InterSol: Package Insert / Prescribing Info
Package insert / product label
Generic name: sodium chloride, sodium acetate, trisodium citrate dihydrate, sodium phosphate, monobasic, monohydrate and sodium phosphate, dibasic
Dosage form: intravenous solution
On This Page
6B7800S
12x 800 mL
Rx only
Store at Controlled Room Temperature. Protect from freezing. Avoid excessive heat. Definition of "Controlled Room Temperature":
"A temperature maintained thermostatically that encompasses the usual and customary working environment of 20° to 25°C (68° to 77°F); that results in a mean kinetic temperature calculated to be not more than 25°C; and that allows for excursions between 15°C and 30°C (59° and 86°F) that are experienced in pharmacies, hospitals, and warehouses. Provided the mean kinetic temperature remains in the allowed range, transient spikes up to 40°C are permitted as long as they do not exceed 24 hours ... The mean kinetic temperature is a calculated value that may be used as an isothermal storage temperature that simulates the non isothermal effects of storage temperature variations."
Reference: United States Pharmacopeia, General Notices. United States Pharmacopeial Convention, Inc. 12601 Twinbrook Parkway, Rockville, MD.
Dispose of waste in appropriate biohazard container or according to local regulatory requirements.
Indications and Usage:
InterSol solution is an isotonic solution designed to replace a proportion of the plasma used in the storage of leukoreduced apheresis platelets under standard blood banking conditions. There is no direct therapeutic effect to be expected from the formulation. The solution should never be infused directly into a patient.
InterSol platelets are leukocyte-reduced apheresis platelet concentrates that are stored in a mix of 65% InterSol and 35% plasma, nominal. InterSol platelets prepared within the ranges described in the apheresis system Operator's Manuals may be stored for up to 5 days at 20-24°C, with continuous agitation.
Dosage and Administration:
InterSol solution is to be used with the AMICUS Separator System or the TRIMA ACCEL system. For full instructions on the use of InterSol solution, see the Operator's Manual for the respective platelet collection system.
Dosage Forms and Strengths:
InterSol solution is provided as a 800 mL sterile and non-pyrogenic solution in a non-PVC plastic container with a sterile and non-pyrogenic fluid path. Each 100 mL contains 305 mg Dibasic Sodium Phosphate, Anhydrous, USP; 93 mg Monobasic Sodium Phosphate, Monohydrate, USP; 318 mg Sodium Citrate, Dihydrate, USP; 442 mg Sodium Acetate, Trihydrate, USP; 452 mg Sodium Chloride, USP; Water for Injection, USP quantity sufficient.
Contraindications:
InterSol solution is added to apheresis-derived leukoreduced platelet concentrates after the apheresis procedure is complete. It is not for direct intravenous infusion. There are no known contraindications associated with the use of InterSol solution for the preparation of InterSol platelets.
Warnings and Precautions:
- •
- InterSol solution is NOT FOR DIRECT INTRAVENOUS INFUSION.
- •
- Do not use if particulate matter is present or if the solution is cloudy.
- •
- Do not use if the container is damaged, leaking or if there is any visible sign of deterioration.
- •
- Do not vent.
- •
- Do not reuse. Discard unused or partially used InterSol solution.
- •
- Protect from sharp objects.
- •
- Verify that the InterSol solution has been securely attached to the PAS Connector line to avoid disconnection and leaks.
Adverse Reactions:
InterSol solution is added to leukoreduced platelet concentrates after the apheresis procedure is complete. It is not for direct intravenous infusion. InterSol solution is not expected to cause adverse events other than those normally associated with platelet transfusion.
Drug Abuse / Dependence:
InterSol solution is used as a storage solution for platelet concentrates and has no pharmacological effect.
Overdosage:
InterSol solution is used as a storage solution for platelet concentrates and it is not for direct intravenous infusion.
Description:
InterSol solution is an isotonic solution designed to replace a proportion of the plasma used in the storage of platelets. The solution contains constituents that are naturally occurring components present in many cellular systems: sodium acetate as a nutrient, sodium citrate to prevent platelet clumping and activation, sodium phosphate for buffering and sodium chloride for osmolarity. InterSol solution does not have a pharmacological effect in vivo, but rather acts to provide the appropriate environment and nutrients in lieu of a portion of the plasma normally used for the storage of platelets.
Clinical Pharmacology:
InterSol solution is used as a storage solution for platelet concentrates and it is not for direct intravenous infusion. This solution has no pharmacological effect.
