Interceptor Flavor Tabs: Package Insert / Prescribing Info
Package insert / product label
Generic name: milbemycin oxime
Dosage form: FOR ANIMAL USE ONLY
On This Page
(milbemycin oxime)
|
INFORMATION FOR DOSING DOGS |
The palatable once-a-month tablet that prevents heartworm disease, controls adult hookworm, and removes and controls adult roundworm and whipworm infections in dogs and puppies.
Caution:
Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. Keep this and all drugs out of the reach of children.
Interceptor Flavor Tabs Description
INTERCEPTOR is available in four tablet sizes in color-coded packages for oral administration to dogs and puppies. Each tablet is formulated to provide a minimum of 0.23 mg/lb (0.5 mg/kg) body weight of milbemycin oxime. Milbemycin oxime consists of the oxime derivatives of 5-didehydromilbemycins in the ratio of approximately 80% A4 (C32H45NO7, MW 555.71) and 20% A3 (C31H43NO7, MW 541.68).
| *for dogs only | |
|
Package color |
Milbemycin oxime tablet |
|
Brown |
2.3 mg* |
|
Green |
5.75 mg |
|
Yellow |
11.5 mg |
|
White |
23.0 mg |
Indications and Usage for Interceptor Flavor Tabs
INTERCEPTOR is indicated for use in the prevention of heartworm disease caused by Dirofilaria immitis, the control of adult Ancylostoma caninum (hookworm), and the removal and control of adult Toxocara canis and Toxascaris leonina (roundworms) and Trichuris vulpis (whipworm) infections in dogs and in puppies four weeks of age or greater and two pounds body weight or greater.
Interceptor Flavor Tabs Dosage and Administration
INTERCEPTOR is given orally, once a month, at the recommended minimum dosage rate of 0.23 mg milbemycin oxime per pound of body weight (0.5 mg/kg).
|
Body Weight |
INTERCEPTOR |
|
2-10 lbs. |
One tablet (2.3 mg) |
|
11-25 lbs. |
One tablet (5.75 mg) |
|
26-50 lbs. |
One tablet (11.5 mg) |
|
51-100 lbs. |
One tablet (23.0 mg) |
Dogs over 100 lbs. are provided the appropriate combination of tablets.
Interceptor Flavor Tabs Dosage and Administration
INTERCEPTOR is palatable and most dogs will consume the tablet willingly when offered by the owner. As an alternative, the dual-purpose tablet may be offered in food or administered as other tablet medications. Watch the dog closely following dosing to be sure the entire dose has been consumed. If it is not entirely consumed, redose once with the full recommended dose as soon as possible.
INTERCEPTOR must be administered monthly, preferably on the same date each month. The first dose should be administered within one month of the dog's first exposure to mosquitoes and monthly thereafter until the end of the mosquito season. If a dose is missed and a 30-day interval between dosing is exceeded, administer INTERCEPTOR immediately and resume the monthly dosing schedule.
If INTERCEPTOR replaces diethylcarbamazine (DEC) for heartworm prevention, the first dose must be given within 30 days after the last dose of DEC.
Palatability:
Palatability trials conducted in 244 dogs from 10 different U.S. veterinary practices demonstrated that INTERCEPTOR was willingly accepted from the owner by over 95% of dogs. The trial was comprised of dogs representing 60 different breeds and both sexes, with weights ranging from 2.1 lbs. to 143.3 lbs., and ages ranging from 8 weeks to 15 years.
Precautions
Do not use in puppies less than four weeks of age or less than two pounds of body weight. Prior to initiation of the INTERCEPTOR treatment program, dogs should be tested for existing heartworm infections. Infected dogs should be treated to remove adult heartworms and microfilariae prior to initiating treatment with INTERCEPTOR. Mild, transient hypersensitivity reactions manifested as labored respiration, vomiting, salivation and lethargy, have been noted in some treated dogs carrying a high number of circulating microfilariae. These reactions are presumably caused by release of protein from dead or dying microfilariae.
