further evidence of bleeding. |
| Major | 80–100 | Consider repeat dose after 6–10 hours, then every 24 hours for 3 days, then every 48 hours until healing achieved. |
Perioperative management:
- Minor surgery: a single infusion to reach FIX level of 50–80 IU/dL may be sufficient. Repeat as needed after 24–48 hours until bleeding stops and healing is achieved. (2.1)
- Major surgery: initial infusion to reach FIX level of 60–100 IU/dL. Consider a repeat dose after 6–10 hours and then every 24 hours for the first 3 days, then every 48 hours until bleeding stops and healing is achieved. (2.1)
Routine prophylaxis:
For adults and adolescents ≥12 years of age, start at 50 IU/kg once weekly or 100 IU/kg once every 10 days. For children <12 years of age, start at 60 IU/kg once weekly. Adjust dosing regimen based on individual response. More frequent or higher doses may be needed in children <12 years of age. (2.1)
Dosage Forms and Strengths
ALPROLIX is available as a lyophilized powder in single-dose vials containing nominally 250, 500, 1000, 2000, 3000, or 4000 international units (IU). (3)
Contraindications
Do not use in individuals who have a known history of hypersensitivity reactions, including anaphylaxis, to the product or its excipients. (4)
Warnings and Precautions
- Hypersensitivity reactions, including anaphylaxis, have been reported. Should symptoms occur, discontinue ALPROLIX and administer appropriate treatment. (5.1)
- Neutralizing antibodies (inhibitors) to FIX have been reported with ALPROLIX. Perform an assay that measures Factor IX inhibitor concentration if plasma Factor IX level fails to increase as expected or if bleeding is not controlled with an appropriate dose. (5.2, 5.4)
- The use of Factor IX products has been associated with the development of thromboembolic complications. (5.3)
- Nephrotic syndrome has been reported following immune tolerance induction with Factor IX-containing products in hemophilia B patients with Factor IX inhibitors and a history of allergic reactions to Factor IX. (5.5)
Adverse Reactions/Side Effects
Previously Treated Patients (PTPs): Common adverse reactions (incidence ≥1%) from clinical trials were headache, oral paresthesia, and obstructive uropathy. (6)
Previously Untreated Patients (PUPs): Common adverse reactions (incidence ≥1%) from clinical trial were Factor IX inhibition, injection site erythema, and hypersensitivity. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Bioverativ Therapeutics Inc. at 1-855-692-5776 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Use In Specific Populations
Pediatric: Higher dose per kilogram body weight or more frequent dosing may be needed in patients <12 years of age. (8.4)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 5/2023
Full Prescribing Information
1. Indications and Usage for Alprolix
ALPROLIX, Coagulation Factor IX (Recombinant), Fc Fusion Protein, is a recombinant DNA derived coagulation Factor IX concentrate indicated in adults and children with hemophilia B (congenital Factor IX deficiency) for:
- On-demand treatment and control of bleeding episodes,
- Perioperative management of bleeding,
- Routine prophylaxis to reduce the frequency of bleeding episodes.
2. Alprolix Dosage and Administration
For intravenous use after reconstitution only
2.1 Dose
- Dose and duration of treatment depend on the severity of the Factor IX deficiency, the location and extent of bleeding, the individual patient's pharmacokinetic profile, and/or the patient's clinical condition.
- Patients may vary in their pharmacokinetic (e.g., half-life, in vivo recovery) and clinical responses. Base the dose and frequency of ALPROLIX on the individual clinical response.
- More frequent or higher doses may be needed in children <12 years of age, especially in children <6 years of age [see Use in Specific Populations (8.4)]. For patients 12 years of age or older, age-based dose adjustment is not usually required.
- In addition to the nominal (target) potency, the actual Factor IX potency in international units (IU), determined by the quality control laboratory at product release, is stated on each ALPROLIX vial label. ALPROLIX potency is assigned using a validated in vitro, activated partial thromboplastin time (aPTT)–based, one-stage clotting assay calibrated against the World Health Organization (WHO) international standard for Factor IX concentrates.
- Factor IX activity measurements in the clinical laboratory may be affected by the type of aPTT reagent or laboratory standard used [see Warnings and Precautions (5.4)].
On average, one IU of ALPROLIX per kg body weight increases the circulating level of Factor IX by approximately 1% (IU/dL) in adults and children ≥6 years of age and by 0.6% (IU/dL) in children under 6 years of age. Estimate the required dose or the expected in vivo peak increase in Factor IX level expressed as IU/dL (or % of normal) using the following formulas:
| IU/dL (or % of normal) = (Total Dose [IU]/Body Weight [kg]) × Recovery (IU/dL per IU/kg) OR Dose (IU) = Body Weight (kg) × Desired Factor IX Rise (IU/dL or, % of normal) × Reciprocal of Recovery (IU/kg per IU/dL) |
- Consider determining the patient's in vivo recovery (in IU/dL per IU/kg) prior to elective major surgery and verify that the target Factor IX level has been achieved prior to major surgery and for major bleeds.
On-demand Treatment and Control of Bleeding Episodes
ALPROLIX dosing for on-demand treatment and control of bleeding episodes is provided in Table 1.
| Type of Bleeding | Circulating Factor IX Level Required (IU/dL or % of normal) | Dosing Interval (hours) |
|---|---|---|
| Minor and Moderate
For example: Uncomplicated hemarthroses, superficial muscle (except iliopsoas) without neurovascular compromise, superficial soft tissue, mucous membranes | 30–60 | Repeat every 48 hours if there is further evidence of bleeding. |
| Major
For example: Iliopsoas and deep muscle with neurovascular injury, or substantial blood loss; Pharyngeal, retropharyngeal, retroperitoneal, CNS | 80–100 | Consider a repeat dose after 6–10 hours and then every 24 hours for the first 3 days. Due to the long half-life of ALPROLIX, the dose may be reduced and frequency of dosing may be extended after day 3 to every 48 hours or longer until bleeding stops and healing is achieved. |
Perioperative Management of Bleeding
ALPROLIX dosing for perioperative management is provided in Table 2.
| Type of Surgery | Circulating Factor IX Level Required (IU/dL or % of normal) | Dosing Interval (hours) |
|---|---|---|
| Minor
(including uncomplicated dental extraction) | 50–80 | A single infusion may be sufficient. Repeat as needed after 24–48 hours until bleeding stops and healing is achieved. |
| Major | 60–100 (initial level) | Consider a repeat dose after 6–10 hours and then every 24 hours for the first 3 days. Due to the long half-life of ALPROLIX, the dose may be reduced and frequency of dosing in the post-surgical setting may be extended after day 3 to every 48 hours or longer until bleeding stops and healing is achieved. |
Routine Prophylaxis
- The recommended starting regimens for adults and adolescents ≥12 years of age are either 50 IU/kg once weekly, or 100 IU/kg once every 10 days.
- For children <12 years of age, start with 60 IU/kg once weekly.
- Adjust dosing regimen based on individual response. More frequent or higher doses may be needed in children <12 years of age, especially in children <6 years of age.
2.2 Reconstitution
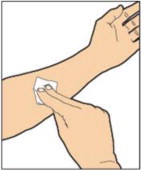
- Use aseptic technique (clean and germ-free) and a flat work surface during the reconstitution procedure.
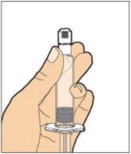
- Allow the vial of ALPROLIX, containing the white to off-white lyophilized powder and the prefilled diluent syringe to reach room temperature before use.
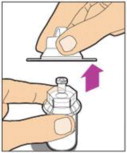
- Remove the plastic cap from the vial and wipe the rubber stopper of the vial with an alcohol wipe. Allow the rubber stopper to dry.
-
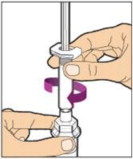
Completely remove the backing from the vial adapter package by peeling back the lid. Do not remove the vial adapter from the package or touch the inside of the package of the adapter.
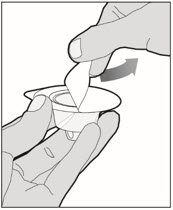
-
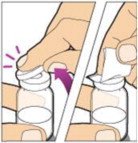
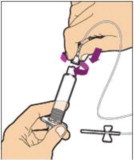
Place the vial on a flat surface and use one hand to hold the vial steady. Use the other hand to place the vial adapter over the vial. Place the adapter spike directly above the center of the rubber stopper and push the adapter straight down until the spike punctures the center of the vial stopper and is fully inserted.
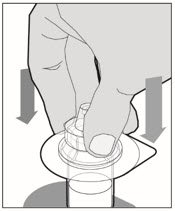
-
Lift the package cover away from the vial adapter and discard the cover.
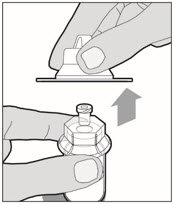
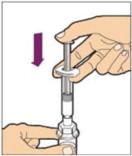
-
Hold the plunger rod at the circular disk. Place the tip of the plunger rod into the end of the syringe. Turn clockwise until it is securely attached. Only use the diluent syringe provided in the ALPROLIX package.
