Ciprodex Prescribing Information
Package insert / product label
Generic name: ciprofloxacin and dexamethasone
Dosage form: otic suspension
Drug class: Otic steroids with anti-infectives
Medically reviewed by Drugs.com. Last updated on Jun 28, 2023.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
CIPRODEX® (ciprofloxacin and dexamethasone), otic suspension
Initial U.S. Approval: 2003
Indications and Usage for Ciprodex
CIPRODEX is a combination of ciprofloxacin, a fluoroquinolone antibacterial and dexamethasone, a corticosteroid, indicated for the treatment of infections caused by susceptible isolates of the designated microorganisms in the specific conditions listed below:
- Acute Otitis Media (AOM) in pediatric patients (age 6 months and older) with tympanostomy tubes due to Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa. (1)
- Acute Otitis Externa (AOE) in pediatric (age 6 months and older), adult, and elderly patients due to Staphylococcus aureus and Pseudomonas aeruginosa. (1)
Ciprodex Dosage and Administration
Dosage Forms and Strengths
Otic Suspension: Each mL of CIPRODEX contains ciprofloxacin hydrochloride 0.3% (equivalent to 3 mg ciprofloxacin base) and dexamethasone 0.1% (equivalent to 1 mg dexamethasone). (3)
Contraindications
- CIPRODEX is contraindicated in patients with a history of hypersensitivity to ciprofloxacin, to other quinolones, or to any of the components in this medication. (4)
- Use of this product is contraindicated in viral infections of the external canal, including herpes simplex infections and fungal otic infections. (4)
Warnings and Precautions
Adverse Reactions/Side Effects
Most common adverse reactions were ear discomfort (3%), ear pain (2.3%), and ear pruritus (1.5%). (6)
To report SUSPECTED ADVERSE REACTIONS, contact Novartis Pharmaceuticals Corporation at 1-888-669-6682 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2020
Related/similar drugs
amoxicillin, cephalexin, azithromycin, ceftriaxone, Augmentin, cefdinir, ceftazidime
Full Prescribing Information
1. Indications and Usage for Ciprodex
CIPRODEX® is indicated for the treatment of infections caused by susceptible isolates of the designated microorganisms in the specific conditions listed below:
- Acute Otitis Media (AOM) in pediatric patients (age 6 months and older) with tympanostomy tubes due to Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa.
- Acute Otitis Externa (AOE) in pediatric (age 6 months and older), adult and elderly patients due to Staphylococcus aureus and Pseudomonas aeruginosa.
2. Ciprodex Dosage and Administration
2.1 Important Administration Instructions
- CIPRODEX is for otic use (ears) only, and not for ophthalmic use, or for injection.
- Shake well immediately before use.
2.2 Dosage
For the Treatment of Acute Otitis Media in Pediatric Patients (age 6 months and older) With Tympanostomy Tubes
The recommended dosage regimen through tympanostomy tubes is as follows:
- Four drops [equivalent to 0.14 mL of CIPRODEX, (consisting of 0.42 mg of ciprofloxacin and 0.14 mg of dexamethasone)] instilled into the affected ear twice daily for seven days.
- The suspension should be warmed by holding the bottle in the hand for one or two minutes to avoid dizziness, which may result from the instillation of a cold suspension.
- The patient should lie with the affected ear upward, and then the drops should be instilled.
- The tragus should then be pumped 5 times by pushing inward to facilitate penetration of the drops into the middle ear.
- This position should be maintained for 60 seconds. Repeat, if necessary, for the opposite ear.
- Discard unused portion after therapy is completed.
For the Treatment of Acute Otitis Externa (age 6 months and older)
The recommended dosage regimen is as follows:
- Four drops [equivalent to 0.14 mL of CIPRODEX, (consisting of 0.42 mg ciprofloxacin and 0.14 mg dexamethasone)] instilled into the affected ear twice daily for seven days.
- The suspension should be warmed by holding the bottle in the hand for one or two minutes to avoid dizziness, which may result from the instillation of a cold suspension.
- The patient should lie with the affected ear upward, and then the drops should be instilled.
- This position should be maintained for 60 seconds to facilitate penetration of the drops into the ear canal. Repeat, if necessary, for the opposite ear.
- Discard unused portion after therapy is completed.
