006 COP Pill: pink, round
Generic Name: fluoride
The pill with imprint 006 COP (Pink, Round, 0mm) has been identified as Luride 1 mg and is used for Prevention of Dental Caries. It belongs to the drug class minerals and electrolytes and is not a controlled substance.
Images for 006 COP

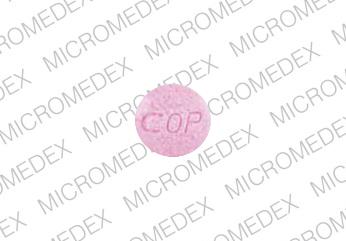
Luride
- Generic Name
- fluoride
- Imprint
- 006 COP
- Strength
- 1 mg
- Color
- Pink
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Minerals and electrolytes
- Pregnancy Category
- N - Not classified
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Colgate Oral Pharmaceuticals Inc
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 00126-0006 (Discontinued) | Colgate Oral Pharmaceuticals |
| 49999-0560 | Lake Erie Medical and Surgical Supply (repackager) |
See also:
More about Luride (fluoride)
Patient resources
Other brands
Altaflor, Fluor-A-Day, Fluorabon, Karidium
Professional resources
Other brands
Fluor-A-Day, Fluorabon Drops, Flura-Drops
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