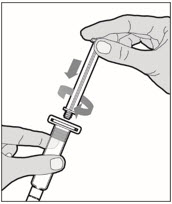
- With one hand, hold the diluent syringe by the ridged part directly under the cap, with the cap pointing up. Do not use if the cap has been removed or is not securely attached.
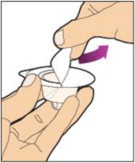
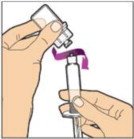
- With your other hand, grasp the cap and bend it at a 90° angle until it snaps off. After the cap snaps off, you will see the glass tip of the syringe. Do not touch the glass tip of the syringe or the inside of the cap.
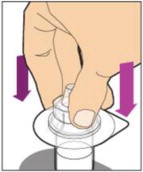
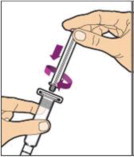
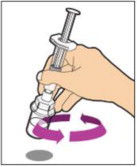
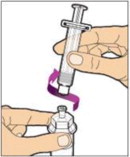
- With the vial sitting on a flat surface, insert the tip of the syringe into the adapter opening. Turn the syringe clockwise until it is securely attached to the adapter.
- Slowly depress the plunger rod to inject all of the diluent into the vial. The plunger rod may rise slightly after this process. This is normal.
- With the syringe still connected to the adapter, gently swirl the vial until the product is completely dissolved. The final solution should be clear to slightly opalescent and colorless. Do not shake. Do not use the reconstituted ALPROLIX if it contains visible particles or is cloudy.
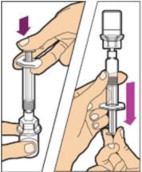
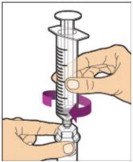
- Make sure the plunger rod is completely depressed. Turn the vial upside-down. Slowly pull on the plunger rod to draw the solution into the syringe. Be careful not to pull the plunger rod completely out of the syringe.
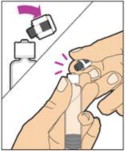
- Gently unscrew the syringe from the vial adapter and dispose of the vial with the adapter still attached. Do not touch the syringe tip or the inside of the cap.
- Use the reconstituted ALPROLIX as soon as possible, but no later than 3 hours after reconstitution. Protect from direct sunlight. Do not refrigerate after reconstitution.
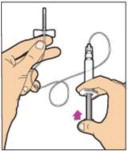
To combine two or more vials of ALPROLIX, after step 12 above, follow these pooling steps:
- Remove the diluent syringe from the vial adapter by turning it counterclockwise until it is completely detached. Do not detach the diluent syringe or the large luer lock syringe until ready to attach the large luer lock syringe to the next vial (with vial adapter attached).
- Leave the vial adapter attached to the vial, as it is needed for attaching a large luer lock syringe.
- Attach a separate, large luer lock syringe by turning clockwise until it is securely in place.
- Slowly pull on the plunger rod to draw the solution into the syringe.
- Repeat this pooling procedure with each vial necessary to obtain the required dose. Once you have pooled the required dose, proceed to administration using the large luer lock syringe.
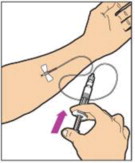
2.3 Administration
For intravenous injection only
- Inspect the reconstituted ALPROLIX solution visually for particulate matter and discoloration prior to administration. Do not use if particulate matter or discoloration is observed.
- Do not administer reconstituted ALPROLIX in the same tubing or container with other medications.
Administration Steps:
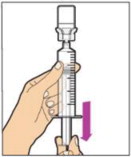
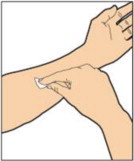
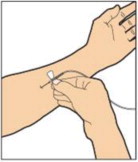
- Attach the syringe to the connector end of the infusion set tubing by turning clockwise until it is securely in place.
- Depress the plunger until all air is removed from the syringe and ALPROLIX has reached the end of the infusion set tubing. Do not push ALPROLIX through the needle.
- Remove the protective needle cover from the infusion set tubing.
- Perform intravenous bolus infusion. The rate of administration should be determined by the patient's comfort level, and no faster than 10 mL per minute.
After infusing ALPROLIX, remove and properly discard the infusion set.
3. Dosage Forms and Strengths
ALPROLIX is available as a white to off-white lyophilized powder in single-dose vials containing nominally (approximately) 250, 500, 1000, 2000, 3000, or 4000 international units (IU) of Factor IX potency per vial. The actual Factor IX potency is stated on each ALPROLIX vial.
4. Contraindications
ALPROLIX is contraindicated in individuals who have a known history of hypersensitivity reactions, including anaphylaxis, to the product or its excipients (sucrose, mannitol, sodium chloride, L-histidine and polysorbate 20).
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Allergic-type hypersensitivity reactions, including anaphylaxis, have been reported with ALPROLIX.
The presence of inhibitors has been associated with allergic reactions with Factor IX replacement therapies, including with ALPROLIX. Evaluate patients experiencing allergic reactions for the presence of an inhibitor. Early signs of allergic reactions, which can progress to anaphylaxis, may include angioedema, chest tightness, hypotension, rash, nausea, vomiting, paresthesia, restlessness, wheezing and dyspnea. Discontinue use of ALPROLIX if hypersensitivity symptoms occur, and initiate appropriate treatment.
5.2 Neutralizing Antibodies
Formation of neutralizing antibodies (inhibitors) to Factor IX has been reported following administration of ALPROLIX. Monitor all patients regularly for the development of inhibitors by appropriate clinical observations and laboratory tests [see Warnings and Precautions (5.4)].
Evaluate patients experiencing allergic reactions for the presence of an inhibitor. Closely observe patients for signs and symptoms of acute hypersensitivity reactions, particularly during the early phases of exposure to the product.
Individuals with Factor IX inhibitors may be at an increased risk of anaphylaxis upon subsequent challenge with ALPROLIX.
5.3 Thromboembolic Complications
The use of Factor IX products has been associated with the development of thromboembolic complications, especially in individuals receiving continuous infusion through a central venous catheter. ALPROLIX should be administered as bolus infusion over several minutes [see Dosage and Administration (2.3)]. The safety of ALPROLIX administration by continuous infusion has not been studied.
5.4 Monitoring Laboratory Tests
- To confirm adequate Factor IX levels have been achieved and maintained, monitor patient plasma Factor IX levels by performing a validated one-stage clotting assay [see Dosage and Administration (2.1)]. Factor IX results can be affected by the type of aPTT reagent used. Measurement with a one-stage clotting assay using a kaolin-based aPTT reagent has been shown to result in an underestimation of Factor IX levels.
- Monitor for the development of Factor IX inhibitors if the expected Factor IX levels in patient plasma are not attained, or if bleeding is not controlled with the recommended dose of ALPROLIX. Perform a Bethesda assay to determine if Factor IX inhibitors are present.
5.5 Nephrotic Syndrome
Nephrotic syndrome has been reported following attempted immune tolerance induction in hemophilia B patients with Factor IX inhibitors and a history of allergic reactions to Factor IX. The safety and efficacy of using ALPROLIX for immune tolerance induction have not been established.
6. Adverse Reactions/Side Effects
The most common adverse reactions (incidence ≥1%) in previously untreated patients (PUPs) were injection site erythema, hypersensitivity, and Factor IX inhibition [see Warnings and Precautions (5.1), (5.2)]. The most common adverse reactions (incidence ≥1%) in previously treated patients (PTPs) were headache, oral paresthesia, and obstructive uropathy.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
Previously Treated Patients (PTPs)
ALPROLIX has been evaluated in three completed studies (an adult and adolescent study, a pediatric study, and an extension study) in previously treated patients (PTPs) with severe to moderately severe hemophilia B (≤2% endogenous FIX activity). Of the 153 subjects treated, 30 (20%) were children (1 to 11 years of age), 11 (7%) were adolescents (12 to 17 years of age), and 112 (73%) were adults (18 to 71 years of age). There were 126 subjects (82.4%) treated for at least 52 weeks, 107 subjects (69.9%) for at least 104 weeks, and 67 (43.8%) treated for at least 208 weeks. The total number of exposure days (EDs) was 26,106 with a median of 165 (range 1–528) EDs per subject. Adverse events were monitored for a total of 561 subject-years.
Adverse reactions (ARs) were reported in 14 of 153 (9.2%) subjects treated with ALPROLIX. They are summarized in Table 3.