3. Dosage Forms and Strengths
Otic Suspension: Each mL of CIPRODEX contains ciprofloxacin hydrochloride 0.3% (equivalent to 3 mg ciprofloxacin base) and dexamethasone 0.1% equivalent to 1 mg dexamethasone.
4. Contraindications
- CIPRODEX is contraindicated in patients with a history of hypersensitivity to ciprofloxacin, to other quinolones, or to any of the components in this medication.
- Use of this product is contraindicated in viral infections of the external canal, including herpes simplex infections and fungal otic infections.
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
CIPRODEX should be discontinued at the first appearance of a skin rash or any other sign of hypersensitivity. Serious and occasionally fatal hypersensitivity (anaphylactic) reactions, some following the first dose, have been reported in patients receiving systemic quinolones. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal, or facial edema), airway obstruction, dyspnea, urticaria, and itching.
5.2 Potential for Microbial Overgrowth with Prolonged Use
Prolonged use of CIPRODEX may result in overgrowth of non-susceptible bacteria and fungi. If the infection is not improved after one week of treatment, cultures should be obtained to guide further treatment. If such infections occur, discontinue use and institute alternative therapy.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Potential for Microbial Overgrowth with Prolonged Use [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In Phases II and III clinical trials, a total of 937 patients were treated with CIPRODEX. This included 400 patients with acute otitis media with tympanostomy tubes (AOMT) and 537 patients with AOE. The reported adverse reactions are listed below:
Acute Otitis Media in Pediatric Patients with Tympanostomy Tubes
The following adverse reactions occurred in 0.5% or more of the patients with non-intact tympanic membranes.
| Adverse Reactions | Incidence (N = 400) |
|---|---|
| Ear discomfort | 3.0% |
| Ear pain | 2.3% |
| Ear precipitate (residue) | 0.5% |
| Irritability | 0.5% |
| Taste Perversion | 0.5% |
The following adverse reactions were each reported in a single patient: tympanostomy tube blockage; ear pruritus; tinnitus; oral moniliasis; crying; dizziness; and erythema.
Acute Otitis Externa
The following adverse reactions occurred in 0.4% or more of the patients with intact tympanic membranes.
| Adverse Reactions | Incidence (N = 537) |
|---|---|
| Ear pruritus | 1.5% |
| Ear debris | 0.6% |
| Superimposed ear infection | 0.6% |
| Ear congestion | 0.4% |
| Ear pain | 0.4% |
| Erythema | 0.4% |
The following adverse reactions were each reported in a single patient: ear discomfort; decreased hearing; and ear disorder (tingling).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of CIPRODEX. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions include auricular swelling, headache, hypersensitivity, otorrhea, skin exfoliation, rash erythematous, and vomiting.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are no available data on CIPRODEX use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal, or fetal outcomes. Because of the minimal systemic absorption of ciprofloxacin and dexamethasone following topical otic administration of CIPRODEX, this product is expected to be of minimal risk for maternal and fetal toxicity when administered to pregnant women [see Clinical Pharmacology (12.3)].
Animal reproduction studies have not been conducted with CIPRODEX. Oral administration of ciprofloxacin during organogenesis at doses up to 100 mg/kg to pregnant mice and rats, and up to 30 mg/kg to pregnant rabbits did not cause fetal malformations (see Data). These doses were at least 200 times the recommended otic human dose (ROHD in mice, rats, and rabbits, respectively, based on body surface area (BSA). With dexamethasone, malformations have been observed in animal studies after ocular and systemic administration.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and of miscarriage is 15% to 20%, respectively.
Data
Animal Data
Ciprofloxacin
Developmental toxicology studies have been performed with ciprofloxacin in rats, mice, and rabbits. The doses used in these studies are, at a minimum, approximately 200 times greater than the recommended otic human dose based on body surface area. In rats and mice, oral doses up to 100 mg/kg administered during organogenesis (Gestation Days [GD], 6-17) were not associated with adverse developmental outcomes, including embryofetal toxicity or malformations. A 30 mg/kg oral dose was associated with suppression of maternal and fetal body weight gain, but fetal malformations were not observed. Intravenous administration of doses up to 20 mg/kg to pregnant rabbits was not maternally toxic and neither embryo-fetal toxicity nor fetal malformations were observed. To mitigate maternal toxicity in these studies, groups of rabbits received ciprofloxacin for a different 5 day dosing period covering organogenesis (GD 6-18).
