Fluad: Package Insert / Prescribing Info
Package insert / product label
Generic name: influenza vaccine, adjuvanted
Dosage form: injection, suspension
Drug class: Viral vaccines
Medically reviewed by Drugs.com. Last updated on Jul 10, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- References
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
FLUAD (Influenza Vaccine, Adjuvanted)
Injectable Emulsion for Intramuscular Use
2025-2026 Formula
Initial U.S. Approval: 2015
Indications and Usage for Fluad
FLUAD is a vaccine indicated for active immunization for the prevention of influenza disease caused by influenza virus subtypes A and type B contained in the vaccine. FLUAD is approved for use in adults 65 years of age and older. (1)
This indication is approved under accelerated approval based on the immune response elicited by FLUAD (1). Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
Fluad Dosage and Administration
For intramuscular use
A single 0.5 mL dose for intramuscular injection. (2.1)
Dosage Forms and Strengths
FLUAD is an injectable emulsion. A single dose is 0.5 mL. (3)
Contraindications
Warnings and Precautions
If Guillain-Barré Syndrome (GBS) has occurred within six weeks of previous influenza vaccination, the decision to give FLUAD should be based on careful consideration of the potential benefits and risks. (5.1)
Adverse Reactions/Side Effects
The most common (≥10%) local and systemic adverse reactions in adults 65 years of age and older who received FLUAD were injection site pain (25%), injection site tenderness (21%), myalgia (15%), fatigue (13%) and headache (13%). (6)
To report SUSPECTED ADVERSE REACTIONS, contact Seqirus at 1-855-358-8966 or VAERS at 1-800-822-7967 and www.vaers.hhs.gov.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2025
Full Prescribing Information
1. Indications and Usage for Fluad
FLUAD is a vaccine indicated for active immunization for the prevention of influenza disease caused by influenza virus subtypes A and type B contained in the vaccine. FLUAD is approved for use in adults 65 years of age and older.
This indication is approved under accelerated approval based on the immune response elicited by FLUAD [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
2. Fluad Dosage and Administration
For intramuscular use
2.1 Dosage and Schedule
Administer FLUAD as a single 0.5 mL intramuscular injection in adults 65 years of age and older.
2.2 Administration
- Gently shake each syringe. FLUAD has a milky-white appearance. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit [see Description (11)]. If either condition exists, FLUAD should not be administered.
- To use a pre-filled syringe fitted with a Luer Lok system, remove the tip cap by unscrewing it in a counter-clockwise direction. Once the tip cap is removed, attach a needle to the syringe by screwing it on in a clockwise direction until it locks. Once the needle is locked in place, remove the needle protector and administer the vaccine.
4. Contraindications
Do not administer FLUAD to anyone with a history of a severe allergic reaction (e.g., anaphylaxis) to any component of the vaccine, including egg protein [see Description (11)], or to a previous influenza vaccine.
5. Warnings and Precautions
5.1 Guillain-Barré Syndrome
If Guillain-Barré syndrome (GBS) has occurred within 6 weeks of receipt of prior influenza vaccine, the decision to give FLUAD should be based on careful consideration of the potential benefits and risks.
The 1976 swine influenza vaccine was associated with an elevated risk of GBS. [see References (1)] Evidence for a causal relationship of GBS with other influenza vaccines is inconclusive; if an excess risk exists, it is probably slightly more than 1 additional case per 1 million persons vaccinated.
5.2 Preventing and Managing Allergic Reactions
Appropriate medical treatment must be immediately available to manage potential anaphylactic reactions following administration of FLUAD.
5.3 Altered Immunocompetence
The immune response to FLUAD in immunocompromised persons, including individuals receiving immunosuppressive therapy, may be lower than in immunocompetent individuals.
6. Adverse Reactions/Side Effects
The most common (≥10%) local and systemic adverse reactions in adults 65 years of age and older who received FLUAD were injection site pain (25%), injection site tenderness (21%), myalgia (15%), fatigue (13%) and headache (13%).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect rates observed in clinical practice.
