PS1 Aciurgy Pack: Package Insert / Prescribing Info
Package insert / product label
Generic name: mupirocin, chlorhexidine gluconate, povidone-iodine
Dosage form: kit
On This Page
PS1 Aciurgy Pack Description
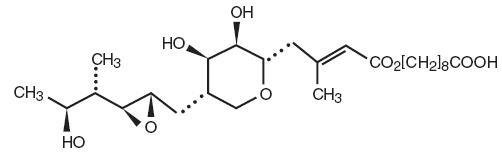
Each gram of Mupirocin Ointment USP, 2% contains 20 mg mupirocin in a bland water miscible ointment base (polyethylene glycol ointment, NF) consisting of polyethylene glycol 400 and polyethylene glycol 3350. Mupirocin is a naturally occurring antibiotic. The chemical name is ( E)-(2 S,3 R,4 R,5 S)-5-[(2 S,3 S,4 S,5 S)-2,3-Epoxy-5-hydroxy-4-methylhexyl]tetrahydro-3,4-dihydroxy-β-methyl-2 H-pyran-2-crotonic acid, ester with 9-hydroxynonanoic acid. The molecular formula of mupirocin is C 26 H 44 O 9 and the molecular weight is 500.62.
The chemical structure is:
PS1 Aciurgy Pack - Clinical Pharmacology
Application of 14C-labeled mupirocin ointment to the lower arm of normal male subjects followed by occlusion for 24 hours showed no measurable systemic absorption (<1.1 nanogram mupirocin per milliliter of whole blood). Measurable radioactivity was present in the stratum corneum of these subjects 72 hours after application.
Following intravenous or oral administration, mupirocin is rapidly metabolized. The principal metabolite, monic acid, is eliminated by renal excretion, and demonstrates no antibacterial activity. In a trial conducted in 7 healthy adult male subjects, the elimination half-life after intravenous administration of mupirocin was 20 to 40 minutes for mupirocin and 30 to 80 minutes for monic acid. The pharmacokinetics of mupirocin has not been studied in individuals with renal insufficiency.
Microbiology -
Mupirocin is an antibacterial agent produced by fermentation using the organism Pseudomonas fluorescens. Mupirocin inhibits bacterial protein synthesis by reversibly and specifically binding to bacterial isoleucyl transfer-RNA (tRNA) synthetase. Due to this unique mode of action, mupirocin does not demonstrate cross-resistance with other classes of antimicrobial agents.
When mupirocin resistance occurs, it results from the production of a modified isoleucyl-tRNA synthetase, or the acquisition of, by genetic transfer, a plasmid mediating a new isoleucyl-tRNA synthetase. High-level plasmid-mediated resistance (MIC >512 mcg/mL) has been reported in increasing numbers of isolates of Staphylococcus aureus and with higher frequency in coagulase-negative staphylococci. Mupirocin resistance occurs with greater frequency in methicillin-resistant than methicillin-susceptible staphylococci. Because of the occurrence of mupirocin resistance in methicillin-resistant Staphylococcus aureu (MRSA), it is appropriate to test MRSA populations for mupirocin susceptibility prior to the use of mupirocin using a standardized method. 1,2,3
Mupirocin is bactericidal at concentrations achieved by topical administration. Mupirocin is highly protein-bound (>97%), and the effect of wound secretions on the MICs of mupirocin has not been determined.
Mupirocin has been shown to be active against susceptible strains of S. aureus and Streptococcus pyogenes, both in vitro and in clinical trials (see INDICATIONS AND USAGE). The following in vitro data are available, but their clinical significance is unknown. Mupirocin is active against most isolates of Staphylococcus epidermidis.
Indications and Usage for PS1 Aciurgy Pack
Mupirocin Ointment USP, 2% is indicated for the topical treatment of impetigo due to: S. aureus and S. pyogenes.
Contraindications
This drug is contraindicated in patients with known hypersensitivity to any of the constituents of the product.
Warnings
Avoid contact with the eyes. In case of accidental contact, rinse well with water.
In the event of sensitization or severe local irritation from Mupirocin Ointment USP, 2%, usage should be discontinued.
Clostridium difficile- associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including mupirocin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Precautions
As with other antibacterial products, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi.
Mupirocin Ointment USP, 2% is not formulated for use on mucosal surfaces. Intranasal use has been associated with isolated reports of stinging and drying.
A paraffin-based formulation - *Bactroban Nasal® (mupirocin calcium ointment ) - is available for intranasal use.