Clinical Studies:
In Vitro Biochemical and Functional Evaluations AMICUS-derived, leukoreduced platelets
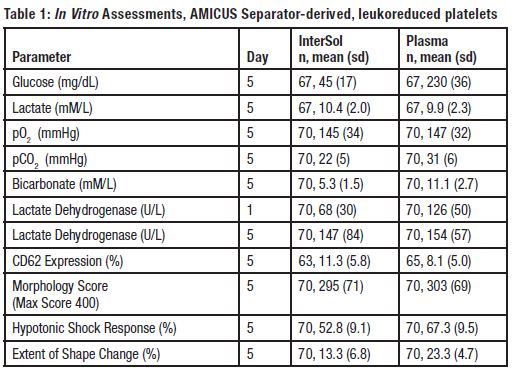
InterSol platelet concentrates (n=70) prepared using the AMICUS separator and stored for 5 days showed a median and mean pH value on Day 5 of 7.2 and 7.2 ± 0.1 (range: 6.9-7.5), respectively, with a lower non-parametric 95%/95% tolerance limit of 6.9.
Supplemental in vitro assessments of InterSol platelets and AMICUS-derived leukoreduced platelets stored in 100% plasma are presented in Table 1.
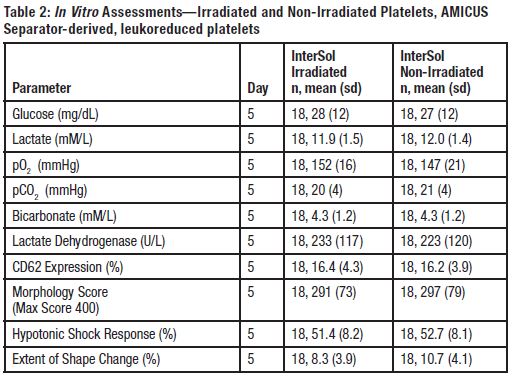
InterSol platelets irradiated (n=18) at either 2500 or 2800 cGray (dependent on site procedures) were compared to non-irradiated InterSol platelets. At the end of storage on Day 5 the platelet yields of the irradiated products were concentrated between 2.5 x 1011 to 3.5 x 1011 and included one product with a yield less than2.5 x 1011 and two products with yields greater than 3.5 x 1011. In vitro testing is not predictive of in vivo performance, which was not evaluated. Summary statistics are presented in Table 2.
In Vivo Recovery and Survival in Healthy Subjects AMICUS-derived, leukoreduced platelets
In vivo evaluation of InterSol platelets at Day 5 (n=33) compared to a fresh platelet control resulted in a mean percent recovery of 46.4 ± 11.9 percent and 58.0 ± 10.7 percent and mean survival of 5.7 ± 1.4 days and 8.0 ± 1.4 days, respectively. The in vivo data collected were used to calculate the upper limit of a two-sided 95% confidence interval of the mean percent recovery of the algebraic expression (0.66 x Fresh – 5 Day), and of the mean percent survival (days) of the algebraic expression (0.58 x Fresh – 5 Day). The upper bound of the two-sided 95% confidence intervals for recovery and survival on Day 5 was –4.6 and –0.6 respectively and met the requirement of less than 0.
Post-Market Transfusion-Related Adverse Events (AE) Study AMICUS-derived, leukoreduced platelets
An open label, non-randomized, retrospective medical record review study was performed in 6 centers to demonstrate that the overall rate of transfusion-related AEs in patients receiving InterSol Platelet transfusions was not more than double the rate in patients receiving apheresis platelets stored in 100% plasma (Plasma Platelets). The study categorized adverse transfusion reactions according to the definitions outlined by the Biovigilance Component of the National Healthcare Safety Network (NHSN) System. All sites that participated were required to have systems in place to observe transfusion-related AEs and to record when no transfusion-related AE was observed. An independent Clinical Events Committee (CEC) blinded to platelet type and study site adjudicated the reported events.
Patients were prescribed platelet transfusions per each site’s standard practice. The type of platelet unit the patient received was based on the site’s inventory at the time the transfusion was ordered. Site personnel observed the platelet transfusion recipient following their standard procedures. Signs and symptoms of a potential transfusion-related reaction were noted using existing reporting systems. A total of 14,005 transfusions from 6 study sites were included in the final analysis. A total of 4,160 InterSol Platelet transfusions were given to a total of 1,444 patients, and 9,845 Plasma Platelet transfusions were given to 2,202 patients. There were 165 CEC-adjudicated adverse reactions reported. Of those, 23 events were associated with InterSol Platelets and 142 events were associated with Plasma Platelets.