Dexamethasone
Dexamethasone has been shown to be teratogenic in mice and rabbits following topical ophthalmic application. In a rat oral developmental toxicity study, no adverse effects were observed at 0.01 mg/kg/day (0.1 times the ROHD based on BSA), although embryotoxicity was observed at higher doses.
8.2 Lactation
Risk Summary
It is not known whether ciprofloxacin and dexamethasone are present in human milk following topical otic administration.
Published literature reports the presence of ciprofloxacin in human milk after oral administration to lactating women. However, because of the minimal systemic absorption of ciprofloxacin following topical otic administration of CIPRODEX, breastfeeding is not expected to result in the exposure of the infant to ciprofloxacin [see Clinical Pharmacology (12.3)].
Systemically administered corticosteroids appear in human milk. Dexamethasone in breast milk could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. However, it is not known whether topical otic administration of CIPRODEX could result in systemic absorption that is sufficient to produce detectable quantities of dexamethasone in human milk.
There are no data on the effects of ciprofloxacin or dexamethasone on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CIPRODEX and any potential adverse effects on the breast-fed child from CIPRODEX.
8.4 Pediatric Use
The safety and efficacy of CIPRODEX have been established in pediatric patients 6 months and older (937 patients) in adequate and well-controlled clinical trials.
No clinically relevant changes in hearing function were observed in 69 pediatric patients (age 4 to 12 years) treated with CIPRODEX and tested for audiometric parameters.
10. Overdosage
Due to the characteristics of this preparation, no toxic effects are to be expected with an otic overdose of this product.
11. Ciprodex Description
CIPRODEX (ciprofloxacin 0.3% and dexamethasone 0.1%) Sterile Otic Suspension contains the quinolone antimicrobial, ciprofloxacin hydrochloride, combined with the corticosteroid, dexamethasone, in a sterile, preserved suspension for otic use. Each mL of CIPRODEX contains ciprofloxacin hydrochloride (equivalent to 3 mg ciprofloxacin base), 1 mg dexamethasone, and 0.1 mg benzalkonium chloride as a preservative. The inactive ingredients are acetic acid, boric acid, edetate disodium, hydroxyethyl cellulose, purified water, sodium acetate, sodium chloride, and tyloxapol. Sodium hydroxide or hydrochloric acid may be added for adjustment of pH.
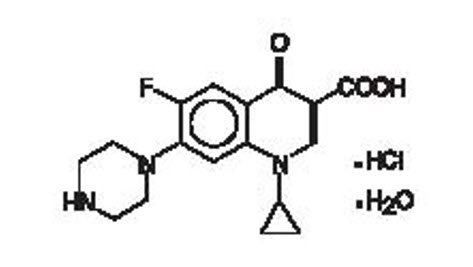
Ciprofloxacin, a quinolone antimicrobial is available as the monohydrochloride monohydrate salt of 1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-7-(1-piperazinyl)-3-quinoline carboxylic acid. The empirical formula is C17H18FN3O3·HCl·H2O. The molecular weight is 385.82 g/mol and the structural formula is:
Figure 1: Structure of Ciprofloxacin
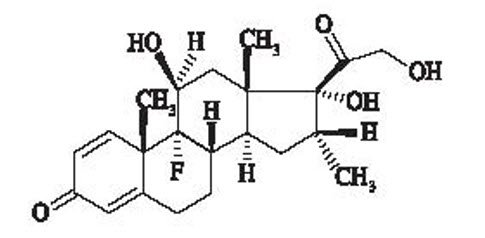
Dexamethasone, 9-fluoro-11(beta),17,21-trihydroxy-16(alpha)-methylpregna-1,4-diene-3,20-dione, is a corticosteroid. The empirical formula is C22H29FO5. The molecular weight is 392.46 g/mol and the structural formula is:
Figure 2: Structure of Dexamethasone
12. Ciprodex - Clinical Pharmacology
12.1 Mechanism of Action
Ciprofloxacin is a fluoroquinolone antibacterial [see Microbiology (12.4)].
Dexamethasone, a corticosteroid, has been shown to suppress inflammation by inhibiting multiple inflammatory cytokines resulting in decreased edema, fibrin deposition, capillary leakage, and migration of inflammatory cells.
