Alprostadil Prescribing Information
Package insert / product label
Dosage form: injection, solution, concentrate
Drug classes: Impotence agents, Vasodilators
Medically reviewed by Drugs.com. Last updated on Jun 25, 2023.
On This Page
WARNING
Apnea is experienced by about 10 to 12% of neonates with congenital heart defects treated with alprostadil injection. Apnea is most often seen in neonates weighing less than 2 kg at birth and usually appears during the first hour of drug infusion. Therefore, respiratory status should be monitored throughout treatment, and alprostadil injection should be used where ventilatory assistance is immediately available.
Alprostadil Description
Alprostadil injection USP for intravascular infusion contains 500 micrograms alprostadil, more commonly known as prostaglandin E1, in 1 mL dehydrated alcohol.
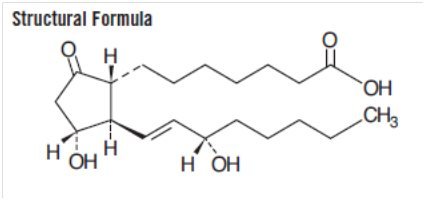
The chemical name for alprostadil is (1R,2R,3R)-3-Hydroxy-2-[(E)-(3S)-3-hydroxy-1-octenyl]-5-oxocyclopentane heptanoic acid, and the molecular weight is 354.49.
Alprostadil is a white to off-white crystalline powder with a melting point between 110° and 116°C. Its solubility at 35°C is 8000 micrograms per 100 mL double distilled water. Alprostadil has a molecular formula of C20H34O5.
Alprostadil - Clinical Pharmacology
Alprostadil (prostaglandin E1) is one of a family of naturally occurring acidic lipids with various pharmacologic effects. Vasodilation, inhibition of platelet aggregation, and stimulation of intestinal and uterine smooth muscle are among the most notable of these effects. Intravenous doses of 1 to 10 micrograms of alprostadil per kilogram of body weight lower the blood pressure in mammals by decreasing peripheral resistance. Reflex increases in cardiac output and rate accompany the reduction in blood pressure.
Smooth muscle of the ductus arteriosus is especially sensitive to alprostadil, and strips of lamb ductus markedly relax in the presence of the drug. In addition, administration of alprostadil reopened the closing ductus of new-born rats, rabbits, and lambs. These observations led to the investigation of alprostadil in infants who had congenital defects which restricted the pulmonary or systemic blood flow and who depended on a patent ductus arteriosus for adequate blood oxygenation and lower body perfusion.
In infants with restricted pulmonary blood flow, about 50% responded to alprostadil infusion with at least a 10 torr increase in blood pO2 (mean increase about 14 torr and mean increase in oxygen saturation about 23%). In general, patients who responded best had low pretreatment blood pO2 and were 4 days old or less.
In infants with restricted systemic blood flow, alprostadil often increased pH in those having acidosis, increased systemic blood pressure, and decreased the ratio of pulmonary artery pressure to aortic pressure.
Alprostadil must be infused continuously because it is very rapidly metabolized. As much as 80% of the circulating alprostadil may be metabolized in one pass through the lungs, primarily by β- and ω-oxidation. The metabolites are excreted primarily by the kidney, and excretion is essentially complete within 24 hours after administration. No unchanged alprostadil has been found in the urine, and there is no evidence of tissue retention of alprostadil or its metabolites.
Indications and Usage for Alprostadil
Alprostadil injection USP is indicated for palliative, not definitive, therapy to temporarily maintain the patency of the ductus arteriosus until corrective or palliative surgery can be performed in neonates who have congenital heart defects and who depend upon the patent ductus for survival. Such congenital heart defects include pulmonary atresia, pulmonary stenosis, tricuspid atresia, tetralogy of Fallot, interruption of the aortic arch, coarctation of the aorta, or transposition of the great vessels with or without other defects.
In infants with restricted pulmonary blood flow, the increase in blood oxygenation is inversely proportional to pretreatment pO2 values; that is, patients with low pO2 values respond best, and patients with pO2 values of 40 torr or more usually have little response.
Alprostadil injection USP should be administered only by trained personnel in facilities that provide pediatric intensive care.
Warnings
See WARNING box.
