Phoxillum Dosage
Generic name: Calcium Chloride 0.139g in 1L, Magnesium Chloride 0.071g in 1L, Sodium Chloride 6.023g in 1L, Potassium Chloride 0.298g in 1L, Sodium Phosphate, Dibasic 0.146g in 1L, Sodium Bicarbonate 2.936g in 1L
Dosage form: renal replacement solution
Medically reviewed by Drugs.com. Last updated on Feb 14, 2025.
Administration Instructions
Visually inspect PRISMASOL and PHOXILLUM for particulate matter and discoloration prior to administration.
Administration should only be under the direction of a physician competent in intensive care treatment including CRRT. Use only with extracorporeal dialysis equipment appropriate for CRRT.
The prepared solution is for single patient use only.
Aseptic technique should be used throughout administration to the patient.
Discard any unused solution.
Dosing Considerations
PRISMASOL replacement solutions contain 4 different combinations of active ingredients (8 different products with varying ingredient amounts). PHOXILLUM replacement solutions contain 2 different combinations of active ingredients (2 different products with varying ingredient amounts). PRISMASOL and PHOXILLUM are supplied in a two-compartment bag that must be mixed immediately prior to use:
- Small compartment A (250 mL) containing an electrolyte solution, and
- Large compartment B (4750 mL) containing the buffer solution.
See Table 1 for the concentrations of the active ingredients (after mixing) in these 10 different replacement solutions (total volume is 5 Liters).
| Ca2+ mEq/L |
HCO3- mEq/L |
K+ mEq/L |
Mg2+ mEq/L |
Na+ mEq/L |
HPO42- mmol/L |
Cl- mEq/L |
Lactate mEq/L |
Dextrose mg/dL |
Osmolarity mOsm/L |
|
|---|---|---|---|---|---|---|---|---|---|---|
| Ca2+ = calcium, HCO3- = bicarbonate, K+ = potassium, Mg2+ = magnesium, Na+ = sodium, HPO42- = phosphate, Cl- = chloride; osmolarity is estimated | ||||||||||
| PRISMASOL Replacement Solutions | ||||||||||
| BGK0/2.5 | 2.5 | 32 | 0 | 1.5 | 140 | 0 | 109.0 | 3.0 | 100 | 292 |
| BGK4/2.5 | 2.5 | 32 | 4.0 | 1.5 | 140 | 0 | 113.0 | 3.0 | 100 | 300 |
| BGK4/3.5 | 3.5 | 32 | 4.0 | 1.0 | 140 | 0 | 113.5 | 3.0 | 100 | 300 |
| BGK2/3.5 | 3.5 | 32 | 2.0 | 1.0 | 140 | 0 | 111.5 | 3.0 | 100 | 296 |
| BGK2/0 | 0 | 32 | 2.0 | 1.0 | 140 | 0 | 108.0 | 3.0 | 100 | 291 |
| B22GK4/0 | 0 | 22 | 4.0 | 1.5 | 140 | 0 | 120.5 | 3.0 | 100 | 296 |
| BGK4/0/1.2 | 0 | 32 | 4.0 | 1.2 | 140 | 0 | 110.2 | 3.0 | 100 | 295 |
| BK0/0/1.2 | 0 | 32 | 0 | 1.2 | 140 | 0 | 106.2 | 3.0 | 0 | 282 |
| PHOXILLUM Replacement Solutions | ||||||||||
| BK4/2.5 | 2.5 | 32 | 4.0 | 1.5 | 140 | 1 | 114.5 | 0 | 0 | 294 |
| B22K4/0 | 0 | 22 | 4.0 | 1.5 | 140 | 1 | 122.0 | 0 | 0 | 290 |
Select the mode of therapy, solute formulation, flow rates, and length of PRISMASOL and PHOXILLIUM replacement therapy in CRRT based on the patient's clinical condition, and fluid, electrolyte, acid-base, glucose balance. Administer either PRISMASOL or PHOXILLUM into the extracorporeal circuit:
- Before (pre-dilution) the hemofilter or hemodiafilter,
- After (post-dilution) the hemofilter or hemodiafilter, or
- Before and after the hemofilter or hemodiafilter.
Preparing the Solution
Use only if the overwrap is not damaged, all seals are intact, and the solution is clear. Press bag firmly to test for any leakage. Do not use if container is damaged or leaking.
The solution may be heated to no more than 40°C/104°F inside of the overwrap. After heating, verify that the solution remains clear and contains no particulate matter.
Follow the instructions below when connecting the solution bags for correct use of the access ports
Figure 1
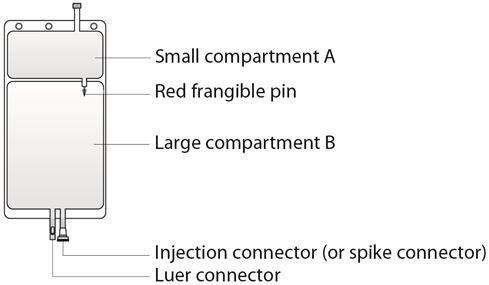
Figure 2 |
Step 1 Immediately before use, remove the overwrap from the bag and mix the solutions in the two different compartments. As soon as the overwrap is removed, the reconstitution of compartments A and B should be done and the mixed solution should be used immediately. After removal of the overwrap, the solution is stable for 24 hours including the duration of the treatment. Open the seal by breaking the red frangible pin between the two compartments of the bag. The frangible pin will remain in the bag. (See Figure 2 beside) |
Figure 3 |
Step 2 Make sure all the fluid from the small compartment A is transferred into the large compartment B. (See Figure 3 beside) |
| Step 3 Rinse the small compartment A twice by pressing the mixed solution back into the small compartment A and then back into the large compartment B. (See Figure 3 beside) | |
Figure 4 |
Step 4 When the small compartment A is empty, shake the large compartment B so that the contents mix completely. (See Figure 4 beside) The solution is now ready to use and the bag can be hung on the equipment. |
Figure 5a |
Step 5 The replacement line may be connected to the bag through either the luer connector or the injection connector (spike connector). Step 5a The luer connector is a needle-less and swabbable connector. Remove the cap with a twist and pull motion, and connect the male luer lock on the replacement line to the female luer receptor on the bag. (See Figure 5a beside) Ensure that the connection is fully seated and tighten. The connector is now open. Verify that the fluid is flowing freely during use. When the replacement line is disconnected from the luer connector, the connector will close and the flow of the solution will stop. |
Figure 5b |
Step 5b If the injection connector (spike connector) is used, first remove the snap-off cap. Then introduce the replacement line spike through the rubber septum of the bag connector. (See Figure 5b beside) Ensure that the spike is fully inserted and verify that the fluid is flowing freely during use. |
Adding Drugs to the Solutions
After mixing, additional drugs may be added to the bag via injection connector (spike connector) in large compartment B. In general, drugs other than phosphate should be administered through a different access line. When introducing additives, use aseptic techniques.
More about Phoxillum (renal replacement solution)
Professional resources
Other brands
See also:
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.