No subject was withdrawn from the trials due to an adverse reaction. In these PTP trials, no inhibitors were detected and no events of anaphylaxis or vascular thromboembolism were reported.
| System Organ Class | Adverse Reactions | Number of Subjects (%) N=153* |
|---|---|---|
| Nervous system disorders | Headache Dizziness Dysgeusia | 2 (1.3) 1 (0.7) 1 (0.7) |
| Gastrointestinal disorders | Paresthesia oral Breath odor | 2 (1.3) 1 (0.7) |
| General disorders and administration site conditions | Fatigue Infusion site pain | 1 (0.7) 1 (0.7) |
| Cardiac disorders | Palpitations | 1 (0.7) |
| Renal and urinary disorders | Obstructive uropathy† | 2 (1.3) |
| Hematuria | 1 (0.7) | |
| Renal colic | 1 (0.7) | |
| Vascular disorders | Hypotension | 1 (0.7) |
| Metabolism and nutritional disorders | Decreased appetite | 1 (0.7) |
Previously Untreated Patients (PUPs)
ALPROLIX safety was also evaluated in one completed study (PUPs study) in 33 subjects with hemophilia B (≤2% endogenous FIX activity). Overall, the median number of weeks on treatment was 83.01 (range: 6.7–226.7 weeks). The number of subjects with at least 10 EDs was 28 (84.8%), at least 20 EDs was 26 (78.8%), and at least 50 EDs was 21 (63.6%). The median number of EDs was 76 (range: 1–137) per subject.
Adverse events were monitored for a total of 57.51 subject-years. Adverse drug reactions were reported in 2 of 33 (6.1%) subjects treated with ALPROLIX. One subject (3.0%) in the PUP study developed the SAE of Factor IX inhibition and the same subject (3.0%) developed hypersensitivity. One additional subject (3.0%) in the PUP study developed injection site erythema.
Immunogenicity
Clinical trial subjects were monitored for neutralizing antibodies to Factor IX, and no inhibitors were detected in PTPs during the study. Of 33 PUPs, a total of 1 subject (3.0%) developed a low-titer neutralizing Factor IX inhibitor.
The detection of antibodies that are reactive to Factor IX is highly dependent on many factors, including the sensitivity and specificity of the assay, assay methodology, sample handling, timing of sample collection, concomitant medications and underlying disease. Therefore, it may be misleading to compare the incidence of antibodies to ALPROLIX with the incidence of antibodies to other products.
6.2 Postmarketing Experience
The following adverse reactions have been identified during the postapproval use of ALPROLIX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Factor IX inhibitor development
Immune system disorders: hypersensitivity, including anaphylaxis
For additional information, refer to the Warnings and Precautions 5.1 and 5.2.
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are no studies of ALPROLIX use in pregnant women to inform a drug-associated risk.
The background risk of major birth defects and miscarriage for the indicated population is unknown; however, the background risk in the U.S. general population of major birth defects is 2%–4% and of miscarriage is 15%–20% of clinically recognized pregnancies.
Animal reproductive and developmental toxicity studies have not been conducted with ALPROLIX. In a placental transfer study, ALPROLIX was detected in murine fetal blood samples at approximately 2.6% of the maternal blood levels (range, 1.7% to 3.3%), 3 to 4 hours following dosing of pregnant mice with 3.3 to 6.6 times the clinical dose of 50 to 100 IU/kg ALPROLIX [see Data].
It is not known whether ALPROLIX can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. If ALPROLIX is clearly needed to treat a pregnant woman, advise the patient that the risks to the mother and to the fetus are unknown.
Data
Animal data
Pregnant, genetically-modified, FIX-deficient mice (HemB mice) were injected intravenously with a single dose of 330 IU/kg ALPROLIX at the end of pregnancy on Gestation Day 18, or with repeat doses of 330 IU/kg ALPROLIX on Gestation Days 18 and 20. Blood samples were collected from the maternal mice and the fetuses after dosing, and FIX activity was measured in both maternal and fetal plasma using a FIX chromogenic assay. After dosing pregnant HemB mice with ALPROLIX, FIX activity in fetal blood was approximately 2.6% of the maternal blood levels, suggesting that placental transfer of ALPROLIX may occur in pregnant female patients. The relevance of these data to humans is unknown.
8.2 Lactation
Risk Summary
It is not known whether ALPROLIX is excreted into human milk. There are no data available to assess the effects of ALPROLIX on milk production or the breastfed child.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ALPROLIX and any potential adverse effects on the breastfed child from ALPROLIX or from the underlying maternal condition.
8.4 Pediatric Use
Safety, efficacy, and pharmacokinetics of ALPROLIX have been evaluated in previously treated patients (PTPs) from the adult and adolescent study (12 to <17 years of age) and from the pediatric study (1 to 11 years of age) [see Adverse Reactions (6), Clinical Trials Experience (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14)]. Safety of ALPROLIX has been evaluated in previously untreated patients (PUPs) less than 18 years of age (median: 0.6 year; range: 0.08–2 years) in the PUPs study [see Adverse Reactions (6), Clinical Trials Experience (6.1), and Clinical Pharmacology (12.3)].
No dose adjustment is required for adolescents. Children under 12 years of age may have higher Factor IX body weight-adjusted clearance and lower recovery. More frequent or higher doses may be needed in children <12 years of age. When calculating target peak doses for treatment of bleeding or surgery, use the average in vivo recovery value of 0.6 IU/dL per IU/kg, or individually determined in vivo recovery, for children under 6 years of age [see Clinical Pharmacology (12.3)].
11. Alprolix Description
ALPROLIX is a sterile, non-pyrogenic, preservative-free, white to off-white, lyophilized powder for reconstitution with the diluent for intravenous injection. After reconstitution, the solution has a clear to slightly opalescent appearance and contains the excipients sucrose, mannitol, sodium chloride, L-histidine and polysorbate 20. ALPROLIX is available in single-dose vials containing nominally (approximately) 250 IU, 500 IU, 1000 IU, 2000 IU, 3000 IU, or 4000 IU of recombinant Factor IX. The actual potency determined by the quality control laboratory at product release is stated directly on each vial label.
Coagulation Factor IX (Recombinant), Fc Fusion Protein (rFIXFc), the active ingredient in ALPROLIX, is a recombinant coagulation Factor IX fusion protein consisting of the human coagulation Factor IX sequence covalently linked to the Fc domain of human immunoglobulin G1 (IgG1). The Factor IX portion of rFIXFc has a primary amino acid sequence that is identical to the Thr148 allelic form of plasma derived Factor IX and has structural and functional properties similar to endogenous Factor IX. The Fc domain of rFIXFc contains the hinge, CH2, and CH3 regions of IgG1. rFIXFc contains 867 amino acids with a molecular weight of approximately 98 kilodaltons.
ALPROLIX is not derived from human blood and contains no preservatives. The recombinant Factor IX Fc fusion protein is expressed in a human embryonic kidney (HEK) cell line, which produces rFIXFc into a defined cell culture medium that does not contain proteins derived from animal or human sources. The purification process for rFIXFc does not include use of a monoclonal antibody reagent. To enhance viral safety, the production process also incorporates two dedicated viral clearance steps – a detergent treatment step for inactivation and a 15 nm filtration step for removal of viruses. The content of activated Factor IX Fc fusion protein (FIXaFc) is limited to ≤0.035 mole percent FIXaFc/FIXFc.
12. Alprolix - Clinical Pharmacology
12.1 Mechanism of Action
ALPROLIX is a recombinant, fusion protein that temporarily replaces the missing coagulation Factor IX needed for effective hemostasis. ALPROLIX contains the Fc region of human IgG1, which binds to the neonatal Fc receptor (FcRn). FcRn is part of a naturally occurring pathway that delays lysosomal degradation of immunoglobulins by cycling them back into circulation, and prolonging their plasma half-life.
12.2 Pharmacodynamics
Hemophilia B is a bleeding disorder characterized by a deficiency of functional coagulation Factor IX (FIX), which leads to a prolonged clotting time in the activated partial thromboplastin time (aPTT) assay, an established in vitro test for the biological activity of Factor IX. Treatment with ALPROLIX shortens the aPTT over the effective dosing period.
12.3 Pharmacokinetics
PK parameters for ALPROLIX were estimated based on the plasma FIX activity measured by the one-stage clotting assay.
Adults (≥18 Years)
The pharmacokinetics (PK) of ALPROLIX (rFIXFc) were evaluated in 22 adults (≥18 years of age) following a 10-minute intravenous infusion of a single dose of 50 IU/kg. Blood samples for PK analysis were collected prior to dosing and up to 240 hours (10 days) after dosing with 50 IU/kg. In addition, PK was assessed for 27 patients receiving the 100 IU/kg dose (Table 4). Blood samples were collected up to 336 hours after dosing. Table 4 presents the PK parameters in adults. The pharmacokinetics of ALPROLIX following single and repeat dosing (at week 26) were similar.
| PK Parameters | ALPROLIX (50 IU/kg) (N=22) | ALPROLIX (100 IU/kg) (N=24) |
|---|---|---|
| Abbreviations: CV = coefficient of variation; IR = incremental recovery; AUCinf = area under the FIX activity time curve to infinity; Terminal T1/2 = terminal phase elimination half-life; MRT = mean residence time; CL = body weight adjusted clearance; Vss = body weight adjusted volume of distribution at steady-state; Time to 1% FIX activity = estimated time in days after dose when FIX activity has declined to approximately 1 IU/dL above baseline | ||
| Cmax (IU/dL) | 46 (68%) | 101 (20%) |
| AUCinf (h*IU/dL) | 1619 (26%) | 3964 (19%) |
| CL (mL/h/kg) | 3.3 (28%) | 2.6 (23%) |
| Vss (mL/kg) | 327 (28%) | 236 (24%) |
| Terminal T1/2 (h) | 86 (37%) | 97 (35%) |
| MRT (h) | 102 (30%) | 91 (23%) |
| IR (IU/dL per IU/kg) | 1.02 (59%) | 1.12 (22%) |
| Time to 1% FIX activity (d) | 12 (24%) | 16 (20%) |
Adolescents (12 to 17 Years)
Pharmacokinetic parameters were evaluated following a 10-minute intravenous infusion in 11 evaluable adolescents (12 to 17 years of age) who received a single dose of 50 IU/kg or 100 IU/kg of ALPROLIX. Blood samples were collected prior to dosing and at multiple time points up to 336 hours (14 days) after dosing. PK parameters of ALPROLIX in adolescents are summarized in Table 5. The pharmacokinetics of ALPROLIX in adolescents are comparable with adults.