12.3 Pharmacokinetics
Following a single bilateral 4-drop (total dose = 0.28 mL, 0.84 mg ciprofloxacin, 0.28 mg dexamethasone) topical otic dose of CIPRODEX to pediatric patients after tympanostomy tube insertion, measurable plasma concentrations of ciprofloxacin and dexamethasone were observed at 6 hours following administration in 2 of 9 patients and 5 of 9 patients, respectively.
Mean ± SD peak plasma concentrations of ciprofloxacin were 1.39 ± 0.880 ng/mL (n = 9). Peak plasma concentrations ranged from 0.543 ng/mL to 3.45 ng/mL and were on average approximately 0.1% of peak plasma concentrations achieved with an oral dose of 250-mg. Peak plasma concentrations of ciprofloxacin were observed within 15 minutes to 2 hours post dose application.
Mean ± SD peak plasma concentrations of dexamethasone were 1.14 ± 1.54 ng/mL (n = 9). Peak plasma concentrations ranged from 0.135 ng/mL to 5.10 ng/mL and were on average approximately 14% of peak concentrations reported in the literature following an oral 0.5-mg tablet dose. Peak plasma concentrations of dexamethasone were observed within 15 minutes to 2 hours post dose application.
Dexamethasone has been added to aid in the resolution of the inflammatory response accompanying bacterial infection (such as otorrhea in pediatric patients with AOMT).
12.4 Microbiology
Mechanism of Action
The bactericidal action of ciprofloxacin results from interference with the enzyme, DNA gyrase, which is needed for the synthesis of bacterial DNA.
Resistance
Cross-resistance has been observed between ciprofloxacin and other fluoroquinolones. There is generally no cross-resistance between ciprofloxacin and other classes of anti-bacterial agents, such as beta-lactams or aminoglycosides.
Antimicrobial Activity
Ciprofloxacin has been shown to be active against most isolates of the following microorganisms, both in vitro and clinically in otic infections [see Indications and Usage (1)].
Aerobic Bacteria
Gram-positive Bacteria
- Staphylococcus aureus
- Streptococcus pneumoniae
Gram-negative Bacteria
- Haemophilus influenzae
- Moraxella catarrhalis
- Pseudomonas aeruginosa
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term carcinogenicity studies in mice and rats have been completed for ciprofloxacin. After daily oral doses of 750 mg/kg (mice) and 250 mg/kg (rats) were administered for up to 2 years, there was no evidence that ciprofloxacin had any carcinogenic or tumorigenic effects in these species. No long-term studies of CIPRODEX have been performed to evaluate carcinogenic potential.
Long-term studies have not been performed to evaluate the carcinogenic potential of topical otic dexamethasone.
Mutagenesis
Eight in vitro mutagenicity tests have been conducted with ciprofloxacin, and the test results are listed below:
- Salmonella/Microsome Test (Negative)
- E. coli DNA Repair Assay (Negative)
- Mouse Lymphoma Cell Forward Mutation Assay (Positive)
- Chinese Hamster V79 Cell HGPRT Test (Negative)
- Syrian Hamster Embryo Cell Transformation Assay (Negative)
- Saccharomyces cerevisiae Point Mutation Assay (Negative)
- Saccharomyces cerevisiae Mitotic Crossover and Gene Conversion Assay (Negative)
- Rat Hepatocyte DNA Repair Assay (Positive)
Thus, 2 of the 8 tests were positive, but results of the following 3 in vivo test systems gave negative results:
- Rat Hepatocyte DNA Repair Assay
- Micronucleus Test (Mice)
- Dominant Lethal Test (Mice)
Dexamethasone has been tested for in vitro and in vivo genotoxic potential and shown to be positive in the following assays: chromosomal aberrations, sister-chromatid exchange in human lymphocytes, and micronuclei and sister-chromatid exchanges in mouse bone marrow. However, the Ames/Salmonella assay, both with and without S9 mix, did not show any increase in His+ revertants.
Impairment of Fertility
Fertility studies performed in male and female rats at oral doses of ciprofloxacin up to 100 mg/kg (approximately 482 times the ROHD of ciprofloxacin based on BSA) revealed no evidence of impairment. Male rats received oral ciprofloxacin for 10 weeks prior to mating and females were dosed for 3 weeks prior to mating through GD 7.