The safety of FLUAD and FLUAD QUADRIVALENT was evaluated in 17 clinical studies in 10,911 adults 65 years of age and older. Data for FLUAD QUADRIVALENT are relevant to FLUAD because both vaccines are manufactured using the same process and have overlapping compositions.
Study 1 (NCT01162122) was a multicenter, observer-blind, randomized controlled study conducted in the United States, Colombia, Panama and the Philippines during the 2010-2011 Northern Hemisphere influenza season. The safety analysis set included 3545 FLUAD recipients and 3537 AGRIFLU (Influenza Vaccine) recipients. The enrolled subject population was 65 to 97 years of age (mean 72 years) and 64% were female. Within each treatment group, 53% were Asian, 28% were Caucasian, 18% were Hispanic, 1% were Black, and fewer than 1% each were Native American/Alaskan, Pacific Islander/Hawaiian, or Other.
Solicited local (injection site) and systemic adverse reactions were collected from subjects who completed a symptom diary card for seven days following vaccination. The reported frequencies of solicited local adverse reactions are presented in Table 1a and systemic adverse reactions are presented in Table 1b.
|
a N = number of subjects with safety data. |
||
|
b Moderate: pain, tenderness, defined as “some limitation in normal daily activity” |
||
|
c Severe: pain, tenderness, defined as “unable to perform normal daily activity” |
||
| Solicited Local Adverse Reactions | FLUAD (Na=3418-3496)
Percentage | AGRIFLU (Na=3420-3488)
Percentage |
| Injection site Pain: Any | 25.0 | 12.2 |
| Injection site Pain: Moderateb | 3.9 | 1.9 |
| Injection site Pain: Severec | 0.3 | 0.2 |
| Tenderness: Any | 21.1 | 11.2 |
| Tenderness: Moderate | 3.0 | 1.0 |
| Tenderness: Severe | 0.1 | 0.2 |
| Erythema: Any | 1.2 | 0.5 |
| Erythema: 25 to ≤ 50 mm | 1.1 | 0.5 |
| Erythema: 51 to ≤ 100 mm | 0.2 | <0.1 |
| Erythema: > 100 mm | 0.0 | 0.0 |
| Induration: Any | 1.3 | 0.5 |
| Induration: 25 to ≤ 50 mm | 1.0 | 0.5 |
| Induration: 51 to ≤ 100 mm | 0.3 | 0.0 |
| Induration: > 100 mm | 0.0 | 0.0 |
| Swelling: Any | 1.2 | 0.4 |
| Swelling: 25 to ≤ 50 mm | 1.0 | 0.4 |
| Swelling: 51 to ≤ 100 mm | 0.2 | <0.1 |
| Swelling: > 100 mm | <0.1 | 0.0 |
|
a N = number of subjects with safety data. |
||
|
b Moderate: myalgia, fatigue, headache, arthralgia, chills, nausea, vomiting defined as “some limitation in normal daily activity”, diarrhea defined as “4 to 5 stools a day”. |
||
|
c Severe: myalgia, fatigue, headache, arthralgia, chills, nausea, vomiting defined as “unable to perform normal daily activity”, diarrhea defined as “6 or more watery stools a day”. |
||
|
d Potentially life threatening (PLT) reaction defined as requiring emergency room visit or hospitalization. |
||
| Solicited Systemic Adverse Reactions | FLUAD (Na=3418-3496) Percentage | AGRIFLU (Na=3420-3488)
Percentage |
| Myalgia: Any | 14.7 | 9.7 |
| Myalgia: Moderateb | 2.6 | 1.8 |
| Myalgia: Severec | 0.3 | 0.7 |
| Fatigue: Any | 13.3 | 10.4 |
| Fatigue: Moderate | 3.1 | 2.4 |
| Fatigue: Severe | 0.4 | 0.6 |
