UlcerGard: Package Insert / Prescribing Info
Package insert / product label
Generic name: omeprazole oral paste
Dosage form: FOR ANIMAL USE ONLY
On This Page
Stomach ulcers are more common in horses than once believed. Studies have demonstrated stomach ulcers in over 90% of racehorses and 60% of performance horses in various disciplines. Horses secrete acid in their stomach 24 hours a day. Horses on grass pasture or eating large quantities of roughage infrequently develop stomach ulcers. Horses without continual access to roughage or in rigorous training are at greater risk of stomach ulceration due to the effects of acid on the stomach lining. The additional stresses of traveling, competition, stall confinement, and strenuous exercise can also contribute to the development of stomach ulcers in horses.
ULCERGARD works to prevent stomach ulcers in horses. The active ingredient in ULCERGARD, omeprazole has been extensively tested for safety and effectiveness. It works by reducing the production of acid in the horse's stomach. To help horses maintain optimal gastric health, it is recommended that ULCERGARD be administered during stressful events or activities that may induce stomach ulcers. Such conditions may include: training, racing, showing, traveling, stall confinement, and competition.
DIRECTIONS FOR USE
Each syringe contains 4 individual daily doses for horses weighing 600-1200 lbs. Please refer to the following dosage chart for help in determining the correct dose for your horse.
| HORSE WEIGHT | DOSE* |
|---|---|
|
|
| Less than 600 lbs | Consult a veterinarian |
| 600-1200 lbs | 1 dose per day |
| over 1200 lbs | 2 doses once per day |
SYRINGE INSTRUCTIONS
- 1)
- To set the syringe plunger, unlock the dose ring by rotating ¼ turn and slide the dose ring along the plunger shaft so that the side nearest the barrel is set at the appropriate daily dose marking.
- 2)
- Rotate ¼ turn to lock ring before dosing.
- 3)
- Make sure horse's mouth contains no feed before administration.
- 4)
- Remove syringe tip cover.
- 5)
- Insert syringe into the corner of the horse's mouth.
- 6)
- Depress the plunger until it stops at the dose ring.
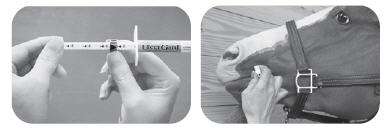
- The entire dose should be deposited on the back of the tongue or deep in the cheek pouch.
- Horses should be observed briefly to assure no part of the dose is lost or rejected.
- If any of the dose is lost, re-dosing is recommended.
- Replace cap if any unused doses remain in the syringe.
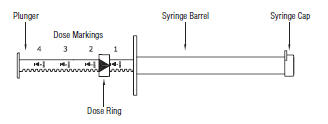
UlcerGard Dosage and Administration
The minimum recommended dosage is 1 mg/kg per day (0.45 mg/lb) or 1/4 syringe. When given once daily during the stressful period, ULCERGARD has been shown to effectively prevent stomach ulcers in horses. This effect was tested in horses exposed to stressful conditions for either 8 or 28 days.
Warnings
Not for use in humans. Keep this and all medications out of the reach of children. In case of ingestion by humans, contact a physician. Do not use in horses intended for human consumption. To obtain product information call 1-877-MERIAL-E.
INFORMATION FOR HORSE OWNERS
- ULCERGARD is intended for use in healthy horses. If you notice any signs of illness prior to or during the use of this product, consult your veterinarian for appropriate diagnosis and treatment recommendations.
- ULCERGARD may be used in horses that weigh at least 600 lbs. The effectiveness of ULCERGARD in the prevention of gastric ulcers in foals and weanlings has not been evaluated.
- ULCERGARD may be used safely in breeding stallions. Safety in pregnant mares has not been determined.
- Once daily administration of ULCERGARD is recommended in horses exposed to stressful conditions which may include: training, racing, showing, traveling, stall confinement, and competition.
- ULCERGARD is intended for use only in the prevention of gastric ulcers in healthy horses. Clinical signs of gastric ulcers may include: decreased appetite, recurrent colic, intermittent loose stools or diarrhea, poor hair coat, poor body condition or poor performance. These signs may also be associated with other diseases as well as existing gastric ulcers. If your horse is exhibiting one or more of these signs, consult your veterinarian for diagnosis and treatment.
Storage and Handling
Store at 68°F-77°F (20-25°C). Excursions between 59°F-86°F (15-30°C) are permitted.
CUSTOMER ASSISTANCE AND WEB SITE
For more information, please call 1-877-MERIAL-E or visit our web site: www.ULCERGARD.com.
| ULCERGARD
omeprazole paste |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Merial Limited (034393582) |