The effect of dexamethasone on fertility has not been investigated following topical otic application. However, the lowest toxic dose of dexamethasone identified following topical dermal application was 1.802 mg/kg in a 26-week study in male rats and resulted in changes to the testes, epididymis, sperm duct, prostate, seminal vesicle, Cowper's gland, and accessory glands. The relevance of this study for short-term topical otic use is unknown.
14. Clinical Studies
In a randomized, multicenter, controlled clinical trial, CIPRODEX dosed 2 times per day for 7 days demonstrated clinical cures in the per protocol analysis in 86% of AOMT patients compared to 79% for ofloxacin solution, 0.3%, dosed 2 times per day for 10 days. Among culture positive patients, clinical cures were 90% for CIPRODEX compared to 79% for ofloxacin solution, 0.3%. Microbiological eradication rates for these patients in the same clinical trial were 91% for CIPRODEX compared to 82% for ofloxacin solution, 0.3%.
In 2 randomized multicenter, controlled clinical trials, CIPRODEX dosed 2 times per day for 7 days demonstrated clinical cures in 87% and 94% of per protocol evaluable AOE patients, respectively, compared to 84% and 89%, respectively, for otic suspension containing neomycin 0.35%, polymyxin B 10,000 units/mL, and hydrocortisone 1.0% (neo/poly/HC). Among culture-positive patients, clinical cures were 86% and 92% for CIPRODEX compared to 84% and 89%, respectively, for neo/poly/HC. Microbiological eradication rates for these patients in the same clinical trials were 86% and 92% for CIPRODEX compared to 85% and 85%, respectively, for neo/poly/HC.
16. How is Ciprodex supplied
How Supplied
CIPRODEX (ciprofloxacin 0.3% and dexamethasone 0.1%) Sterile Otic Suspension is a white-to off-white suspension supplied as follows: 7.5 mL fill in a system consisting of a natural polyethylene bottle and natural plug, with a white polypropylene closure. Tamper evidence is provided with a shrink band around the closure and neck area of the package.
7.5 mL fill NDC 0078-0799-75
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature].
Avoid freezing. Protect from light.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
For Otic Use Only
Advise patients that CIPRODEX is for otic use (ears) only. This product must not be used in the eye [see Dosage and Administration (2.2)].
Administration Instructions
Instruct patients to warm the bottle in their hand for one to two minutes prior to use and shake well immediately before using [see Dosage and Administration (2.1, 2.2)].
Allergic Reactions
Advise patients to discontinue use immediately and contact their physician, if rash or allergic reaction occurs [see Warnings and Precautions (5.1)].
Avoid Contamination of the Product
Advise patients to avoid contaminating the tip with material from the ear, fingers, or other sources [see Instructions for Use].
Duration of Use
Advise patients that it is very important to use the eardrops for as long as their doctor has instructed, even if the symptoms improve [see Patient Information].
Protect from Light
Advise patients to protect the product from light [see How Supplied/Storage and Handling (16)].
Unused Product
Advise patients to discard unused portion after therapy is completed [see Dosage and Administration (2.2)].
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
T2020-141
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: November 2020 | |
| PATIENT INFORMATION
CIPRODEX® (CI-PRO-DEX) (ciprofloxacin and dexamethasone) otic suspension |
||
| What is CIPRODEX?
CIPRODEX is a prescription medicine used in the ear only (otic use) that contains 2 medicines, a quinolone antibiotic medicine called ciprofloxacin and a corticosteroid medicine called dexamethasone. CIPRODEX is used in adults and children 6 months of age or older to treat certain types of infections caused by certain germs called bacteria. These bacterial infections include:
|
||
| Who should not use CIPRODEX?
Do not use CIPRODEX if you:
|
||
| What should I tell my doctor before using CIPRODEX?
Before using CIPRODEX, tell your doctor about all of your medical conditions, including if you:
|
||
How should I use CIPRODEX?
|
||
| What are the possible side effects of CIPRODEX? CIPRODEX may cause serious side effects, including:
|
||
|
|
|
The most common side effects of CIPRODEX include:
|
||
How should I store CIPRODEX?
|
||
| General information about the safe and effective use of CIPRODEX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use CIPRODEX for a condition for which it was not prescribed. Do not give CIPRODEX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about CIPRODEX that is written for health professionals. |
||
| What are the ingredients in CIPRODEX?