NOTE: Alprostadil injection must be diluted before it is administered. See dilution instructions in DOSAGE AND ADMINISTRATION section.
The administration of alprostadil injection to neonates may result in gastric outlet obstruction secondary to antral hyperplasia. This effect appears to be related to duration of therapy and cumulative dose of the drug. Neonates receiving alprostadil injection at recommended doses for more than 120 hours should be closely monitored for evidence of antral hyperplasia and gastric outlet obstruction.
Alprostadil injection should be infused for the shortest time and at the lowest dose that will produce the desired effects. The risks of long-term infusion of alprostadil injection should be weighed against the possible benefits that critically ill infants may derive from its administration.
Precautions
General Precautions
Cortical proliferation of the long bones, first observed in dogs, has also been observed in infants during long-term infusions of alprostadil. The cortical proliferation in infants regressed after withdrawal of the drug.
In infants treated with alprostadil injection at the usual doses for 10 hours to 12 days and who died of causes unrelated to ductus structural weakness, tissue sections of the ductus and pulmonary arteries have shown intimal lacerations, a decrease in medial muscularity and disruption of the medial and internal elastic lamina. Localized and aneurysmal dilatations and vessel wall edema also were seen compared to a series of pathological specimens from infants not treated with alprostadil injection. The incidence of such structural alterations has not been defined.
Because alprostadil inhibits platelet aggregation, use alprostadil injection cautiously in neonates with bleeding tendencies.
Alprostadil injection should not be used in neonates with respiratory distress syndrome. A differential diagnosis should be made between respiratory distress syndrome (hyaline membrane disease) and cyanotic heart disease (restricted pulmonary blood flow). If full diagnostic facilities are not immediately available, cyanosis (pO2 less than 40 torr) and restricted pulmonary blood flow apparent on an X-ray are appropriate indicators of congenital heart defects.
Necessary Monitoring
In all neonates, arterial pressure should be monitored intermittently by umbilical artery catheter, auscultation, or with a Doppler transducer. Should arterial pressure fall significantly, decrease the rate of infusion immediately.
In infants with restricted pulmonary blood flow, measure efficacy of alprostadil injection by monitoring improvement in blood oxygenation. In infants with restricted systemic blood flow, measure efficacy by monitoring improvement of systemic blood pressure and blood pH.
Drug Interactions
No drug interactions have been reported between alprostadil injection and the therapy standard in neonates with restricted pulmonary or systemic blood flow. Standard therapy includes antibiotics, such as penicillin and gentamicin; vasopressors, such as dopamine and isoproterenol; cardiac glycosides; and diuretics, such as furosemide.
Adverse Reactions/Side Effects
Central Nervous System
Apnea has been reported in about 12% of the neonates treated (see WARNING box). Other common adverse reactions reported have been fever in about 14% of the patients treated and seizures in about 4%. The following reactions have been reported in less than 1% of the patients: cerebral bleeding, hyperextension of the neck, hyperirritability, hypothermia, jitteriness, lethargy, and stiffness.
Cardiovascular System
The most common adverse reactions reported have been flushing in about 10% of patients (more common after intraarterial dosing), bradycardia in about 7%, hypotension in about 4%, tachycardia in about 3%, cardiac arrest in about 1%, and edema in about 1%. The following reactions have been reported in less than 1% of the patients: congestive heart failure, hyperemia, second degree heart block, shock, spasm of the right ventricle infundibulum, supraventricular tachycardia, and ventricular fibrillation.
Respiratory System
The following reactions have been reported in less than 1% of the patients: bradypnea, bronchial wheezing, hypercapnia, respiratory depression, respiratory distress, and tachypnea.
Gastrointestinal System
See WARNINGS.
The most common adverse reaction reported has been diarrhea in about 2% of the patients. The following reactions have been reported in less than 1% of the patients: gastric regurgitation, and hyperbilirubinemia.
Hematologic System
The most common hematologic event reported has been disseminated intravascular coagulation in about 1% of the patients. The following events have been reported in less than 1% of the patients: anemia, bleeding, and thrombocytopenia.
Miscellaneous
Sepsis has been reported in about 2% of the patients. Peritonitis has been reported in less than 1% of the patients. Hypokalemia has been reported in about 1%, and hypoglycemia and hyperkalemia have been reported in less than 1% of the patients.