Pediatrics (2 to 10 Years)
In the pediatric study, pharmacokinetic parameters were evaluated following a 10-minute intravenous infusion in 24 evaluable children (2 to 10 years of age) who received a single 50 IU/kg dose of ALPROLIX. PK samples were collected prior to dosing and at multiple time points up to 168 hours (7 days) after dosing.
Table 5 presents the PK parameters in adolescents and children. Compared to adults, incremental recovery was lower and body weight adjusted clearance was higher in children under 12 years of age, particularly in children under 6 years of age. Incremental recovery in children 2 to 4 years and 6 to 10 years was lower by 41% and 27%, respectively. Compared to adults, body weight-adjusted clearance in children 2 to 4 years and 6 to 10 years was higher by 36% and 11%, respectively. This may result in a need for per kg body weight dose and/or interval adjustments in children under 12 years of age [see Use in Specific Populations (8.4)].
| PK Parameters | <6 years (range: 2 to 4 years) (N=11, 50 IU/kg) | 6 to <12 years (range: 6 to 10 years) (N=13, 50 IU/kg) | 12 to 17 years (N=8, 50 IU/kg) | 12 to 17 years (N=3, 100 IU/kg) |
|---|---|---|---|---|
|
Abbreviations: CV = Coefficient of Variation; IR = incremental recovery; AUCinf = area under the FIX activity time curve to infinity; Terminal T ½= terminal phase elimination half-life; MRT = mean residence time; CL = body weight adjusted clearance; Vss = body weight adjusted volume of distribution at steady-state |
||||
| Cmax (IU/dL) | 30 (19%) | 37 (29%) | 43 (44%) | 96 (9%) |
| IR (IU/dL per IU/kg) | 0.60 (20%) | 0.74 (29%) | 0.87 (44%) | 0.96 (10%) |
| AUCinf (h*IU/dL) | 1169 (15%) | 1471 (27%) | 1439 (24%) | 3420 (13%) |
| Terminal T½ (h) | 68 (24%) | 72 (23%) | 80 (15%) | 94 (24%) |
| MRT (h) | 86 (23%) | 84 (19%) | 95 (17%) | 95 (29%) |
| CL (mL/h/kg) | 4.4 (17%) | 3.6 (25%) | 3.7 (26%) | 3.0 (12%) |
| Vss (mL/kg) | 373 (23%) | 302 (29%) | 345 (24%) | 275 (20%) |
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate the carcinogenic potential of ALPROLIX, or studies to determine the effects of ALPROLIX on genotoxicity or fertility have not been performed. An assessment of the carcinogenic potential of ALPROLIX was completed and no carcinogenic risk from product use has been identified.
14. Clinical Studies
The safety and efficacy of ALPROLIX was evaluated in a multi-center, prospective, open-label study in previously treated patients (PTPs): adult and adolescent patients 12 to 71 years of age and a study in pediatric patients 1 to 11 years of age.
Adult and Adolescent Study (12 to 71 Years)
The non-randomized adult and adolescent study compared each of two prophylactic treatment regimens (fixed weekly and individualized interval) to episodic (on-demand) treatment; determined hemostatic efficacy in the treatment of bleeding episodes; and determined hemostatic efficacy during perioperative management of subjects undergoing major surgical procedures. A total of 123 previously treated patients (PTPs) with severe to moderately severe hemophilia B (≤2% endogenous FIX activity) were followed for up to 77 weeks. Subjects who were on prophylaxis before the study were assigned to one of the prophylaxis arms. Subjects who were treating on-demand before the study were assigned to either one of the two prophylaxis arms or to the on-demand treatment arm. Treatment arm assignments were based on standard of care and discussion between the investigator and the subject.
Sixty-three subjects in the fixed weekly interval arm received ALPROLIX for routine prophylaxis starting at an initial dose of 50 IU/kg. The dose was adjusted to maintain FIX trough level between 1% and 3% above baseline or higher, as clinically indicated to prevent bleeding. Fifty subjects (79%) required at least one dose adjustment and the median number of dose adjustments was one. The overall median dose on study was 45.2 IU/kg (interquartile range: 38.1, 53.7). The median weekly dose during the last 6 months on study in 58 subjects who were on study for at least 9 months was 40.7 IU/kg (interquartile range: 32.3, 54.1).
Twenty-nine subjects in the individualized interval arm initially received ALPROLIX for routine prophylaxis at a dose of 100 IU/kg every 10 days, with the interval adjusted to maintain FIX trough level between 1% and 3% above baseline or higher, as clinically indicated to prevent bleeding. The overall median interval on study was 12.5 days (interquartile range: 10.4, 13.4). The median interval during the last 6 months in 26 subjects who were on study for at least 9 months was 13.8 days (interquartile range: 10.5, 14.0).
Twenty-seven subjects received ALPROLIX as needed for the treatment of bleeding episodes in the episodic (on-demand) treatment arm.
Twelve subjects received ALPROLIX for perioperative management in 14 major surgical procedures. Major surgery was defined as any surgical procedure with or without general anesthesia in which a major body cavity was penetrated and exposed, or a substantial impairment of physical or physiological functions was produced. Four subjects in this arm did not participate in the other arms.
Pediatric Study (1 to 11 Years)
The pediatric study enrolled a total of 30 previously treated male pediatric patients with severe to moderately severe hemophilia B (≤2% endogenous FIX activity). Subjects were less than 12 years of age (15 were 1 to 5 years of age and 15 were 6 to 11 years of age). All subjects received treatment with ALPROLIX and were followed for up to 52 weeks.
All 30 subjects were treated with ALPROLIX on an individualized prophylactic dose regimen starting with 50–60 IU/kg every 7 days, with adjustment of dose to a maximum of 100 IU/kg and dosing interval adjusted between once and twice weekly. The median dosing interval was 7.0 days (interquartile range: 6.9 to 7.0) with no difference between the age cohorts. The median average weekly dose of ALPROLIX was 59.4 IU/kg and (interquartile range: 53.0 to 64.8) for subjects 1 to 5 years of age and 57.8 IU/kg (interquartile range: 51.7 to 65.0) for subjects 6 to 11 years of age. At the end of the trial, the median prescribed once weekly prophylactic dose was 60 IU/kg across both pediatric age subgroups (range: 40 to 70 IU/kg).
On-demand Treatment and Control of Bleeding Episodes
Adult and adolescent study (12 to 71 years)
A total of 636 bleeding events were observed by 114 subjects in the fixed weekly interval prophylaxis, individualized interval prophylaxis, and the episodic (on-demand) arms. The median total dose to treat a bleeding episode was 47.0 IU/kg (interquartile range: 33.3, 62.5). Assessment of response to each injection was recorded by subjects at 8–12 hours after treatment. Efficacy in control of bleeding episodes is summarized in Table 6.
| Bleeding Episodes | (N=636) |
|---|---|
| Number of injections to treat bleeding episodes | |
|
|
| 1 injection | 575 (90.4%) |
| 2 injections | 44 (6.9%) |
| 3 injections | 17 (2.7%) |
| Response to first injection* | (N=613) |
| Excellent or good | 513 (83.7%) |
| Moderate | 90 (14.7%) |
| None | 10 (1.6%) |
Pediatric study (1 to 11 years)
A total of 60 bleeding events were observed by 20 subjects during the study. The median total dose to treat a bleeding episode was 68.2 IU/kg (interquartile range: 50.9, 126.2). Assessment of response to each injection was recorded by subjects at 8 to 12 hours post treatment. Efficacy in control of bleeding episodes is summarized in Table 7.
| 1 to 5 Years | 6 to 11 Years | Total (1 to 11 Years) |
|
|---|---|---|---|
| Bleeding episodes | (N=22) | (N=38) | (N=60) |
| Number of injections to treat bleeding episodes | |||
|
|||
| 1 injection | 19 (86.4%) | 26 (68.4%) | 45 (75.0%) |
| 2 injections | 2 (9.1 %) | 8 (21.1%) | 10 (16.7%) |
| 3 injections | 1 (4.5%) | 4 (10.5%) | 5 (8.3%) |
| Response to first injection* | (N=53) | ||
| Excellent or good | 47 (88.7%) | ||
| Moderate | 5 (9.4%) | ||
| None | 1 (1.9%) | ||
Perioperative Management
Thirty-five major surgical procedures were performed in 22 subjects (age range: 10–63 years) in the adult and adolescent study and in the extension study. Of the 35 major surgeries, 28 surgeries (80.0%) required a single perioperative dose to maintain hemostasis during surgery. The median average dose per injection to maintain hemostasis during surgery was 94.7 IU/kg (range: 49 to 152). Perioperative Factor IX replacement with ALPROLIX was by bolus infusion only. The safety of continuous infusion was not evaluated.