Active ingredients: ciprofloxacin hydrochloride, dexamethasone, and benzalkonium chloride as a preservative Inactive ingredients: acetic acid, boric acid, edetate disodium, hydroxyethyl cellulose, purified water, sodium acetate, sodium chloride, and tyloxapol. Sodium hydroxide or hydrochloric acid may be added for adjustment of pH. Distributed by: Novartis Pharmaceuticals Corporation East Hanover, New Jersey 07936 © Novartis |
||
T2020-142
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: November 2020 |
| INSTRUCTIONS FOR USE CIPRODEX® (CI-PRO-DEX) (ciprofloxacin and dexamethasone) otic suspension |
|
| This “Instructions for Use” contains information on how to use CIPRODEX. | |
| Important Information You Need to Know Before Using CIPRODEX.
Read this Instructions for Use that comes with CIPRODEX before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or treatment.
|
|
| How should I use CIPRODEX?
Step 1. Wash your hands with soap and water (see Figure A). Figure A  |
|
| Step 2. Warm the bottle of CIPRODEX by rolling the bottle between your hands for 1 to 2 minutes (see Figure B). Shake the bottle of CIPRODEX well. Figure B  |
|
|
Step 3. Remove the CIPRODEX cap. Put the cap in a clean and dry area. Do not let the tip of the bottle touch your ear, fingers or other surfaces. Step 4. Lie down on your side so that the affected ear faces upward (see Figure C). Figure C 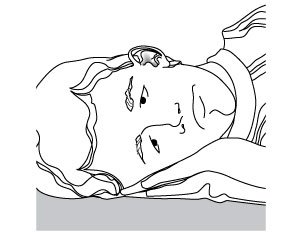 |
|
|
Step 5. Hold the bottle of CIPRODEX between your thumb and index finger (see Figure D). Place the tip of the bottle close to your ear. Be careful not to touch your fingers or ear with the tip of the bottle. Figure D 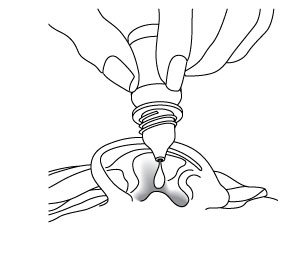 |
|
|
Step 6. Gently squeeze the bottle and let 4 drops of CIPRODEX fall into the affected ear. If a drop misses your ear, follow the instructions in Step 5 again. |
|
|
Step 7. Stay on your side with the affected ear facing upward (see Figure C). It is important that you follow the instructions below for your specific ear infection, to allow CIPRODEX to enter the affected part of your ear. |
|
|
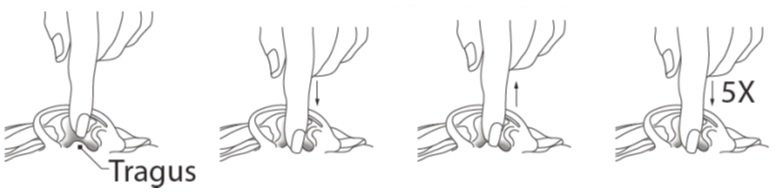
Step 8. If you use CIPRODEX to treat a middle ear infection, and you have a tube in your eardrum known as a tympanostomy:
 |
|
|
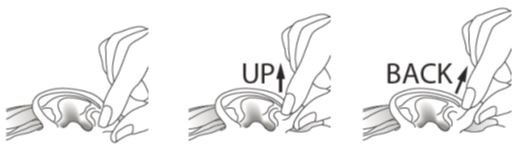
If you use CIPRODEX to treat an outer ear canal infection:
 |
|
|
Step 9. If your doctor has told you to use CIPRODEX in both ears, repeat steps 5-8 for your other ear. |
|
|
Step 10. Put the cap back on the bottle and close it tightly. |
|
|
Step 11. After you have used all of your CIPRODEX doses, there may be some CIPRODEX left in the bottle. Throw the bottle away. |
|
How should I store CIPRODEX?
If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for more information about CIPRODEX that is written for health professionals. |
|
| Distributed by: Novartis Pharmaceuticals Corporation East Hanover, New Jersey 07936 © Novartis |
|
T2020-143
| CIPRODEX
ciprofloxacin and dexamethasone suspension/ drops |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Novartis Pharmaceuticals Corporation (002147023) |
More about Ciprodex (ciprofloxacin / dexamethasone otic)
- Compare alternatives
- Reviews (100)
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- Drug class: otic steroids with anti-infectives