Polyethylene glycol can be absorbed from open wounds and damaged skin and is excreted by the kidneys. In common with other polyethylene glycol-based ointments, Mupirocin Ointment USP, 2% should not be used in conditions where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment.
Mupirocin Ointment USP, 2% should not be used with intravenous cannulae or at central intravenous sites because of the potential to promote fungal infections and antimicrobial resistance.
Information for Patients -
Use this medication only as directed by the healthcare provider. It is for external use only. Avoid contact with the eyes. If Mupirocin Ointment USP, 2% gets in or near the eyes, rinse thoroughly with water. The medication should be stopped and the healthcare provider contacted if irritation, severe itching, or rash occurs. If impetigo has not improved in 3 to 5 days, contact the healthcare provider.
Carcinogenesis, Mutagenesis, Impairment of Fertility -
Long-term studies in animals to evaluate carcinogenic potential of mupirocin have not been conducted.
Results of the following studies performed with mupirocin calcium or mupirocin sodium in vitro and in vivo did not indicate a potential for genotoxicity: Rat primary hepatocyte unscheduled DNA synthesis, sediment analysis for DNA strand breaks, Salmonella reversion test (Ames), Escherichia coli mutation assay, metaphase analysis of human lymphocytes, mouse lymphoma assay, and bone marrow micronuclei assay in mice.
Reproduction studies were performed in male and female rats with mupirocin administered subcutaneously at doses up to 14 times a human topical dose (approximately 60 mg mupirocin per day) on a mg/m 2 basis and revealed no evidence of impaired fertility and reproductive performance from mupirocin.
Pregnancy:
Teratogenic Effects:
Pregnancy Category B -
Reproduction studies have been performed in rats and rabbits with mupirocin administered subcutaneously at doses up to 22 and 43 times, respectively, the human topical dose (approximately 60 mg mupirocin per day) on a mg/m 2 basis and revealed no evidence of harm to the fetus due to mupirocin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers -
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Mupirocin Ointment USP, 2% is administered to a nursing woman.
Pediatric Use -
The safety and effectiveness of Mupirocin Ointment USP, 2% have been established in the age range of 2 months to 16 years. Use of mupirocin ointment USP, 2% in these age groups is supported by evidence from adequate and well-controlled trials of mupirocin ointment USP, 2% in impetigo in pediatric subjects studied as part of the pivotal clinical trials (see CLINICAL STUDIES).
Adverse Reactions/Side Effects
The following local adverse reactions have been reported in connection with the use of mupirocin ointment USP, 2%: burning, stinging, or pain in 1.5% of subjects; itching in 1% of subjects; rash, nausea, erythema, dry skin, tenderness, swelling, contact dermatitis, and increased exudate in less than 1% of subjects. Systemic allergic reactions, including anaphylaxis, urticaria, angioedema and generalized rash have been reported in patients treated with mupirocin formulations.
PS1 Aciurgy Pack Dosage and Administration
A small amount of Mupirocin Ointment USP, 2% should be applied to the affected area 3 times daily. The area treated may be covered with a gauze dressing if desired. Patients not showing a clinical response within 3 to 5 days should be re-evaluated.
Clinical Studies
The efficacy of topical mupirocin ointment USP, 2% in impetigo was tested in 2 trials. In the first, subjects with impetigo were randomized to receive either mupirocin ointment USP, 2% or vehicle placebo 3 times daily for 8 to 12 days. Clinical efficacy rates at end of therapy in the evaluable populations (adults and pediatric subjects included) were 71% for mupirocin ointment USP, 2% (n=49) and 35% for vehicle placebo (n=51). Pathogen eradication rates in the evaluable populations were 94% for mupirocin ointment USP, 2% and 62% for vehicle placebo. There were no side effects reported in the group receiving mupirocin ointment USP, 2%. In the second trial, subjects with impetigo were randomized to receive either mupirocin ointment USP, 2% 3 times daily or 30 to 40 mg/kg oral erythromycin ethylsuccinate per day (this was an unblinded trial) for 8 days. There was a follow-up visit 1 week after treatment ended. Clinical efficacy rates at the follow-up visit in the evaluable populations (adults and pediatric subjects included) were 93% for mupirocin ointment USP, 2% (n=29) and 78.5% for erythromycin (n=28). Pathogen eradication rates in the evaluable populations were 100% for both test groups. There were no side effects reported in the group receiving mupirocin ointment USP, 2%.