| Fatigue: PLTd | 0.0 | <0.1 |
| Headache: Any | 13.2 | 11.2 |
| Headache: Moderate | 3.0 | 2.6 |
| Headache: Severe | 0.4 | 0.6 |
| Headache: PLT | 0.0 | <0.1 |
| Arthralgia: Any | 8.5 | 7.8 |
| Arthralgia: Moderate | 1.6 | 1.6 |
| Arthralgia: Severe | 0.2 | 0.6 |
| Chills: Any | 6.7 | 4.7 |
| Chills: Moderate | 1.5 | 1.2 |
| Chills: Severe | 0.3 | 0.3 |
| Chills: PLT | <0.1 | 0.0 |
| Diarrhea: Any | 4.8 | 4.5 |
| Diarrhea: Moderate | 1.3 | 0.9 |
| Diarrhea: Severe | 0.3 | 0.2 |
| Diarrhea: PLT | <0.1 | <0.1 |
| Fever: Any | 3.6 | 3.4 |
| Fever: ≥38.0°C to ≤ 38.4°C | 1.8 | 1.7 |
| Fever: ≥ 38.5°C to ≤ 38.9°C | 1.3 | 1.3 |
| Fever: 39.0°C to ≤ 40.0°C | 0.4 | 0.4 |
| Fever: ≥ 40.0°C | 0.1 | 0.0 |
| Nausea: Any | 2.9 | 2.8 |
| Nausea: Moderate | 0.4 | 0.6 |
| Nausea: Severe | 0.1 | 0.1 |
| Nausea: PLT | <0.1 | 0.0 |
| Vomiting: Any | 1.4 | 1.7 |
| Vomiting: Moderate | 0.4 | 0.5 |
| Vomiting: Severe | <0.1 | 0.1 |
| Vomiting: PLT | <0.1 | 0.0 |
Unsolicited Adverse Events (AEs): The clinical safety of FLUAD was assessed in fifteen (15) randomized, controlled studies. The total safety population in these trials included 10,952 adults 65 years of age and older, comprising 5,754 who received FLUAD and 5,198 who received other US licensed influenza vaccines. The percentage of subjects with an unsolicited AE within 30 days following vaccination was similar between vaccine groups (16.9% FLUAD vs. 18.0% active comparator).
Serious Adverse Events (SAEs) and Deaths: In Study 1, in which subjects were followed for SAEs and deaths for one year following vaccination (N=3,545 FLUAD, N=3,537 AGRIFLU), the percentages of subjects with an SAE were similar between vaccine groups (7% FLUAD vs. 7% AGRIFLU). Four SAEs (1 FLUAD and 3 AGRIFLU) were assessed as related to study vaccination over one year of observation and 2 of these occurred (1 FLUAD and 1 AGRIFLU) within 21 days following study vaccination. There were 98 deaths (N=52 FLUAD, N=46 AGRIFLU) over one year of which none occurred within the first 21 days following vaccination.
In 14 additional randomized, controlled studies, SAEs were collected over a 3 to 4-week period in 4 studies, over a 8-week period in 1 study, and over a 6-month period in 9 studies (N= 2,209 FLUAD, N=1,661 US licensed influenza vaccines). The percentages of subjects with an SAE within 30 days (1.1% FLUAD vs. 1.8% AGRIFLU) or within 6 months (4.3% FLUAD vs. 5.9% AGRIFLU) were similar between vaccine groups. The percentages of deaths within 30 days (0.3% FLUAD vs. 0.6% active comparator) or within 6 months (1.0% FLUAD vs. 1.5% active comparator) were also similar.
Adverse Events of Special Interest (AESIs): Rates of new onset neuroinflammatory and immune mediated diseases were assessed in a post hoc analysis of the 15 randomized controlled studies over the time periods specified above for SAEs. The percentage of subjects with an AESI at any time after vaccination was similar between vaccine groups (0.9% FLUAD vs. 0.9% active comparator). There were no notable imbalances for specific AESIs.
