Sterile Water for Irrigation: Package Insert / Prescribing Info
Package insert / product label
Dosage form: irrigation
Medically reviewed by Drugs.com. Last updated on Sep 2, 2025.
On This Page
Sterile Water for Irrigation Description
Sterile Water for Irrigation USP is a sterile, hypotonic, nonpyrogenic irrigating fluid or pharmaceutic aid (solvent) entirely composed of Sterile Water for Injection USP. It is prepared by distillation and contains no antimicrobial or bacteriostatic agents or added buffers. The pH is 5.7 (5.0–7.0)
The plastic container is a copolymer of ethylene and propylene formulated and developed for parenteral drugs. The copolymer contains no plasticizers and exhibits virtually no leachability. The plastic container is also virtually impermeable to vapor transmission and therefore, requires no overwrap to maintain the proper drug concentration. The safety of the plastic container has been confirmed by biological evaluation procedures. The material passes Class Vl testing as specified in the U.S. Pharmacopeia for Biological Tests — Plastic Containers. These tests have shown that the container is nontoxic and biologically inert.
Not made with natural rubber Latex, PVC or DEHP.
Sterile Water for Irrigation - Clinical Pharmacology
Sterile Water for Irrigation USP is utilized for a variety of clinical indications. Because of its low refractive index (1.3325), water provides excellent visibility during endoscopic urological procedures. It is also utilized as a pharmaceutic aid, as well as in the preparation of enteral nutrient products.
Water is hypotonic and will cause hemolysis and will be readily absorbed by the tissues during surgical procedures; therefore, its use under such conditions is not recommended.
Indications and Usage for Sterile Water for Irrigation
Sterile Water for Irrigation USP is indicated for use as an irrigating fluid or pharmaceutic aid. Sterile Water may also be used as an adjunct in the preparation of non-intravenously administered nutrient mixtures (see DOSAGE AND ADMINISTRATION).
Warnings
Sterile Water for Irrigation USP is hypotonic and will cause hemolysis, and is not recommended for use during surgical procedures.
After opening container, its contents should be used promptly to minimize the possibility of bacterial growth or pyrogen formation.
Discard unused portion of irrigating solution since it contains no preservative.
Sterile Water for Irrigation Dosage and Administration
Irrigation
Use as directed by physician.
This drug product should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Nutrient Mixtures
Sterile Water for Irrigation USP may be used to prepare non-intravenously administered nutrient mixtures. It contains no electrolytes or other added substances. Refer to preparation instructions of particular mixture to be used. The plastic container may be used for administration of non-intravenous nutrient mixture to the patient as appropriate.
How is Sterile Water for Irrigation supplied
Sterile Water for Irrigation USP is supplied sterile and nonpyrogenic in plastic irrigating containers. The 1000 mL and 500 mL containers are packaged 16 per case.
| NDC | Cat. No. | Size |
|---|---|---|
| Sterile Water for Irrigation USP (Canada DIN 01963961) | ||
| 0264-2101-00 | R5000-01 | 1000 mL |
| 0264-2101-10 | R5001-01 | 500 mL |
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.
Do not warm above 150°F (66°C).
Directions for Use of PIC™ (Plastic Irrigation Container)
Not for injection.
Aseptic technique is required.
- Caution – Before use, perform the following checks:
(a) Read the label. Ensure solution is the one ordered and is within the expiration date.
(b) Invert container and inspect the solution in good light for cloudiness, haze, or particulate matter; check the container for leakage or damage. Any container which is suspect should not be used.
Use only if solution is clear and container and seal are intact.
Single-dose container. Discard unused portion.
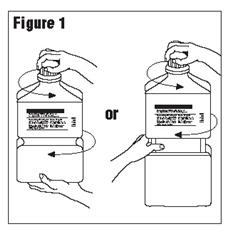
- Outer Closure Removal – Grasp the container with one hand and turn the breakaway ring counterclockwise with the other hand until slight resistance is felt. Then, twisting the container in the opposite direction, turn the breakaway ring sharply until the entire outer cap is loose and can be lifted off. (Figure 1)
- Connect the administration set through the sterile set port according to set instructions (Figure 2) or remove screw cap and pour.
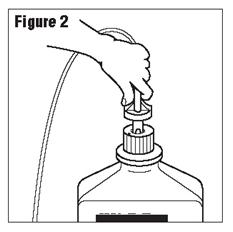
- Do not warm above 150°F (66°C) to assure minimal bottle distortion. Keep bottles upright.
