MTL1 Leucoflex: Package Insert / Prescribing Info
Package insert / product label
Generic name: anticoagulant citrate phosphate dextrose
Dosage form: kit
On This Page
MTL1 Leucoflex Description
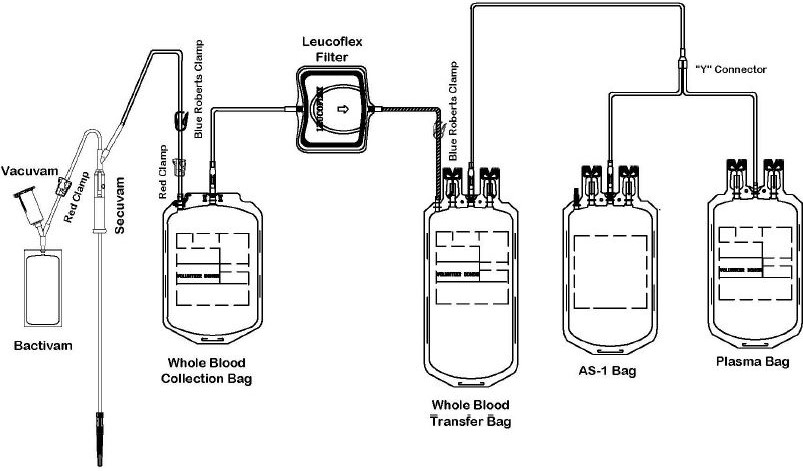
The Leucoflex MTL1 product consists of a blood collection system with 70mL of CPD for the collecion of 500 +/- 50mL of blood and 110mL of AS-1 Additive Solution. They are supplied with sterile, non-pyrogenic fluid pathways. The product as supplied includes an in-line leukoreduction filter known as Leucoflex MTL1, storage containers for blood components and tubing, a sample diversion pouch (Bactivam), a vacuum tube adapter (Vacuvam), and a protective shield for the used needle (Secuvam).
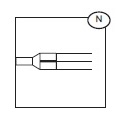
Representative Product Drawing
Intended Use
The "Leucoflex MTL1 Leukocyte Reduction Filter for Whole Blood" is intended for the pre-storage leukoreduction of whole blood initiated between 4 and 7 hours after collection if whole blood is stored at ambient temperature, or between 4 and 8 hours of storage at 1 to 6 C. The collection set provides for subsequent preparation of AS-1 Red Blood Cells, Leukocytes Reduced (adenine saline added) and Plasma, Leukocytes Reduced in a closed system.
The AS-1 Red Blood Cells, Leukocytes Reduced and Plasma, Leukocytes Reduced may then be stored for the maximum allowable dating periods.
Warnings
- Avoid contact with sharp objects.
- DO NOT USE if the overwrap or blood bag system shows any signs of deterioration.
- DO NOT USE if the solutions are not clear.
- Dispose of all system components that have been contaminated with blood in a biohazard container as per your institution's SOP.
- Dispose of all sharps as per your institution's SOP.
Precautions
- Do not fold or squeeze the Leucolex MTL1 filter. Inappropriate handling may adversely affect filtration.
- Check for kinks in the tubing prior to collection and filtration.
- Rx only.
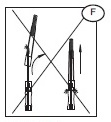
The protective shield for needle Secuvam (A) and the vacuum tube adapter Vacuvam (B) features of the collection set aid in the safe collection of vacuum tube samples from the sample diversion pouch Bactivam (C) and the disposal of donor needles after blood collection by providing protection from accidental needlestick.
All or part of this medical device is made of PVC plasticized with DEHP. According to some studies, DEHP could potentially be harmful to the reproductive system of male fetuses. The prescriber is solely responsible for choosing to use this device on women who are either pregnant or breast-feeding, or on young male infants. Nevertheless, DEHP-plasticized PVC is in compliance with the European Pharmacopeia.
Risks linked to the reuse: Septic risk.