Safety of Annual Revaccination: In 5 of the randomized, controlled trials, subjects were followed for SAEs and deaths for 6 months following revaccination (N=492 FLUAD, N=330 US licensed and non-US licensed influenza vaccines). After the second annual vaccination, the percentages of subjects with an SAE were similar between vaccine groups (6.1% FLUAD vs.
5.5% comparator influenza vaccines); 23 deaths (N=17 FLUAD, N=6 comparator influenza vaccines) were reported. Causes of death included cardiovascular events, malignancy, trauma, gastrointestinal disorders, and respiratory failure. Clinical characteristics of the deaths, including the variable causes, timing since vaccination, and underlying medical conditions, do not provide evidence for a causal relationship with FLUAD.
Safety of FLUAD QUADRIVALENT
Study 2 (NCT02587221) was a multi-center, randomized, observer-blind, non-influenza comparator-controlled efficacy and safety study conducted in 12 countries during the 2016-2017 Northern Hemisphere and 2017 Southern Hemisphere seasons. In this study, 3381 subjects received FLUAD QUADRIVALENT and 3380 subjects received a US-licensed non-influenza comparator vaccine (Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine, Boostrix® [GlaxoSmithKline Biologicals]).
Serious adverse events (SAEs) and potentially immune-mediated adverse events of special interest (AESIs) were collected up to 366 days after vaccination. SAEs were reported by 238 (7.0%) FLUAD QUADRIVALENT recipients and 234 (6.9%) comparator recipients. There were no SAEs, AESIs or deaths in this study that were related to FLUAD QUADRIVALENT.
Study 3 (NCT03314662) was a multicenter, randomized, double-blind, comparator-controlled study conducted during the 2017-18 Northern Hemisphere influenza season. In this study, 888 subjects received FLUAD QUADRIVALENT, 444 subjects received FLUAD (aTIV-1) and 444 subjects received FLUAD (aTIV-2). FLUAD (aTIV-1) contained hemagglutinin from a Type B virus of the Victoria lineage, and FLUAD (aTIV-2) contained hemagglutinin from a Type B virus of the Yamagata lineage.
Serious AEs and AESIs were collected up to 181 days after vaccination. Within 6 months after vaccination, 37 (4.2%) FLUAD QUADRIVALENT recipients, 28 (6.3%) FLUAD (aTIV-1) and 18 (4.1%) FLUAD (aTIV-2) recipients experienced an SAE. There were no SAEs, AESIs or deaths in this study that were related to the study vaccine. There were no AEs leading to withdrawal from the study.
6.2 Postmarketing Experience
In addition to the adverse reactions observed during clinical trials, the following adverse events were reported from postmarketing surveillance in individuals 65 years of age and older for FLUAD or FLUAD QUADRIVALENT. Data for FLUAD QUADRIVALENT are relevant to FLUAD because both vaccines are manufactured using the same process and have overlapping compositions. Because these events were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or to establish, for all events, a causal relationship to vaccine exposure.
Blood and lymphatic system disorders:
Thrombocytopenia (some cases were severe with platelet counts less than 5,000 per mm3), lymphadenopathy
Immune system disorders:
Allergic reactions including anaphylactic shock (in rare cases), anaphylaxis
Nervous system disorders:
Encephalomyelitis, Guillain-Barré Syndrome, convulsions, neuritis, neuralgia, paresthesia, syncope, presyncope, dizziness
Vascular disorders:
Vasculitis which may be associated with transient renal involvement
Skin and subcutaneous tissue disorders:
Generalized skin reactions including erythema multiforme, urticaria, pruritis or non-specific rash, erythema, angioedema
Musculoskeletal and connective tissue disorders:
Muscular weakness, pain in extremity
General disorders and administration site conditions:
Extensive swelling of injected limb, injection site cellulitis-like reaction, injection site swelling, peripheral swelling, asthenia, malaise, pyrexia
Related/similar drugs
8. Use In Specific Populations
Data for FLUAD QUADRIVALENT are relevant to FLUAD because both vaccines are manufactured using the same process and have overlapping compositions.