B. Braun Medical Inc.
Bethlehem, PA 18018-3524 USA
1-800-227-2862
Imported by:
B. Braun of Canada, Ltd.
Scarborough, Ontario M1H 2W4
Y36-003-075 LD-774-1
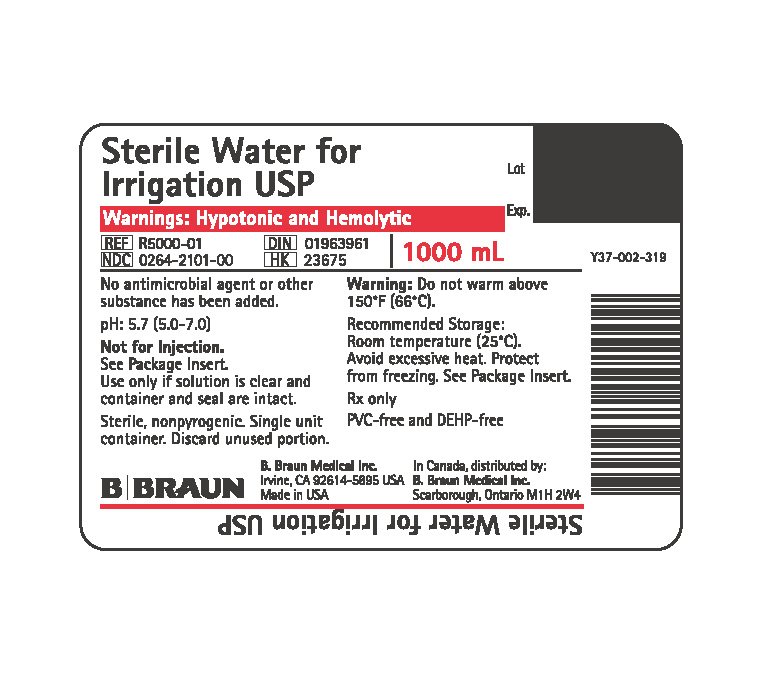
PRINCIPAL DISPLAY PANEL - 1000 mL Container
Sterile Water for Irrigation USP
Warnings: Hypotonic and Hemolytic
REF R5000-01
NDC 0264-2101-00
DIN 01963961
HK 23675
1000 mL
PIC™ Container
Rx only
Lot
Exp.
No antimicrobial agent or other substance has been added.
pH: 5.7 (5.0-7.0)
Not for Injection.
See Package Insert.
Use only if solution is clear and container and seal are intact.
Sterile, nonpyrogenic. Single-dose container. Discard unused portion.
Warning: Do not warm above 150°F (66°C).
Recommended Storage: Room temperature (25°C). Avoid excessive heat. Protect from freezing.
Dosage: See Prescribing Information.
Not made with natural rubber latex, PVC or DEHP.
Y37-002-594 LD-775-1
B. Braun Medical Inc.
Bethlehem, PA 18018-3524 USA
1-800-227-2862
Imported by:
B. Braun of Canada, Ltd.
Scarborough, Ontario M1H 2W4
Sterile Water for Irrigation USP
PRINCIPAL DISPLAY PANEL - 500 mL Container
Sterile Water for Irrigation USP
Warnings: Hypotonic and Hemolytic
REF R5001-01
NDC 0264-2101-10
DIN 01963961
HK 23675
500 mL
PIC™ Container
Rx only
Lot
Exp.
No antimicrobial agent or other substance has been added.
pH: 5.7 (5.0-7.0)
Not for Injection.
See Package Insert.
Use only if solution is clear and container and seal are intact.
Sterile, nonpyrogenic. Single-dose container. Discard unused portion.
Warning: Do not warm above 150°F (66°C).
Recommended Storage: Room temperature (25°C). Avoid excessive heat. Protect from freezing.
Dosage: See Prescribing Information.
Not made with natural rubber latex, PVC or DEHP.
Y37-002-595 LD-776-1
B. Braun Medical Inc.
Bethlehem, PA 18018-3524 USA
1-800-227-2862
Imported by:
B. Braun of Canada, Ltd.
Scarborough, Ontario M1H 2W4
Sterile Water for Irrigation USP

| STERILE WATER
water irrigant |
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - B. Braun Medical Inc. (002397347) |