Hemostasis was assessed by the investigator after surgery. Assessment of intraoperative and postoperative hemostatic response for major surgeries in the adult and adolescent study and the ongoing extension study are summarized in Table 8.
| Response | |||||
|---|---|---|---|---|---|
| Major Surgery | Number of Procedures (Number of Subjects)* | Excellent | Good | Fair | Poor/None |
| Ablation of Liver Lesion | 1 (1) | 1 | |||
| Arthroscopy | 2 (2) | 2 | |||
| Closure of Rectal Fistula | 1 (1) | 1 | |||
| Craniotomy† | 1 (1) | 1 | |||
| Dental Abscess | 1 (1) | 1 | |||
| Finger Amputation or Partial Amputation | 2 (1) | 2 | |||
| Hip Replacement or Repair | 2 (2) | 1 | 1 | ||
| Install or Removal of External Ilizarov Fixation | 2 (1) | 2 | |||
| Liver Transplant | 1 (1) | 1 | |||
| Liver Resection | 1 (1) | 1 | |||
| Orchiectomy | 1 (1) | 1 | |||
| Percutaneous-Ablation of Hepatic Carcinoma | 1 (1) | 1 | |||
| Patellar Resurfacing | 1 (1) | 1 | |||
| Pilonidal Cyst | 1 (1) | 1 | |||
| Pin Release | 1 (1) | 1 | |||
| Spinal Surgery | 1 (1) | 1 | |||
| Tendon Transfer in Right Arm | 1 (1) | 1 | |||
| Tonsillectomy | 1 (1) | 1 | |||
| Unilateral Ankle Fusion | 2 (2) | 2 | |||
| Unilateral Ankle Replacement or Revision | 1 (1) | 1 | |||
| Unilateral Knee Replacement or Revision | 8 (8) | 6 | 2 | ||
There were an additional 62 minor surgical procedures in 37 subjects in the adult and adolescent study, the pediatric study, and the extension study. Hemostatic response was assessed for 38 minor surgeries; 36 minor surgeries were rated as excellent or good and 2 as fair.
Routine Prophylaxis
Adult and adolescent study (12 to 71 years)