8.1 Pregnancy
Risk Summary
FLUAD is not approved for use in persons < 65 years of age.
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. The data are insufficient to establish if there is vaccine-associated risk with FLUAD in pregnant women.
A developmental toxicity study was performed in female rabbits administered FLUAD prior to mating and during gestation. A 0.5 mL dose was injected on each occasion (a single human dose is 0.5 mL). Animals were administered FLUAD by intramuscular injection twice prior to gestation, during the period of organogenesis (gestation day 7) and later in pregnancy (gestation day 20), 0.5 mL (45 mcg)/rabbit/occasion. No vaccine-related fetal malformations or variations and no adverse effects on pre-weaning development were observed in the study. FLUAD did not affect female fertility in the rabbit developmental toxicity study.
8.2 Lactation
Risk Summary
FLUAD is not approved for use in persons < 65 years of age. It is not known whether FLUAD is excreted in human milk. Data are not available to assess the effects of FLUAD on the breastfed infant or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for FLUAD and any potential adverse effects on the breastfed child from FLUAD or from the underlying maternal condition. For preventive vaccines, the underlying maternal condition is susceptibility to disease prevented by the vaccine.
8.3 Pediatric Use
Safety and effectiveness of FLUAD and FLUAD QUADRIVALENT were evaluated in clinical trials conducted in children 6 months to <72 months of age. Data for FLUAD QUADRIVALENT are relevant to FLUAD because both vaccines are manufactured using the same process and have overlapping compositions. Data from these trials are inconclusive to demonstrate the safety and effectiveness of FLUAD in children 6 months to <72 months of age. The safety and effectiveness of FLUAD in infants less than 6 months of age and in children older than 72 months of age have not been evaluated.