Adverse Reactions/Side Effects
The following adverse reactions have been reported following the use of INTERCEPTOR: Depression/lethargy, vomiting, ataxia, anorexia, diarrhea, convulsions, weakness and hypersalivation.
Efficacy:
INTERCEPTOR eliminates the tissue stage of heartworm larvae and the adult stage of hookworm (Ancylostoma caninum), roundworms (Toxocara canis, Toxascaris leonina) and whipworm (Trichuris vulpis) infestations when administered orally according to the recommended dosage schedule.The anthelmintic activity of milbemycin oxime is believed to be a result of interference with invertebrate neurotransmission.
Safety:
Milbemycin oxime has been tested safely in over 75 different breeds of dogs, including collies, pregnant females, breeding males and females, and puppies over two weeks of age. In well-controlled clinical field studies, 786 dogs completed treatment with milbemycin oxime. Milbemycin oxime was used safely in animals receiving frequently used veterinary products such as vaccines, anthelmintics, antibiotics, steroids, flea collars, shampoos and dips.
Two studies in heartworm-infected dogs were conducted which demonstrated mild, transient hypersensitivity reactions in treated dogs with high microfilaremia counts (see Precautions for reactions observed). Safety studies in pregnant dogs demonstrated that high doses (1.5 mg/kg =3X) of milbemycin oxime given in an exaggerated dosing regimen (daily from mating through weaning), resulted in measurable concentrations of the drug in milk. Puppies nursing these females which received exaggerated dosing regimens demonstrated milbemycin-related effects. These effects were directly attributable to the exaggerated experimental dosing regimen. The product is normally intended for once-a-month administration only. Subsequent studies included using 3X daily from mating to one week before weaning and demonstrated no effects on the pregnant females or their litters. A second study where pregnant females were dosed once at 3X the monthly use rate either before, on the day of or shortly after whelping resulted in no effects on the puppies.
Some nursing puppies, at 2, 4, and 6 weeks of age, given greatly exaggerated oral milbemycin oxime doses (9.6 mg/kg = 19X) exhibited signs typified by tremors, vocalization and ataxia. These effects were all transient and puppies returned to normal within 24 to 48 hours. No effects were observed in puppies given the recommended dose of milbemycin oxime (0.5 mg/kg). This product has not been tested in dogs less than 1 kg weight.
A rising-dose safety study conducted in rough-coated collies, manifested a clinical reaction consisting of ataxia, pyrexia and periodic recumbency, in one of fourteen dogs treated with milbemycin oxime at 12.5 mg/kg (25X monthly use rate). Prior to receiving the 12.5 mg/kg dose (25X monthly use rate) on day 56 of the study, all animals had undergone an exaggerated dosing regimen consisting of 2.5 mg/kg milbemycin oxime (5X monthly use rate) on day 0, followed by 5.0 mg/kg (10X monthly use rate) on day 14 and 10.0 mg/kg (20X monthly use rate) on day 32. No adverse reactions were observed in any of the collies treated with this regimen up through the 10.0 mg/kg (20X monthly use rate) dose.
How is Interceptor Flavor Tabs supplied
INTERCEPTOR is available in four tablet sizes (see Dosage section), formulated according to the weight of the dog. Each tablet size is available in color-coded packages of 6 or 12 tablets each, which are packaged 10 per display carton.
Storage and Handling
INTERCEPTOR should be stored at room temperature, between 59° and 77°F (15-25°C).
Interceptor™
(milbemycin oxime)
|
INFORMATION FOR DOSING CATS |
The palatable once-a-month tablet that prevents heartworm disease and removes adult roundworms and hookworms in cats and kittens.
Caution:
Federal (USA) law restricts this drug to use by or on the order of a licensed veterinarian. Keep this and all drugs out of the reach of children.
Interceptor Flavor Tabs Description
INTERCEPTOR for Cats is available in three tablet sizes in color-coded packages for oral administration to cats and kittens. Each tablet is formulated to provide a minimum of 0.9 mg/lb (2.0 mg/kg) body weight of milbemycin oxime. Milbemycin oxime consists of the oxime derivatives of 5-didehydromilbemycins in the ratio of approximately 80% A4 (C32H45NO7, MW 555.71) and 20% A3 (C31H43NO7, MW 541.68).