Using a negative binomial model, a reduction in annualized bleeding rate (ABR) of 83% (76%–89%) for subjects in the fixed weekly interval arm and a reduction of 87% (80%–92%) for subjects in the individualized interval arm compared to the episodic (on-demand) treatment arm was observed.
The median duration of treatment on study was 51.4 weeks (range <1–77). A comparison of the ABRs in subjects evaluable for efficacy is summarized in Table 9.
| Bleeding Episodes | Prophylaxis Fixed Weekly Interval (N=61) | Prophylaxis Individualized Interval (N=26) | Episodic (On-Demand) (N=27) |
|---|---|---|---|
|
|||
| Overall ABR
(IQR)* | 2.95 (1.01, 4.35) | 1.38 (0.00, 3.43) | 17.69 (10.77, 23.24) |
| Spontaneous ABR (IQR)* | 1.04 (0.00, 2.19) | 0.88 (0.00, 2.30) | 11.78 (2.62, 19.78) |
| Joint ABR
(IQR)* | 1.11 (0.00, 4.01) | 0.36 (0.00, 3.24) | 13.58 (6.13, 21.61) |
Pediatric study (1 to 11 years)
The median duration of treatment on study was 49.4 weeks (range 12–52). A comparison of the median ABRs in pediatric subjects evaluable for efficacy is summarized in Table 10.
| Bleeding Episodes | 1 to 5 Years (N=15) | 6 to 11 Years (N=15) | Total (<12 Years) (N=30) |
|---|---|---|---|
|
|||
| Overall ABR (IQR)* | 1.09 (0.00, 2.90) | 2.13 (0.00, 4.17) | 1.97 (0.00, 3.13) |
| Spontaneous ABR (IQR)* | 0.00 (0.00, 1.09) | 0.00 (0.00, 2.09) | 0.00 (0.00, 1.16) |
| Joint ABR (IQR)* | 0.00 (0.00, 0.00) | 1.06 (0.00, 2.09) | 0.00 (0.00, 1.12) |
16. How is Alprolix supplied
How Supplied
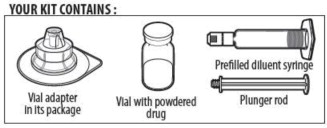
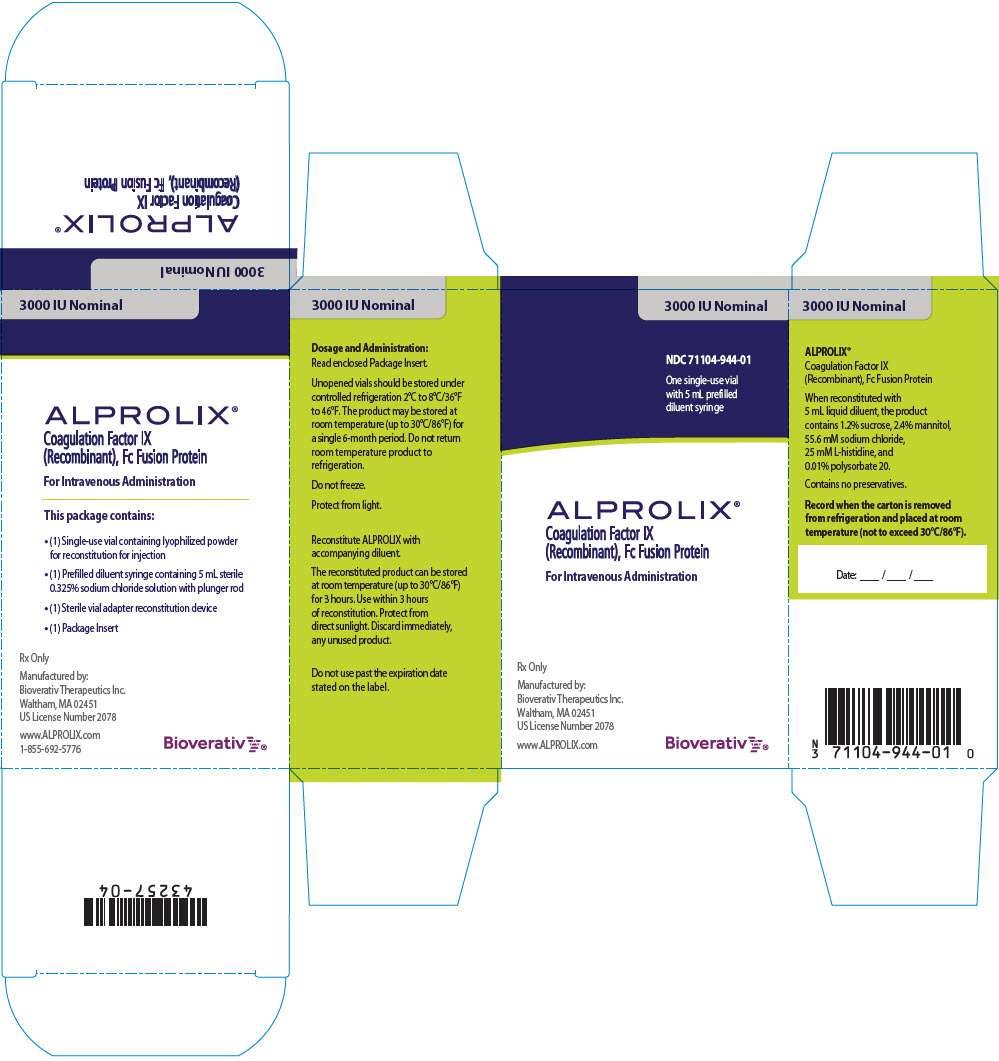
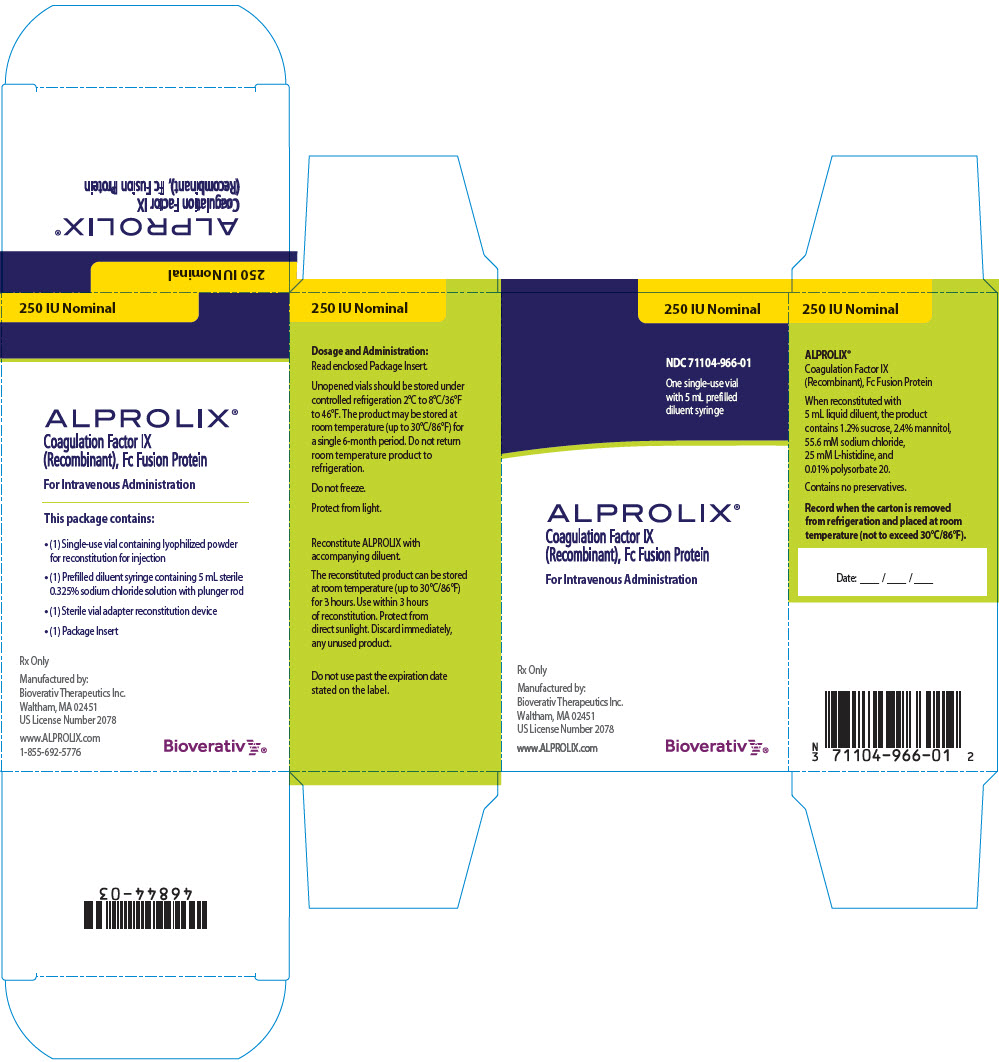
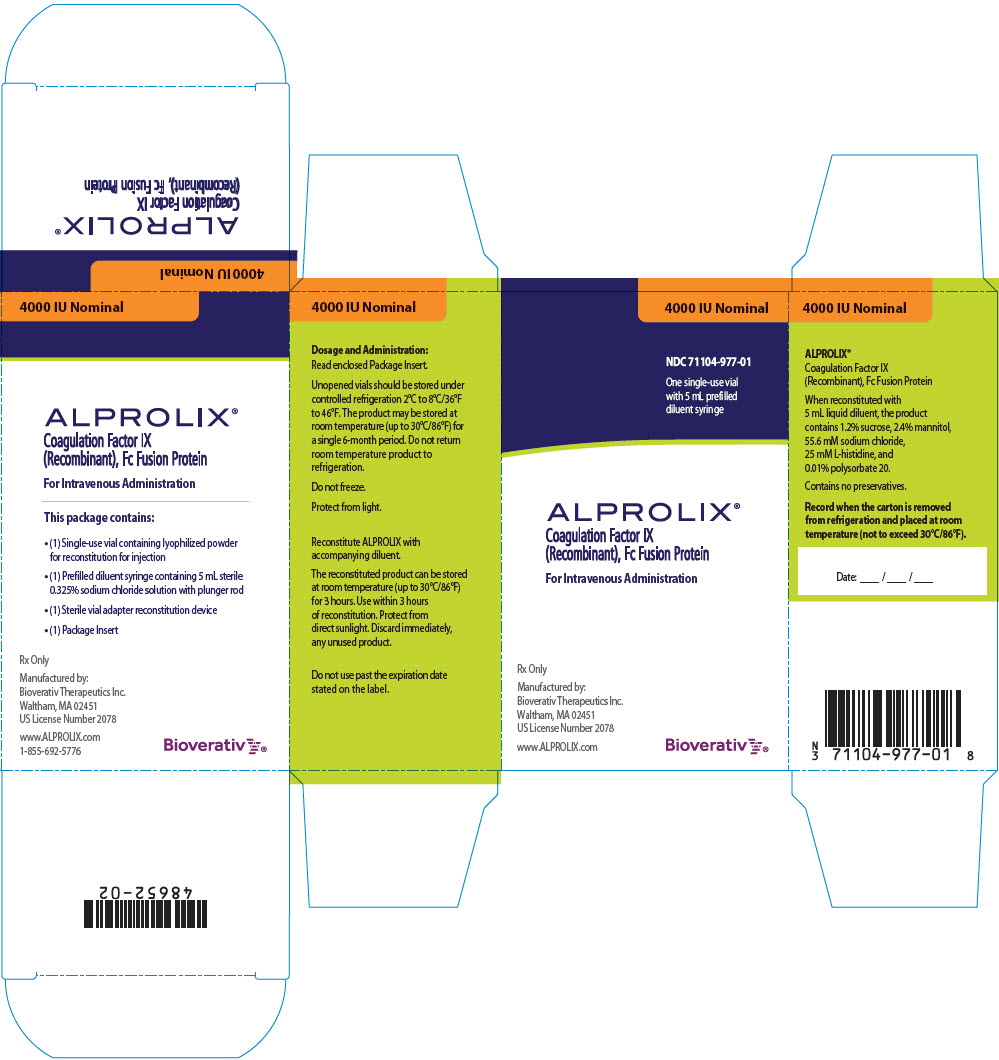
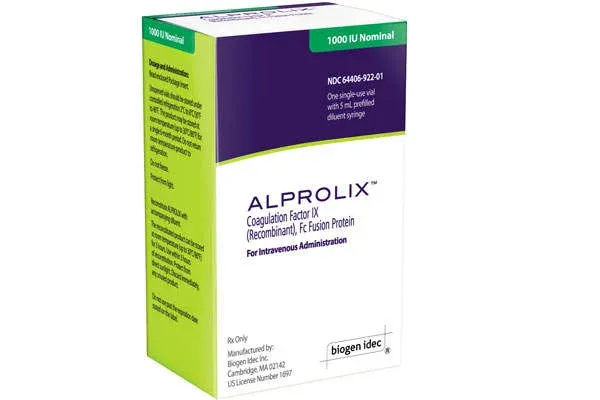
ALPROLIX is supplied as a kit comprising:
- one single-dose glass vial containing rFIXFc powder,
- one prefilled syringe containing 5 mL diluent and sealed with a plunger stopper and tip-cap, and
- one sterile vial adapter (reconstitution device).