All maunfacturing processes and all components in contact with the donors, the users and the blood components dedicated to the patients are not made with natural rubber latex.
COLLECTION AND SAMPLING PROCEDURE
Before Donation
1) Peel open the transparent overwrap (D) using the corner flap.
2) Support blood bag system as far as possible below donor arm, ideally on an automated blood mixer.
Note: it is recommended that the primary bag on the mixer lies with the tubing on the lower side of the mixer holding plate.
3) Apply tourniquet or pressure cuff (inflate to approximately 60 mm Hg).
4) Prepare venipuncture site as per your SOP.
5) Ensure that the donor line is not twisted or tangled.
Venipucture
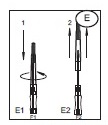
1) Open the needle cap (E1) using the following procedure:
a) Twist the needle cap, thus breaking the seal.
b) Lift the cap off of the needle (E2) in a straight line taking special care not to touch the needle in the process (F). Keep hands behind the needle at all times during set use and disposal.
2) Perform the venipuncture
3) After the needle has been inserted, introduce the Secuvam onto the hub of the needle.
4) Tape the Secuvam onto the donor's arm.
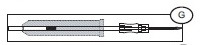
Note: if the Secuvam causes discomfort to the donor or decreases the blood flow because of the angle of the needle, leave the Secuvam on the tubing, behind the needle hub (G) during collection. Secure the needle into the Secuvam at the end of the donation (H).
After Venipuncture
1) Ensure that the clamp between the Bactivam pouch and the donation line is open.
2) Ensure that the Bactivam pouch is held below the level of the venipuncture site, and is supported at all times.
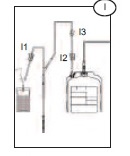
3) Allow blood to run into the Bacivam pouch until the pouch fills (approximately 30ml) (I).
4) Close the red clamp (I1) on the transfer line to the Bactivam pouch, by holding the clamp in one hand and squeezing the two edges together, until it locks (a click my be heard).
Note: the red clamp cannot be opened after it has snapped shut.
5) Break the break-away cannula (I2) on the donor line by holding the cannula in one hand and the plastic Y-junction in the other and snapping the cannula both left and right to ensure a clean break. Allow the donation procedure to continue, ensuring that the blood bag is mixed immediately when blood flows into the primary bag.
Note: This step may be done prior to venipuncture if the Blue Roberts clamp (I3) on the donor line is closed prior to breaking the break-away cannula.
6) If using an automated mixer, open the mixer clamp and start the mixer at this point.
Note: it is recommended that the primary bag on the mixer lies with the tubing on the lower side of the mixer holding plate.
7) After approximately 50 ml of blood enters the primary bag, mix thoroughly (when possible).
Note: Collect the quantity of blood stated on the blood bag label.
Sampling Procedure
Note: this is done during the donation. Avoid air in the collection of sample tubes from the diversion pouch to ensure adequate sample volume and avoid hemolysis.
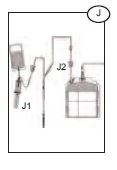
1) Hold the Bactivam pouch with the Vacuvam below it (J), to ensure good blood flow from the pouch to the sampling device, and no introduction of air into the sampling tubes.
2) Remove the cap of the Vacuvam sampling device, and retain for later use (when possible).
3) Introduce a vacuum sampling tube by holding the barrel of the Vacuvam and directing the sample tube cap directly onto the needle inside the device (J1).
4) Blood is drawn automatically into the sampling tube, stopping at the appropriate volume.
5) When the sample tube stops filling, withdraw the tube, ensuring that the tube is pulled out of the barrel in a straight line.
6) Repeat the sampling procedure steps (3 to 5) with all remaining sampling tubes.
7) When all samples have been taken, replace the Vacuvam cap to prevent accidental access to the interior needle (when possible).
8) Mix the blood in the primary bag regularly (approximately every 100 ml or every minute).
