Chondroprotec
This treatment applies to the following species: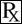 Company: Neogen
Company: Neogen
Polysulfated Glycosaminoglycan
INDICATIONS FOR USE:
● Partial and Full Thickness Wounds
● Secreting and Bleeding Dermal Lesions
● 1st and 2nd Degree Burns
● Pressure Ulcers (Stages I-IV)
● Surgical Incisions
● Donor Sites
● Venous Stasis Ulcers
FOR VETERINARY USE ONLY
Directions For Use
1. Irrigate wound with normal saline solution.
2. Either pour contents of vial or dispense from syringe to withdraw solution and cover wound site.
3. Cover with a non-adherent dressing such as PUF (polyurethane film) or Telfa®. Note: Subsequent applications of CHONDROPROTEC can be injected through the dressing as to not disturb the wound bed.
4. Reapply CHONDROPROTEC daily and redress wound site every 5 - 7 days or as needed.
Store at a temperature between 15° and 30°C (59°F - 86°F).
Protect from freezing. Discard unused portion.
Sterile when unopened.
For use on dogs, cats, and horses.
Not for food animal use.
NEOGEN®
Distributed by: Neogen Corporation, Lexington, KY 40511
800-525-2022
animalsafety.neogen.com
|
Net Contents: |
|
|
10 mL/1000 mg |
L145-0518 |
CPN: 1491021.4
944 NANDINO BLVD, LEXINGTON, KY, 40511-1205
| Telephone: | 859-254-1221 | |
| Order Desk: | 800-525-2022 | |
| Fax: | 859-255-5532 | |
| Website: | http://animalsafety.neogen.com | |
| Email: | inform@neogen.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |

Copyright © 2025 Animalytix LLC. Updated: 2025-08-27
