The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Arthramid Vet
This page contains information on Arthramid Vet for veterinary use.The information provided typically includes the following:
- Arthramid Vet Indications
- Warnings and cautions for Arthramid Vet
- Direction and dosage information for Arthramid Vet
Arthramid Vet
This treatment applies to the following species: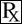 Company: Contura Vet US
Company: Contura Vet US
Instruction For Use
Arthramid Vet Caution
Federal law restricts this device to sale by or on the order of a licensed veterinarian and it must be used under the supervision of a licensed veterinarian for the application of intra-articular administration.
Product Description
Arthramid® Vet is a non-resorbable, injectable, transparent, hydrophilic gel, for intra-articular administration in horses only.
Arthramid® Vet consists of a backbone of cross-linked polyacrylamide, with water molecules loosely bound to the polymer matrix. Nominal proportions of the Arthramid® Vet gel are 2.5% cross-linked polyacrylamide and 97.5% non-pyrogenic water for injection. Arthramid® Vet is biocompatible and non-biodegradable and is sterilized by moist heat.
Arthramid® Vet is supplied in a pre-filled, sterile, 1 mL syringe sealed with a Luer lock fitting. It is intended to be injected with a sterile 16-23G hypodermic needle.
Indication
Arthramid® Vet is indicated for the management of non-infectious causes of joint disease in horses, including both early and late stages of osteoarthritis and degenerative joint disease.
Contraindications
Arthramid® Vet is not to be injected into actively infected joints, infected surrounding joint soft tissues or infected skin overlaying the joint.
Warnings
● Do not inject Arthramid® Vet intravascularly. Injection into blood vessels may cause vascular occlusion leading to possible embolism.
● Do not use Arthramid® Vet if the package is opened or damaged.
● Do not re-sterilize Arthramid® Vet.
● Do not mix Arthramid® Vet with any other product.
● Do not use Arthramid® Vet in combination with any other products intended for intra-articular injection, for a period of no less than 30 days.
● Do not use Arthramid® Vet after the expiraton date printed on the packaging.
Precautions
Arthramid® Vet is not recommended for use in horses with acute/chronic diseases receiving treatment with systemic corticosteroids or antibiotics.
The injection site should be disinfected prior to treatment.
Medical history records addressing ongoing infections, concomitant medication, surgery, etc. must be reviewed before injection to prevent possible infections.
After use, syringes and needles should be handled as potential biohazards. Dispose in accordance with accepted medical practice and applicable local, state and federal requirements.
Method of Administration
Arthramid® Vet must be administered by a qualified veterinarian trained in these types of procedures.
Arthramid® Vet is administered via percutaneous infiltration into the articular cavity of the affected joint delivering the required volume as advised depending on the joint being injected.
Dose Recommendation
The following dosage recommendations have been made, based on observed clinical responses to administration;
● Distal Interphalangeal (DIP/ Coffin) - 1-2 mL
● Metacarpo/tarso-phalangeal (Fetlock) - 2 mL
● Carpus - 2 mL
● Tarso-metatarsal (TMT)/ Distal-intertarsal (DIT) - 1 mL
● Tarsocrural - 2-3 mL
● Shoulder - 3 mL
● Stifles - 1-2 mL per compartment or 3-4 mL for medial-femorotibial joint
A single injection of the above advised volumes is considered adequate administration to achieve effect. Published clinical trials indicate horses that partially respond to an initial treatment may benefit from a second dose at 4 to 6 weeks later.
Studies indicate that concurrent treatment of multiple joints in the same animal is safe.
Peri-Operative Procedures
1. Sedate or restrain the horse.
2. Clip the hair overlaying the joint to be injected. For antisepsis, wash and disinfect thoroughly in a radius of 3 inches round the injection site. Injection of Arthramid® Vet may be performed under local anaesthesia.
Aseptic technique must be used to avoid contamination of the sterile Arthramid® Vet syringe and injection site. The 16-23G hypodermic needle should be placed intra-articularly. Synovial fluid should be observed. Care should be taken to avoid unnecessary damage of the intra-articular tissue, as this may result in diffuse swelling lasting for 24 to 48 hours. Remove the protective tip cap from the Arthramid® Vet syringe. The syringe should then be firmly attached into the luer lock socket on the hypodermic needle. Make certain that the syringe is correctly mounted prior to use. If more than 1 mL of Arthramid® Vet is required for the joint, detach the empty Arthramid® Vet syringe from the hypodermic needle and repeat the process until the required dose is delivered. Upon completion, withdraw the hypodermic needle and syringe and dispose in accordance with accepted medical practice and applicable local, state and federal requirements. Cover the injection site with a sterile non-adhesive dressing.
Post-Operative Procedures
An ointment can be applied on the injection site immediately after treatment. A cold pack can be applied on the injection site in case of an edema. A bandage should be applied around the injection site if possible. Local or systemic corticosteroids should not be administered to the animal within two weeks of injection of Arthramid® Vet, since this may mask a possible infection. The animal should be rested for 48 hours after the treatment.
Non-steroidal anti-inflammatory drugs (NSAIDs) can be administered for pain relief and to reduce swelling. Allergic reactions to Arthramid® Vet have never been observed.
Complications
As with any intra-articular procedure, an Arthramid® Vet injection carries a risk of infection. Standard precautions and strict aseptic injection technique are essential. In the event of infection, the use of broad-spectrum antibiotics is recommended as a first line treatment. Any use of corticosteroids is contraindicated in case of infection.
Some animals can develop pain within the first few hours, post-administration. In addition, there is also a slight risk of hematoma and mild edema at the site of injection. Within 1-2 weeks after treatment there is a slight risk that the animal may develop a transient edema and tenderness at the treatment site. If not caused by infection, these reactions are self-limiting and should resolve within a couple of weeks.
Information to the Owner
The owner of the horse should be informed about the indications, expected results, contraindications, precautions, warnings, and potential complications. The owner of the horse should be advised that in case of complications, the veterinarian who performed the Arthramid® Vet injections should be contacted immediately for necessary treatment.
Owners should be counselled that one or more repeat Arthramid® Vet injection procedures may be required to achieve a satisfactory level of improvement in disease symptoms.
Complaints
Please report any malfunction or complaints to Contura Vet at Info@conturavet.com.
Storage
Arthramid® Vet must be stored protected from direct sunlight. Do not freeze. Do not store unsealed syringes for later use.
Symbols used on packaging

Contura Vet Limited, Riverside One, Sir John Rogerson’s Quay, Dublin 2, Ireland
Manufactured by: Contura International A/S, Sydmarken 23, 2860 Soeborg, Denmark
10095-001 September 2020
CPN: 2112000.0
3401 MALLORY LANE, SUITE 100, FRANKLIN, TN, 37067
| Toll-Free: | 800-689-2909 | |
| Website: | www.conturavetus.com | |
| Email: | usorders@conturavet.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2024 Animalytix LLC. Updated: 2024-02-27