After Donation
1) Once the required volume of blood has been collected, close the red clamp on the donor line (J2), at least 12 inches above the primary bag.
Note: if necessary, place a Hemostat on the collection line just below the Secuvam.
2) Release the pressure cuff or tourniquet, and immediately mix the whole blood in the primary bag.
Note: if the needle was taped to the arm, and not the Secuvam, remove the tape from the needle before pulling the needle into the Secuvam (disregard step 3).
3) Remove the tape holding the Secuvam in place.
4) Remove the needle by gently pulling the needle into the barrel of the Secuvam while supporting the Secuvam with one hand (a click may be heard).
Note: if necessary, step 4 may be done before step 3.
5) Remove and discard the cap (if relevant) from the barrel of the Vacuvam, into an appropriate container.
6) Insert the Secuvam needle protector into the barrel of the Vacuvam sampling device.
7) After removing the blood from the mixer/trip scale, mix the blood unit thoroughly.
8) If the donor line is to be used for quality control, strip the line as per procedure (at least 3 times), heat-seal (place clips) and cut (between the clips), leaving sufficient line for testing purposes.
Note: if it is necessary to strip the donor line, it is recommended to strip within 4 minutes of the end of the donation, in order to avoid introducing clots into the blood bag.
9) Ideally, heat-seal the donor line as close as possible to the pack (~1 inch). Alternatively, place clips close to the blood pack and cut between the clips. Mix thoroughly during and immediately after the procedure.
Note: this is an in-line filter system; there is no need to keep the donor line for cross-matching segments.
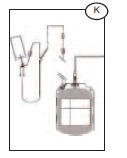
10) Remove and discard the donor line (K) with the two red clamps, Bactivam, Vacuvam and Secuvam as per SOP.
Note: the completely protected needle after securing the Secuvam inside the rigid Vacuvam sleeve can be disposed of in general biowaste vs. a sharps container
COMPONENT PREPARATION GENERAL
1 to 6 C Filtration: Ensure that the whole blood to be filtered at cold temperature has reached 1 to 6 C and has been stored between 4 and 8 hours at 1 to 6 C. The height of the unit for filtration should be 72 to 76 inches.
Ambient (20 to 24 C) Filtration: Whole blood filtration may be initiated at room temperature between 4 and 7 hours after collection. Filtrations performed at ambient temperatures should be completed within eight hours of collection. The height of the unit for filtration should be 55 to 60 inches.
CAUTION:
· Do not fold or manhandle the filters (L).
· Do not apply pressure, either mechanical or human, to increase the flow rate of the filter.
· Units with decreased or extended filtration times may not be adequately leukoreduced, and should be evaluated for residual leukocytes and red blood cell recovery before release. Consult training materials for more information on expected length of filtration times and the applicable hold periods in clinical study data.
· Failure to achieve and maintain a closed system throughout processing results in a product that must be stored at 1 to 6 C and transfused within 24 hours.
· Fresh Frozen Plasma should be prepared between 4 and 8 hours of the whole blood donation.
· Fresh Frozen Plasma should be stored at minus 18 C or colder.
· The AS-1 additive solution must be added to the red blood cells without delay after the separation of the plasma. Whole blood in CPD alone may be stored at 1 to 6 C for up to 21 days.
· The filter has been designed and validated to meet the criteria of a leukoreduction filter for whole blood. The filter's performance must be validated using your facility's standard operating procedure (SOP) on component preparation and handling. Any filtration that fails to meet the filtration specifications established by the facility's validation should be evaluated for residual leukocytes and red blood cell (RBC) recovery after filtration.
Note that unusual attributes such as sickle cell trait, cold agglutinins, blood clotting and gel formation, and poor mixing during collection and processing of components can contribute to the failure of a filtration to meet filtration specifications. Faiure to adequately re-suspend RBC by mixing the whole blood unit thoroughly prior to filtration can result in blocked filters.