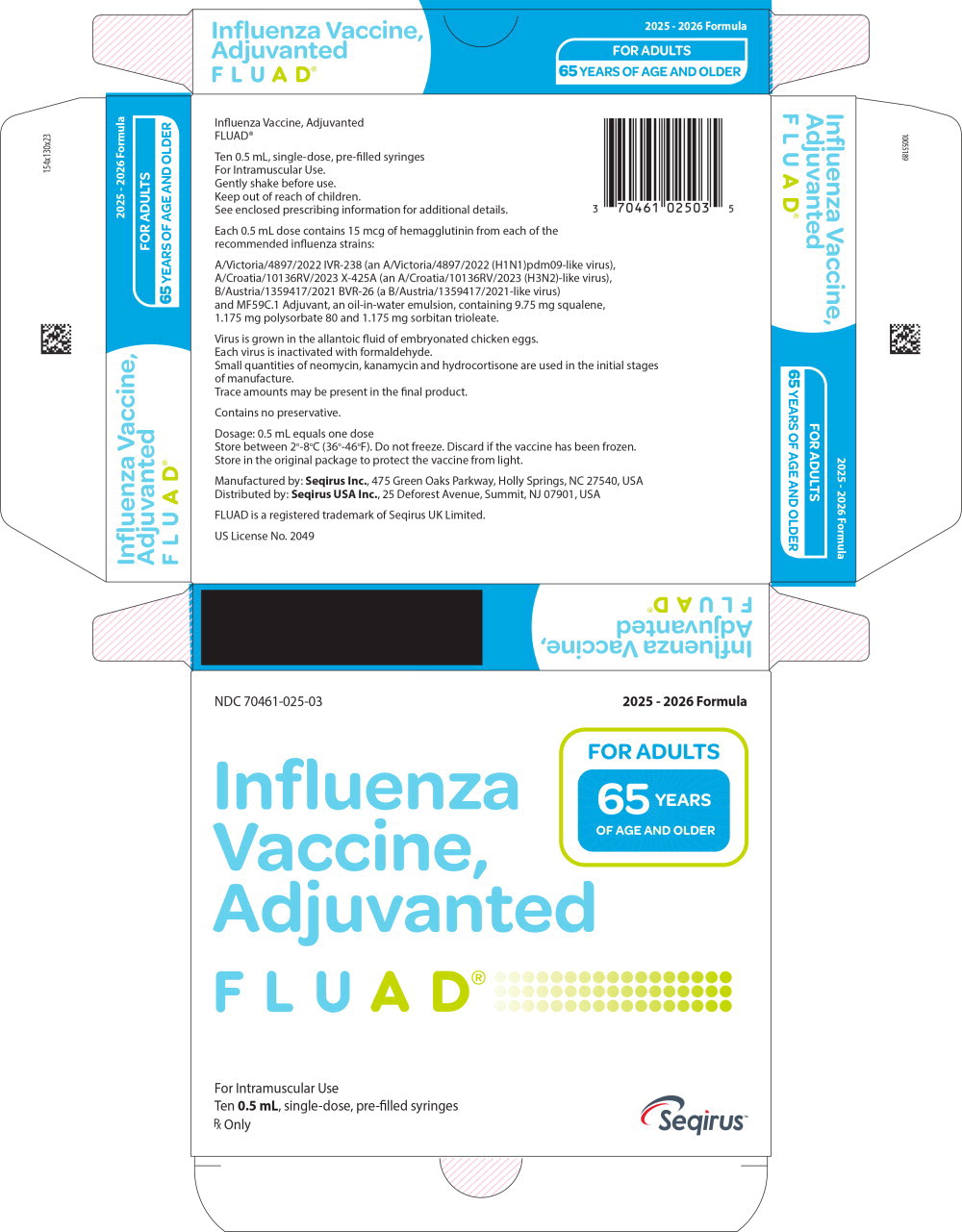
11. Fluad Description
FLUAD (Influenza Vaccine, Adjuvanted), a sterile injectable emulsion for intramuscular use, is a trivalent inactivated influenza vaccine prepared from virus propagated in the allantoic cavity of embryonated hens' eggs inoculated with a specific type of influenza virus.
FLUAD is standardized according to United States Public Health Service requirements and each 0.5 mL dose is formulated to contain 15 mcg of hemagglutinin (HA) from each of the following influenza strains recommended for the 2025-2026 influenza season: A/Victoria/4897/2022 IVR-238 (an A/Victoria/4897/2022 (H1N1)pdm09-like virus), A/Croatia/10136RV/2023 X-425A (an A/Croatia/10136RV/2023 (H3N2)-like virus), B/Austria/1359417/2021 BVR-26 (a B/Austria/1359417/2021-like virus). FLUAD also contains MF59C.1 adjuvant (MF59®), a squalene based oil-in-water emulsion. Each of the strains is harvested and clarified separately by centrifugation and filtration prior to inactivation with formaldehyde. The inactivated virus is concentrated and purified by zonal centrifugation. The surface antigens, hemagglutinin and neuraminidase, are obtained from the influenza virus particle by further centrifugation in the presence of cetyltrimethylammonium bromide (CTAB). The antigen preparation is further purified.
FLUAD is prepared by combining the three virus antigens with the MF59C.1 adjuvant. After combining, FLUAD is a sterile, milky-white injectable emulsion supplied in single-dose pre-filled syringes containing 0.5 mL dose. Each 0.5 mL dose contains 15 mcg of hemagglutinin (HA) from each of the three recommended influenza strains and MF59C.1 adjuvant (9.75 mg squalene, 1.175 mg of polysorbate 80, 1.175 mg of sorbitan trioleate, 0.66 mg of sodium citrate dihydrate and 0.04 mg of citric acid monohydrate) at pH 6.9-7.7.