Pediatrics -
There were 91 pediatric subjects aged 2 months to 15 years in the first trial described above. Clinical efficacy rates at end of therapy in the evaluable populations were 78% for mupirocin ointment USP, 2% (n=42) and 36% for vehicle placebo (n=49). In the second trial described above, all subjects were pediatric except 2 adults in the group receiving mupirocin ointment USP, 2%. The age range of the pediatric subjects was 7 months to 13 years. The clinical efficacy rate for mupirocin ointment USP, 2% (n=27) was 96%, and for erythromycin it was unchanged (78.5%).
How is PS1 Aciurgy Pack supplied
Mupirocin Ointment USP, 2% is available as follows:
22 g tube (NDC 45802- 112-22)
Store at 20-25°C (68-77°F) [see USP Controlled Room Temperature].
*Bactroban Nasal® is a registered trademark of GlaxoSmithKline.
REFERENCES
1. Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard -Tenth Edition. CLSI document M07-A10 [2015], Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA.
2. Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Diffusion Susceptibility Tests; Approved Standard – Twelfth Edition. CLSI document M02-A12 [2015], Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA.
3. Finlay JE, Miller LA, Poupard JA. Interpretive criteria for testing susceptibility of staphylococci to mupirocin. Antimicrob Agents Chemother 1997;41(5):1137-1139.
Made in Israel
Manufactured By Perrigo
Yeruham 80500, Israel
Distributed By
Perrigo®
Allegan, MI 49010 • www.perrigo.com
Rev 05-15
1N200 RC J6
Indications and Usage for PS1 Aciurgy Pack
- surgical hand scrub: significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care
- healhcare personnel handwash: helps reduce bacteria that potentially can cause disease
- patient preoperative skin preparation: for the preparation of the patient's skin prior to surgery
- skin wound and general skin cleansing
Warnings
For external use only
Alergy alert:
This product may cause a severe allergic reaction. Symptoms may incloude
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
if an allergic reaction occurs, stop use and seek medical help right away
Do not use
- if you or the patient is allergic to chlorhexidine gluconate or any other ingredient in this product
- in contact with meninges
- in the genital area
- as a preoperative skin preparation of the head or face
When using this product
- keep out of the eyes, ears and mouth. May cause serious and permenant eye injury if placed or kept in the eye during surgical procedures or may cause deafness when instilled in the middle ear through perforated ear drums.
- if solution should contact these areas, rinse our promptly and thoroughly with water
- wounds which involve more than the superficial layers of the skin should not be routinely treated
- repeated general skin cleansing of large body areas should not be done ecept when advised be a health care provider
Stop use and ask a doctor if
irritation, sensitization, or allergic reaction occurs. These may be signs of a serious condition.
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
PS1 Aciurgy Pack Dosage and Administration
use with care in premature infants or infants under 2 months of age. These products may cause irritation or chemical burns.
Surgical hand scrub:
- wet hands and forearms with water
- scrub for 3 minutes with about 5 mL of product and a wet brush paying close attention to the nails, cuticles amd interdigital spaces
- a seprate nail cleaner may be used
- rinse thoroughly
- wash for an additional 3 minutes with 5 mL of product and rinse under running water
- dry thoroughly
Healthcare personnel handwash:
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 15 seconds
- rinse and dry thoroughly
Patient preoperative skin preparation:
- apply product liberally to surgical site and swab for at least 2 minutes and dry with a sterile towel
- repeat procedure for an additional 2 minutes and dry with a sterile towel
Skin wound and general skin cleaning:
- thoroughly rinse the area to be cleaned with water
- apply the minimum amount of product necessary to cover the skin or wound area and wash gently
- rinse again thoroughly
Inactive ingredients
cocamide DEA, fragrance, glucono-delta-lactone, hydroxethylcellulose, isopropyl alcohol, lauramine oxide, PEG-75 lanolin, purified water, tridecyl alcohol
Indications and Usage for PS1 Aciurgy Pack
- First aid antiseptic to help prevent infection in minor cuts, scrapes and burns
Warnings
For external use only.
Do not use
- in the eyes
- longer than 1 week unless directed by a physician
- on individuals who are allergic or sensitive to iodine
- or apply over large areas of the body
| PS1 ACIURGY PACK
mupirocin 2%, chlorhexidine gluconate 4%, povidone-iodine kit |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Oaklock, LLC (079559179) |