Overall, 1.13% of all transfusions resulted in an AE. The percentage of InterSol Platelet transfusions which led to AEs was 0.55%, while 1.37% of Plasma Platelet transfusions resulted in AEs. The 97.5% upper confidence limit for the relative risk of transfusion-related AE associated with InterSol Platelets relative to Plasma Platelets was 0.66, indicating that the study objective of ruling out a doubling of transfusion-related AEs for InterSol vs. Plasma Platelets was met.
The majority of the 165 CEC-adjudicated reactions were Allergic (n=93) or Febrile Non-Hemolytic Transfusions Reactions (FNHTR, n=56), at 0.66% and 0.40% of total transfusions, respectively. There were allergic reactions associated with 0.29% of InterSol Platelet transfusions and 0.82% of Plasma Platelet transfusions. FNHTR events were associated with 0.17% of InterSol Platelet transfusions and 0.50% of Plasma Platelet transfusions.
No InterSol Platelet transfusion was associated with more than one reaction. Seven (7) Plasma Platelet transfusions (0.07% of total transfusions) were associated with two (2) separate adverse reactions each. Two (2) InterSol Platelet and five (5) Plasma Platelet adverse reactions were classified as severe. All other reactions were classified as non-severe. All adverse reactions were reported with an outcome of “Minor or No Sequelae”.
No adverse safety trends were identified after review of all the AEs by the independent Clinical Events Committee.
In Vitro Biochemical and Functional Evaluations, TRIMA ACCEL-derived, leukoreduced platelets
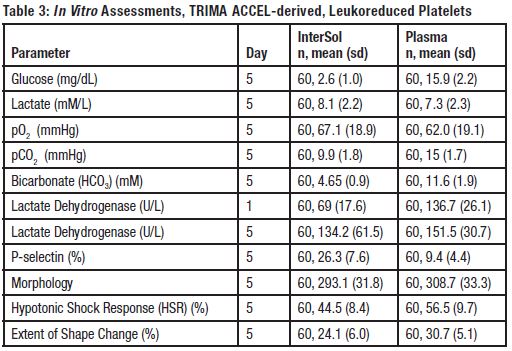
InterSol platelet concentrates (n = 60) prepared using the Trima Apheresis system and stored for 5 days showed a mean pH value on Day 5 of 7.2 ± 0.1 (range 7.0 – 7.3), with a lower one-sided 95% confidence interval of 0.951.
Supplemental in vitro assessments of TRIMA ACCEL-derived leukoreduced platelets stored in InterSol platelet and 100% plasma are presented in Table 3.
In Vivo Recovery and Survival in Healthy Subjects, TRIMA ACCEL-derived, leukoreduced platelets
In vivo evaluations of TRIMA ACCEL-derived platelets stored in InterSol at Day 5 (n = 24) compared to fresh platelet control resulted in a mean percent recovery of 45.2 ± 12.3 percent and 56.0 ± 13.2 percent and a mean survival of 5.4 ± 1.0 days and 7.9 ± 1.5 days, respectively. The in vivo data collected were used to calculate the upper limit of a one-sided 97.5% confidence interval of the mean percent recovery of the algebraic expression of (0.66 x Fresh – 5 Day) and the mean percent survival (days) of the algebraic expression (0.58 x Fresh – 5 Day). The lower limit of the one-sided 97.5% confidence intervals for recovery and survival on Day 5 was 4.0 and 0.4 respectively and met the requirement of greater than 0.
How Supplied/Storage and Handling:
800 mL sterile solution in a non-PVC plastic container with a sterile, non-pyrogenic fluid path. The InterSol solution container is supplied in a vented plastic overwrap covering that serves as a dust cover for the container. The dust cover is not a sterility barrier and is not an element that defines the expiration date of the InterSol product.


Fresenius Kabi AG
Else-Kröner-Str. 1
61352 Bad Homburg
Germany
Tel.: +49 (0) 61 72 / 686-0
www.fresenius-kabi.com
www.fresenius-kabi.com/us/symbolglossary
For US: 1-800-933-6925
All trademarks shown are property of their respective owners.
© 2025 Fresenius Kabi AG. All rights reserved.
472317814 [B] 2025/03
| INTERSOL
platelet additive 3 solution |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| Labeler - Fenwal, Inc. (794519020) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Fenwal International, Inc. | 091164590 | MANUFACTURE(0942-9605) | |