WHOLE BLOOD LEUKOREDUCTION
1) Mix the blood well.
2) Suspend the blood bag at a height of 55 to 60 inches if filtering at room temperature (20 to 24 C) or 72 to 76 inches if filtering at cold temperature (1 to 6 C).
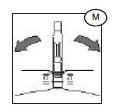
3) Break the break-away cannula (M) on the blood bag leading to the Leucoflex MTL1 filter, by bending the cannula with a side-to-side motion.
4) Allow the blood to flow by gravity through the Leucoflex MTL1 filter into the transfer bag.
Note: Determine the status of the filtation as follows: If there is no blood flowing from below the filter, the filter is flat and the filter media is visible on the inlet side, filtration is complete.
5) Ensure there is a temporary clamp between the Leucoflex MTL1 filter and the leukoreduced blood.
6) If required, purge the air from the leukoreduced blood back into the flexible Leucoflex MTL1 filter (the filter will expand), filling the segment tubing with blood.
7) Close the temporary clamp before separating the blood and attached satellites.
8) Heat seal the segment tubing just below the Leucoflex MTL1 filter, and all along the segment tubing between the printed numbers.
Note: Do not strip the tubing below the leukocyte reduction filter. Such stripping may pull leukocytes through the filter.
9) Heat seal and discard the empty collection bag, Leucoflex MTL1 filter, temporary clamp and tubing as per SOP.
Note: Ensure that the temporary clamp is not left on the segment tubing.
10) Centrifuge the blood, satellite bag, and additive solution bag together under conditions designed to produce a separation of plasma and red blood cells.
11) Place the centrifuged blood carefully into a plasma extractor, and gently release the pressure plate.
12) Break the break-away cannula on the blood bag by bending the cannula with a side-to-side motion.
13) Express Plasma into the empty transfer bag. If Cryoprecipitate AHF (Anti- Hemophilic Factor) will be manufactured from this Plasma, put a temporary clamp between the "Y" connector (N) and the plasma bag; otherwise, add a permanent clamp.
14) Break the break-away cannula on the additive solution bag by bending the cannula with a side-to-side motion.
15) Suspend the additive solution (AS-1) bag above the RBC bag so that the additive solution drains completely into the RBC without delay.
Note: The additive solution must be mixed thoroughly with the AS-1 Red Blood Cells, Leukocytes Reduced.
16) Heat seal and disconnect the Plasma, Leukocytes Reduced (and any attached transfer bags) as per SOP. Quality control testing should be performed monthly to demonstrate that the WBC content of the plasma is <5 x 10 6 per container if the Plasma or Cryoprecipitated AHF will be labeled as Leukocytes Reduced.
17) Store the AS-1 Red Blood Cells, Leukocytes Reduced at 1-6 C for up to 42 days.
18) Should Cryoprecipitated AHF be manufactured, process the plasma bag and attached empty transfer bag as per SOP.
Clinical Studies Holding Time, Residual WBC and Percent RBC Recovery
WARNING: If unit is spiked for any reason, storage is limited to 24 hours at 1 to 6 C before transfusion.
|
Filtration Temperature | n |
Holding Time (Hrs) Mean (Range) |
Residual WBC (x 10 6/ unit) Mean +/-SD |
Percentage RBC Recovery Mean +/-SD |
| 20 to 24 C | 61 | 5.1 (4.1 - 6.7) | 0.417 +/-0.533 | 91.1 +/-3.0 |
| 1 to 6 C | 30 | 5.1 (4.6 - 6.1) | 0.197 +/-0.131 | 89.9 +/-1.9 |
NOTE: If either face of the filter remains white, the Whole Blood Unit has not been leucoreduced and must be discarded.
| MTL1 LEUCOFLEX
anticoagulant citrate phosphate dextrose solution with as-1 kit |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Maco Productions (265492868) |
| Registrant - Maco Productions (265492868) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Maco Productions | 265492868 | manufacture(14498-001) | |