FLUAD may contain trace amounts of neomycin (≤ 0.02 mcg by calculation), kanamycin (≤ 0.03 mcg by calculation) and hydrocortisone (≤ 0.005 ng by calculation) which are used during the initial stages of manufacture, as well as residual egg protein (ovalbumin) (≤ 0.4 mcg), formaldehyde (≤ 10 mcg) or CTAB (≤ 12 mcg).
FLUAD does not contain a preservative. The syringe, syringe plunger stopper and tip caps are not made with natural rubber latex.
12. Fluad - Clinical Pharmacology
Mechanism of Action
Specific levels of hemagglutination inhibition (HI) antibody titers induced by vaccination with inactivated influenza virus vaccine have not been correlated with protection from influenza illness. In some human studies, HI antibody titers of 1:40 or greater have been associated with protection from influenza illness in up to 50% of subjects. [see References (2,3)]
Antibody against one influenza virus type or subtype confers limited or no protection against another. Furthermore, antibody to one antigenic variant of influenza virus might not protect against a new antigenic variant of the same type or subtype. Frequent development of antigenic variants through antigenic drift is the virologic basis for seasonal epidemics and the reason for the usual change of one or more new strains in each year's influenza vaccine.
14. Clinical Studies
Study 1 (NCT01162122) evaluated the safety and immunogenicity of FLUAD in comparison to AGRIFLU. A total of 7082 subjects were randomized and vaccinated with FLUAD (N=3541) or AGRIFLU (N=3541). The primary immunogenicity analyses were conducted on all vaccinated subjects with a blood sample collected at Day 22 (N=3225-3227 [91%] and 3,256- 3,259 [92%] in the FLUAD and AGRIFLU groups, respectively). Non-inferiority of FLUAD compared with AGRIFLU was demonstrated for all three vaccine strains based on pre-defined thresholds for seroconversion rate differences and GMT ratios (Table 2a & 2b).
|
GMT = Geometric mean antibody titer; CI = Confidence Interval. |
|||
|
a Results obtained following vaccination with influenza vaccine formulated for the 2010-2011 season. |
|||
|
b N is the number of vaccinated participants with available data for the immunologic endpoint listed. |
|||
|
c FLUAD met non-inferiority criteria based on GMT ratios if the lower limit of the 95% CI [FLUAD:AGRIFLU] for each strain was > 0.67. |
|||
| GMTs Against
FLUAD and AGRIFLU Vaccine Strains | FLUAD
N b= 3225-3227 GMT (95% CI) | AGRIFLU
Nb =3256-3259 GMT (95% CI) | FLUAD and AGRIFLU
GMT Ratioc (95% CI) |
| A/California/7/2009-like (H1N1) | 99 (93-106) | 70 (66-75) | 1.4 (1.32-1.49) |
| A/Perth/16/2009-like (H3N2) | 272 (257-288) | 169 (159-179) | 1.61 (1.52-1.7) |
| B/Brisbane/60/2008- like | 28 (26-29) | 24 (23-26) | 1.15 (1.08-1.21) |
| Seroconversionc for Vaccine Strains: | FLUAD
Nb= 3225-3227 % of Subjects (95% CI) | AGRIFLU
Nb =3256-3259 % of Subjects (95% CI) | FLUAD and AGRIFLU
Difference in Seroconversion Rated (95% CI) |
|---|---|---|---|
|
a Results obtained following vaccination with influenza vaccine formulated for the 2010-2011 season. |
|||
|
b N is the number of vaccinated participants with available data for the immunologic endpoint listed. |
|||
|
c Seroconversion was defined as prevaccination HI titer <10 and postvaccination HI titer ≥ 40 or at least a 4-fold increase in HI from prevaccination HI titer ≥ 10. |
|||
|
d FLUAD met non-inferiority criteria based on seroconversion rate differences if the lower limit of the 95% CI |
|||
|
[FLUAD -AGRIFLU] for each strain was >-10% . |
|||
| A/California/7/2009- like (H1N1) | 69% (67%–70%) | 58% (57%–60%) | 9.8% (7.5%–12.1%) |
| A/Perth/16/2009-like (H3N2) | 73% (71%–74%) | 58% (56%–60%) | 13.9% (11.7%–16.1%) |
| B/Brisbane/60/2008- like | 33% (31%–35%) | 29% (28%–31%) | 3.2% (1.1%–5.3%) |
15. References
-
Lasky T, Terracciano GJ, Magder L, et al. The Guillain-Barre syndrome and the 1992-1993 and 1993-1994 influenza vaccines. N Engl J Med 1998; 339(25): 1797-1802.
