PLIVA 483 Pill: white, round, 10mm
The pill with imprint PLIVA 483 (White, Round, 10mm) has been identified as Theophylline Extended-Release 100 mg and is used for Asthma, acute, Asthma, Maintenance, Apnea of Prematurity, and Asthma. It belongs to the drug class methylxanthines and is not a controlled substance.
Images for PLIVA 483
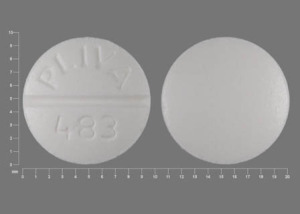



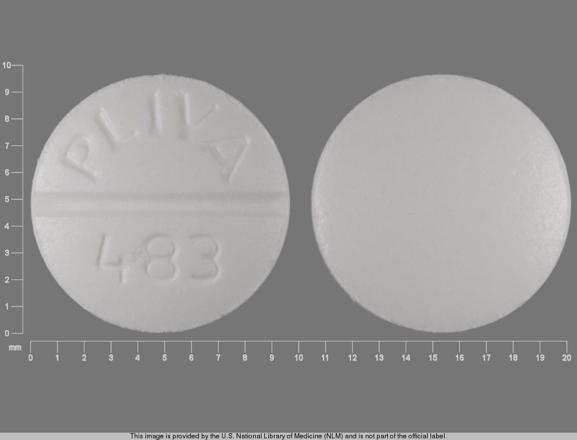
Theophylline Extended-Release
- Imprint
- PLIVA 483
- Strength
- 100 mg
- Color
- White
- Size
- 10.00 mm
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Methylxanthines
- Pregnancy Category
- C - Risk cannot be ruled out
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Pliva Inc.
- National Drug Code (NDC)
- 50111-0483 (Discontinued)
Related images for "PLIVA 483"
More about theophylline
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (19)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: methylxanthines
- Breastfeeding
- En español
Patient resources
- Theophylline drug information
- Theophylline (Intravenous) (Advanced Reading)
- Theophylline (Oral) (Advanced Reading)
Other brands
Theo-Dur, Theo-24, Elixophyllin, Quibron-T/SR, ... +6 more
Professional resources
- Theophylline prescribing information
- Theophylline Elixir (FDA)
- Theophylline Oral Solution (FDA)
- Theophylline in Dextrose (FDA)
Other brands
Theo-24, Elixophyllin, Quibron-T/SR
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.