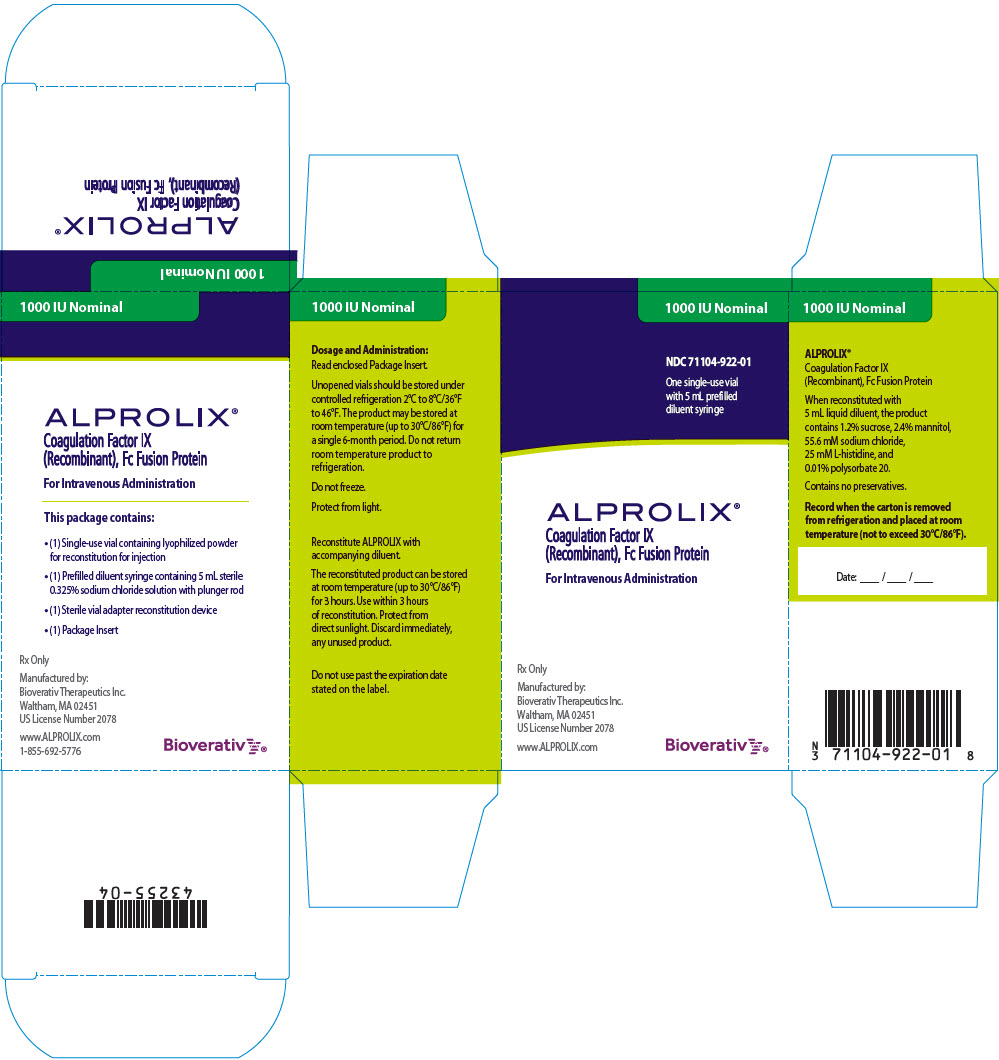
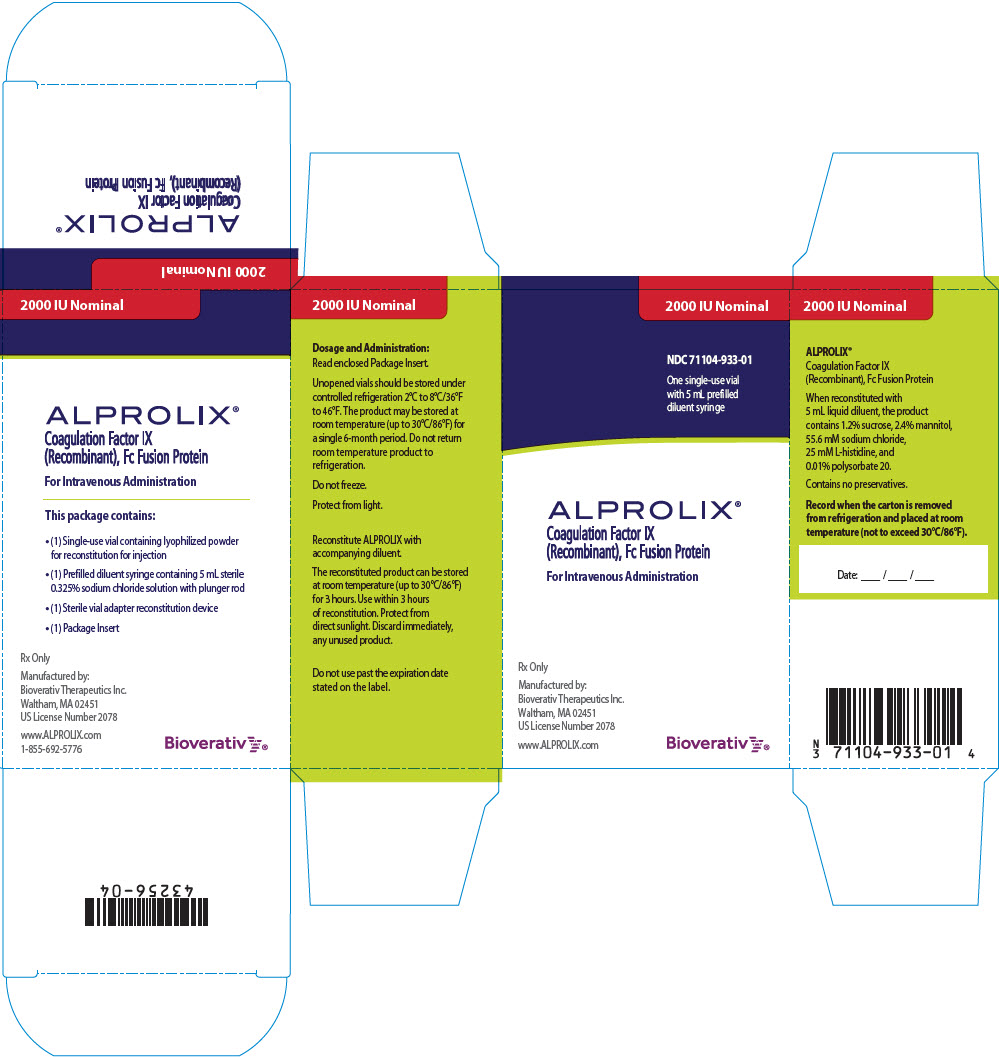
ALPROLIX is available in 250 IU, 500 IU, 1000 IU, 2000 IU, 3000 IU or 4000 IU nominal (approximate) dosage strengths. The actual Factor IX potency, expressed in IU, is stated on each rFIXFc vial and carton label. Not made with natural rubber latex.
| Nominal (Approximate) Strength | Potency Color Code | Kit NDC Number |
|---|---|---|
| 250 IU | Yellow | 71104-966-01 |
| 500 IU | Blue | 71104-911-01 |
| 1000 IU | Green | 71104-922-01 |
| 2000 IU | Red | 71104-933-01 |
| 3000 IU | Grey | 71104-944-01 |
| 4000 IU | Orange | 71104-977-01 |
Storage and Handling
- Store ALPROLIX in the original package in order to protect it from light.
- Store ALPROLIX at 2°C to 8°C (36°F to 46°F). Do not freeze. Freezing will damage the prefilled diluent syringe.
- ALPROLIX can be stored at room temperature, not to exceed 30°C (86°F), for a single period of up to 6 months within the expiration date printed on the carton and vial label. If stored at room temperature, record the date on the carton when the product was removed from refrigeration. Use the product before the end of this 6-month period or discard it. Do not place the product back into refrigeration after warming to room temperature. The shelf-life then expires after storage at room temperature for 6 months, or after the expiration date on the product vial, whichever is earlier.
- Do not use product or diluent after the expiration date printed on the carton, vial, or syringe.
- Reconstituted product may be stored at room temperature, not to exceed 30°C (86°F) for no longer than 3 hours. Protect from direct sunlight. Discard any product not used within 3 hours after reconstitution.
17. Patient Counseling Information
Advise the patient to:
- Read the FDA-approved patient labeling (Patient Information and Instructions for Use).
- Report any adverse reactions or problems following ALPROLIX administration to their physician or healthcare provider.
- Contact their healthcare provider or treatment facility for further treatment and/or assessment if they experience a lack of a clinical response to Factor IX therapy, as this may indicate the development of an inhibitor [see Warnings and Precautions (5.2)].
- Discontinue use of the product and contact their healthcare provider if early signs of hypersensitivity reactions (including hives, chest tightness, wheezing, difficulty breathing, and swelling of the face) and anaphylaxis occur. [see Warnings and Precautions (5.1)].
- Contact their healthcare provider or seek emergency care immediately if a thrombotic/thromboembolic event should occur [see Warnings and Precautions (5.3)].
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
A SANOFI COMPANY
US License Number 2078
©2023 Bioverativ Therapeutics Inc. All rights reserved.
For patent information: https://www.sanofi.us/en/products-and-resources/patents
Patient Information
ALPROLIX® /all' prō liks /
[Coagulation Factor IX (Recombinant), Fc Fusion Protein]
Please read this Patient Information carefully before using ALPROLIX and each time you get a refill, as there may be new information. This Patient Information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is ALPROLIX?
ALPROLIX is an injectable medicine that is used to help control and prevent bleeding in people with hemophilia B. Hemophilia B is also called congenital Factor IX deficiency.
Your healthcare provider may give you ALPROLIX when you have surgery.
Who should not use ALPROLIX?
You should not use ALPROLIX if you are allergic to ALPROLIX or any of the other ingredients in ALPROLIX. Tell your healthcare provider if you have had an allergic reaction to any Factor IX product prior to using ALPROLIX.
What should I tell my healthcare provider before using ALPROLIX?
Tell your healthcare provider about all of the medicines you take, including all prescription and non-prescription medicines, such as over-the-counter medicines, supplements, or herbal medicines.
Tell your doctor about all of your medical conditions, including if you:
- are pregnant or planning to become pregnant. It is not known if ALPROLIX may harm your unborn baby.
- are breastfeeding. It is not known if ALPROLIX passes into breast milk or if it can harm your baby.