-
Hannoun C, Megas F, Piercy J. Immunogenicity and protective efficacy of influenza vaccination. Virus Res 2004; 103:133-138.
-
Hobson D, Curry RL, Beare A, et. al. The role of serum hemagglutinin-inhibiting antibody in protection against challenge infection with influenza A2 and B viruses. J Hyg Camb 1972; 767-777.
16. How is Fluad supplied
FLUAD is supplied in the product presentation listed below:
| Presentation | Carton
NDC Number | Components |
| Pre-Filled Syringe | 70461-025-03 | 0.5 mL dose in a pre-filled syringe (needle not supplied), package of 10 syringes per carton [NDC 70461-025-04] |
Store FLUAD refrigerated at 2°C to 8°C (36°F to 46°F). Protect from light. Do not freeze. Discard if the vaccine has been frozen. Do not use after expiration date.
The syringe, syringe plunger stopper and tip cap are not made with natural rubber latex.
17. Patient Counseling Information
- Inform vaccine recipients of the potential benefits and risks of immunization with FLUAD.
- Educate vaccine recipients regarding the potential side effects. Clinicians should emphasize that (1) FLUAD contains non-infectious particles and cannot cause influenza and (2) FLUAD is intended to help provide protection against illness due to influenza viruses only.
- Instruct vaccine recipients to report adverse reactions to their healthcare provider and/or to Vaccine Adverse Event Reporting System (VAERS) at 1-800-822-7967 and www.vaers.hhs.gov. Provide vaccine recipients with the Vaccine Information Statements which are required by the National Childhood Vaccine Injury Act of 1986. These materials are available free of charge at the Centers for Disease Control and Prevention (CDC) website (www.cdc.gov/vaccines).
- Inform vaccine recipients that annual vaccination is recommended.
FLUAD, FLUAD QUADRIVALENT and MF59 are registered trademarks of Seqirus UK Limited or its affiliates.
Manufactured by: Seqirus Inc., 475 Green Oaks Parkway, Holly Springs, NC 27540, USA
Distributed by: Seqirus USA Inc., 25 Deforest Avenue, Summit, NJ 07901, USA
Tel: 1-855-358-8966
US License No. 2049
| FLUAD
influenza a virus a/victoria/4897/2022 ivr-238 (h1n1) antigen (formaldehyde inactivated), influenza a virus a/croatia/10136rv/2023 x-425a (h3n2) antigen (formaldehyde inactivated), influenza b virus b/austria/1359417/2021 bvr-26 antigen (formaldehyde inactivated) injection, suspension |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Seqirus, Inc. (080102141) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Seqirus Inc | 080102141 | MANUFACTURE, LABEL, ANALYSIS, PACK | |
Frequently asked questions
- How and where is a flu shot injection given?
- What flu vaccine can I use with an egg allergy?
- Where can I get the flu vaccine right now?
- How can I get a flu vaccine without a needle?
- Which flu vaccines are available this year?
- How well does the flu vaccine work?
More about Fluad (influenza virus vaccine, inactivated)
- Check interactions
- Compare alternatives
- Reviews (6)
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: viral vaccines
Patient resources
Professional resources
Other brands
Afluria, Fluzone High-Dose, Flucelvax, Fluarix, ... +10 more