To report SUSPECTED ADVERSE REACTIONS, contact Teva Pharmaceuticals USA, Inc. at 1-888-838-2872 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Overdosage
Apnea, bradycardia, pyrexia, hypotension, and flushing may be signs of drug overdosage. If apnea or bradycardia occurs, discontinue the infusion, and provide appropriate medical treatment. Caution should be used in restarting the infusion. If pyrexia or hypotension occurs, reduce the infusion rate until these symptoms subside. Flushing is usually a result of incorrect intraarterial catheter placement, and the catheter should be repositioned.
Alprostadil Dosage and Administration
The preferred route of administration for alprostadil injection is continuous intravenous infusion into a large vein. Alternatively, alprostadil injection may be administered through an umbilical artery catheter placed at the ductal opening. Increases in blood pO2 (torr) have been the same in neonates who received the drug by either route of administration.
Begin infusion with 0.05 to 0.1 micrograms alprostadil per kilogram of body weight per minute. A starting dose of 0.1 micrograms per kilogram of body weight per minute is the recommended starting dose based on clinical studies; however, adequate clinical response has been reported using a starting dose of 0.05 micrograms per kilogram of body weight per minute. After a therapeutic response is achieved (increased pO2 in infants with restricted pulmonary blood flow or increased systemic blood pressure and blood pH in infants with restricted systemic blood flow), reduce the infusion rate to provide the lowest possible dosage that maintains the response. This may be accomplished by reducing the dosage from 0.1 to 0.05 to 0.025 to 0.01 micrograms per kilogram of body weight per minute. If response to 0.05 micrograms per kilogram of body weight per minute is inadequate, dosage can be increased up to 0.4 micrograms per kilogram of body weight per minute although, in general, higher infusion rates do not produce greater effects.
Dilution Instructions
To prepare infusion solutions, dilute 1 mL of alprostadil injection with sodium chloride injection, USP or dextrose injection, USP. Undiluted alprostadil injection may interact with the plastic sidewalls of volumetric infusion chambers causing a change in the appearance of the chamber and creating a hazy solution. Should this occur, the solution and the volumetric infusion chamber should be replaced.
When using a volumetric infusion chamber, the appropriate amount of intravenous infusion solution should be added to the chamber first. The undiluted alprostadil injection should then be added to the intravenous infusion solution, avoiding direct contact of the undiluted solution with the walls of the volumetric infusion chamber.
Dilute to volumes appropriate for the pump delivery system available. Prepare fresh infusion solutions every 24 hours. Discard any solution more than 24 hours old.
|
Add 1 vial (500 micrograms) alprostadil to: | Approximate
Concentration of resulting solution (micrograms/mL) |
lnfusion rate (mL/min per kg) (of body weight) |
|---|---|---|
|
250 mL |
2 |
0.05 |
|
100 mL |
5 |
0.02 |
|
50 mL |
10 |
0.01 |
|
25 mL |
20 |
0.005 |
|
Example: |
To provide 0.1 micrograms/kilogram of body weight per minute to an infant weighing 2.8 kilograms using a solution of 1 vial alprostadil injection in 100 mL of saline or dextrose: INFUSION RATE = 0.02 mL/min per kg × 2.8 kg = 0.056 mL/min or 3.36 mL/hr. |
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
How is Alprostadil supplied
Each mL contains 500 micrograms alprostadil in dehydrated alcohol.
|
NDC Number |
Alprostadil per Microgram/mL |
Volume |
|---|---|---|
|
NDC 0703-1501-02 |
500 micrograms/mL |
1 mL Single Dose Vial |
Store alprostadil injection USP in a refrigerator at 2° to 8°C (36° to 46°F).
Distributed by:
Teva Pharmaceuticals USA, Inc.
Parsippany, NJ 07054
Rev. B 2/2020
| ALPROSTADIL
alprostadil injection, solution, concentrate |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Teva Parenteral Medicines, Inc. (794362533) |
More about alprostadil
- Check interactions
- Compare alternatives
- Reviews (182)
- Side effects
- Dosage information
- During pregnancy
- Drug class: impotence agents
- En español
Patient resources
Professional resources
Other brands
Edex, Caverject, Muse, Prostin VR Pediatric, Caverject Impulse