- have been told that you have inhibitors to Factor IX (because ALPROLIX may not work for you).
How should I use ALPROLIX?
ALPROLIX should be administered as ordered by your healthcare provider. You should be trained on how to do infusions by your healthcare provider. Many people with hemophilia B learn to infuse their ALPROLIX by themselves or with the help of a family member.
See the Instructions for Use for directions on infusing ALPROLIX. The steps in the Instructions for Use are general guidelines for using ALPROLIX. Always follow any specific instructions from your healthcare provider. If you are unsure of the procedure, please ask your healthcare provider. Do not use ALPROLIX as a continuous intravenous infusion.
Contact your healthcare provider immediately if bleeding is not controlled after using ALPROLIX.
What are the possible side effects of ALPROLIX?
Common side effects of ALPROLIX include headache, abnormal sensation in the mouth, and pain in your side with blood in your urine, which may be a sign of clot formation in the urinary collecting system.
Allergic reactions may occur. Call your healthcare provider or get emergency treatment right away if you have any of the following symptoms: hives, chest tightness, wheezing, difficulty breathing, or swelling of the face.
Redness to the skin at the injection site may also occur.
ALPROLIX may increase the risk of forming abnormal blood clots in your body, especially if you have risk factors for developing blood clots. Call your healthcare provider or seek emergency care if you have symptoms of a possible abnormal blood clot, which may include: chest pain, difficulty breathing, unexpected swelling of an arm or leg with or without pain or tenderness.
Your body can also make antibodies called "inhibitors" against ALPROLIX, which may stop ALPROLIX from working properly. Your healthcare provider may need to test your blood for inhibitors from time to time.
These are not all the possible side effects of ALPROLIX.
Talk to your healthcare provider about any side effect that bothers you or that does not go away.
How should I store ALPROLIX?
Store ALPROLIX vials at 2°C to 8°C (36°F to 46°F). Do not freeze.
ALPROLIX vials may also be stored at room temperature up to 30°C (86°F) for a single 6-month period.
If you choose to store ALPROLIX at room temperature:
- Note on the carton the date on which the product was removed from refrigeration.
- Use the product before the end of this 6-month period or discard it.
- Do not return the product to the refrigerator.
Do not use product or diluent after the expiration date printed on the carton, vial, or syringe.
After Reconstitution:
- Use the reconstituted product as soon as possible; however, you may store the reconstituted product at room temperature up to 30°C (86°F) for up to 3 hours. Protect the reconstituted product from direct sunlight. Discard any product not used within 3 hours after reconstitution.
- Do not use ALPROLIX if the reconstituted solution is cloudy, contains particles or is not colorless.
What else should I know about ALPROLIX?
Medicines are sometimes prescribed for purposes other than those listed here. Do not use ALPROLIX for a condition for which it was not prescribed. Do not share ALPROLIX with other people, even if they have the same symptoms that you have.
This Patient Information has been approved by the US Food and Drug Administration.
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
A SANOFI COMPANY
US License Number 2078
©2023 Bioverativ Therapeutics Inc. All rights reserved.
Revised: May 2023
ALPROLIX®
Coagulation Factor IX (Recombinant), Fc Fusion Protein
INSTRUCTIONS FOR USE
Read the Instructions for Use before you start using ALPROLIX® and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Your healthcare provider should show you or your caregiver how to reconstitute and administer ALPROLIX the first time ALPROLIX is used.
Check the expiration date on the ALPROLIX kit.
Do not use the product if past the expiration date.
Allow the ALPROLIX vial and the diluent to come to room temperature.
Do not use external heat sources such as putting the vial and/or diluent in hot water.
Find a clean, flat work surface and collect all the supplies you will need to reconstitute and administer ALPROLIX.
Wash your hands with soap and water. Aseptic technique (clean and germ free) should be used.
YOUR KIT CONTAINS:
RECONSTITUTION
TURN OVER FOR POOLING AND ADMINISTRATION
TURN OVER TO FRONT SIDE FOR RECONSTITUTION
POOLING
POOLING is the process of combining two or more reconstituted vials into a larger syringe (not into the diluent syringe) prior to intravenous administration.
If you are using two or more vials, follow these pooling steps.
Be sure to leave the vial adapter attached to the vial as you will need it for attaching a large luer lock syringe.
Do not detach the diluent syringe or the large luer syringe until you are ready to attach the large luer lock syringe to the next vial (with vial adapter attached).
ADMINISTRATION (Intravenous Injection)
ALPROLIX is administered by intravenous infusion after reconstitution of the drug powder with the diluent.
Your healthcare provider should teach you how to infuse ALPROLIX. Once you have been taught to self-infuse, you can follow these instructions.
Do not administer reconstituted ALPROLIX if it contains particulate matter, is discolored, or is cloudy.
STORAGE CONDITIONS - PRODUCT KIT
Keep refrigerated until use.
Keep away from direct sunlight.
STORAGE CONDITIONS - RECONSTITUTED
ALPROLIX should be administered within 3 hours after reconstitution.
Do not refrigerate after reconstitution.
Keep away from direct sunlight.
This Instructions for Use has been approved by the US Food and Drug Administration.
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
A SANOFI COMPANY
US License Number 2078
©2023 Bioverativ Therapeutics Inc. All rights reserved.
For more information go to www.ALPROLIX.com or call 1-855-692-5776
Revised: May 2023
PRINCIPAL DISPLAY PANEL - Kit Carton - 500 IU
500 IU Nominal
NDC 71104-911-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
PRINCIPAL DISPLAY PANEL - Kit Carton - 1000 IU
1000 IU Nominal
NDC 71104-922-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
PRINCIPAL DISPLAY PANEL - Kit Carton - 2000 IU
2000 IU Nominal
NDC 71104-933-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
PRINCIPAL DISPLAY PANEL - Kit Carton - 3000 IU
3000 IU Nominal
NDC 71104-944-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
PRINCIPAL DISPLAY PANEL - Kit Carton - 250 IU
250 IU Nominal
NDC 71104-966-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
PRINCIPAL DISPLAY PANEL - Kit Carton - 4000 IU
4000 IU Nominal
NDC 71104-977-01
One single-use vial
with 5 mL prefilled
diluent syringe
ALPROLIX®
Coagulation Factor IX
(Recombinant), Fc Fusion Protein
For Intravenous Administration
Rx Only
Manufactured by:
Bioverativ Therapeutics Inc.
Waltham, MA 02451
US License Number 2078
www.ALPROLIX.com
Bioverativ®
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| ALPROLIX
coagulation factor ix (recombinant), fc fusion protein kit |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Bioverativ Therapeutics Inc. (070517011) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Vetter Pharma Fertigung GmbH & Co. KG (Langenargen Eisenbahnstrasse) | 344217323 | ANALYSIS(71104-045, 71104-911, 71104-922, 71104-933, 71104-944, 71104-951, 71104-952, 71104-953, 71104-954, 71104-955, 71104-956, 71104-966, 71104-977) , MANUFACTURE(71104-045, 71104-911, 71104-922, 71104-933, 71104-944, 71104-951, 71104-952, 71104-953, 71104-954, 71104-955, 71104-956, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Vetter Pharma Fertigung GmbH & Co. KG (Ravensburg Schuetzenstrasse) | 316126754 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , MANUFACTURE(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Eurofins Biopharma Product Testing Munich GmbH | 313046917 | ANALYSIS(71104-045, 71104-911, 71104-922, 71104-933, 71104-944, 71104-951, 71104-952, 71104-953, 71104-954, 71104-955, 71104-956, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Vetter Pharma Fertigung GmbH & Co. KG (Ravensburg Mooswiesen) | 312670654 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , PACK(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , LABEL(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Packaging Coordinators, LLC | 078525133 | PACK(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , LABEL(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Lonza Biologics, Inc. | 093149750 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , API MANUFACTURE(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| PPD Development Ireland Ltd. | 985036175 | ANALYSIS(71104-045, 71104-911, 71104-922, 71104-933, 71104-944, 71104-951, 71104-952, 71104-953, 71104-954, 71104-955, 71104-956, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Avista Pharma Solutions, Inc. | 079509111 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Eurofins Biopharma Product Testing Ireland Limited | 238239933 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Rechon Life Science AB | 775207769 | PACK(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , LABEL(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Biogen U.S. Corporation | 078734950 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , MANUFACTURE(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sharp Packaging Services, LLC | 143696495 | PACK(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , LABEL(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Biogen MA Inc. | 841087823 | ANALYSIS(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) , API MANUFACTURE(71104-911, 71104-922, 71104-933, 71104-944, 71104-966, 71104-977) | |
More about Alprolix (coagulation factor ix)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Drug images
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: miscellaneous coagulation modifiers
- Breastfeeding
- En español
Patient resources
- Alprolix drug information
- Alprolix (Factor ix fc fusion protein recombinant Intravenous) (Advanced Reading)
Professional resources
Other brands
Idelvion, BeneFix, Rebinyn, Alphanine SD, ... +3 more