Indications and Usage for Interceptor Flavor Tabs
INTERCEPTOR for Cats is indicated for use in the prevention of heartworm disease caused by Dirofilaria immitis, and the removal of adult Ancylostoma tubaeforme (hookworm) and Toxocara cati (roundworm) in cats and kittens six weeks of age or greater and 1.5 lbs. body weight or greater.
Dosage Forms and Strengths
INTERCEPTOR for Cats is given orally, once a month, at the recommended minimum dosage rate of 0.9 mg milbemycin oxime per pound of body weight (2.0 mg/kg).
|
Body Weight |
INTERCEPTOR |
|
1.5 to 6 lbs. |
One tablet (5.75 mg) |
|
6.1-12 lbs. |
One tablet (11.5 mg) |
|
12.1-25 lbs. |
One tablet (23.0 mg) |
Cats over 25 lbs. are provided the appropriate combination of tablets.
Interceptor Flavor Tabs Dosage and Administration
INTERCEPTOR for Cats is palatable and may be offered by the owner as a treat. As an alternative, the tablet may be offered in food or administered as other tablet medications. The tablets can be broken for ease of administration. Watch the cat closely following dosing to be sure the entire dose has been consumed. If it is not entirely consumed, redose once with the full recommended dose as soon as possible.
INTERCEPTOR for Cats must be administered monthly, preferably on the same date each month. The first dose should be administered within one month of the cat's first exposure to mosquitoes and monthly thereafter until the end of the mosquito season. If a dose is missed and a 30-day interval between dosing is exceeded, administer INTERCEPTOR for Cats immediately and resume the monthly dosing schedule. It is recommended that cats be tested for existing heartworm infection prior to starting treatment with INTERCEPTOR for Cats (see Precautions).
Palatability:
Palatability trials conducted in 72 cats demonstrated that cats were successfully dosed with INTERCEPTOR for Cats by the owner when they either offered the tablet as a treat, placed the tablet in the cat's mouth or placed the tablet in the cat's food in 72% of cats. About 16% of the cats were dosed manually and 13% of the cats were not successfully dosed according to the protocol.
Precautions
Do not use in kittens less than six weeks of age or less than 1.5 lbs. body weight. Safety in heartworm-positive cats has not been established. Safety in breeding, pregnant, and lactating queens and breeding toms has not been established.
Efficacy:
INTERCEPTOR for Cats eliminate the tissue stage of heartworm larvae and hookworm (Ancylostoma tubaeforme) and roundworm (Toxocara cati) infections when administered orally according to the recommended dosage schedule.The anthelmintic activity of milbemycin oxime is believed to be a result of interference with invertebrate neuro- transmission.
Safety:
Milbemycin oxime has been tested safely in over 8 different breeds of cats. In well-controlled clinical field studies 141 cats completed treatment with milbemycin oxime. Milbemycin oxime was used safely in animals receiving frequently used veterinary products such as vaccines, anthelmintics, anesthetics, antibiotics, steroids, flea collars, shampoos and dips.
Safety studies were conducted in young cats and kittens and doses of 1X, 3X and 5X the minimum recommended dose of 2.0 mg/kg demonstrated no drug-related effects. Tolerability studies at exaggerated doses of 1OX also demonstrated no drug-related adverse effects in kittens and young adult cats.
How is Interceptor Flavor Tabs supplied
INTERCEPTOR for Cats is available in three tablet sizes (see Dosage section), formulated according to the weight of the cat. Each tablet size is available in color-coded packages of 6 or 12 tablets each, which are packaged 10 per display carton.
Storage and Handling
INTERCEPTOR for Cats should be stored at room temperature, between 59° and 77°F (15-25°C).
Manufactured for: Elanco US Inc.
Greenfield, IN 46140, USA
Approved by FDA under NADA # 140-915
Interceptor, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
© 2021 Elanco or its affiliates
Revision date: February 2021
Elanco™
PA103156X
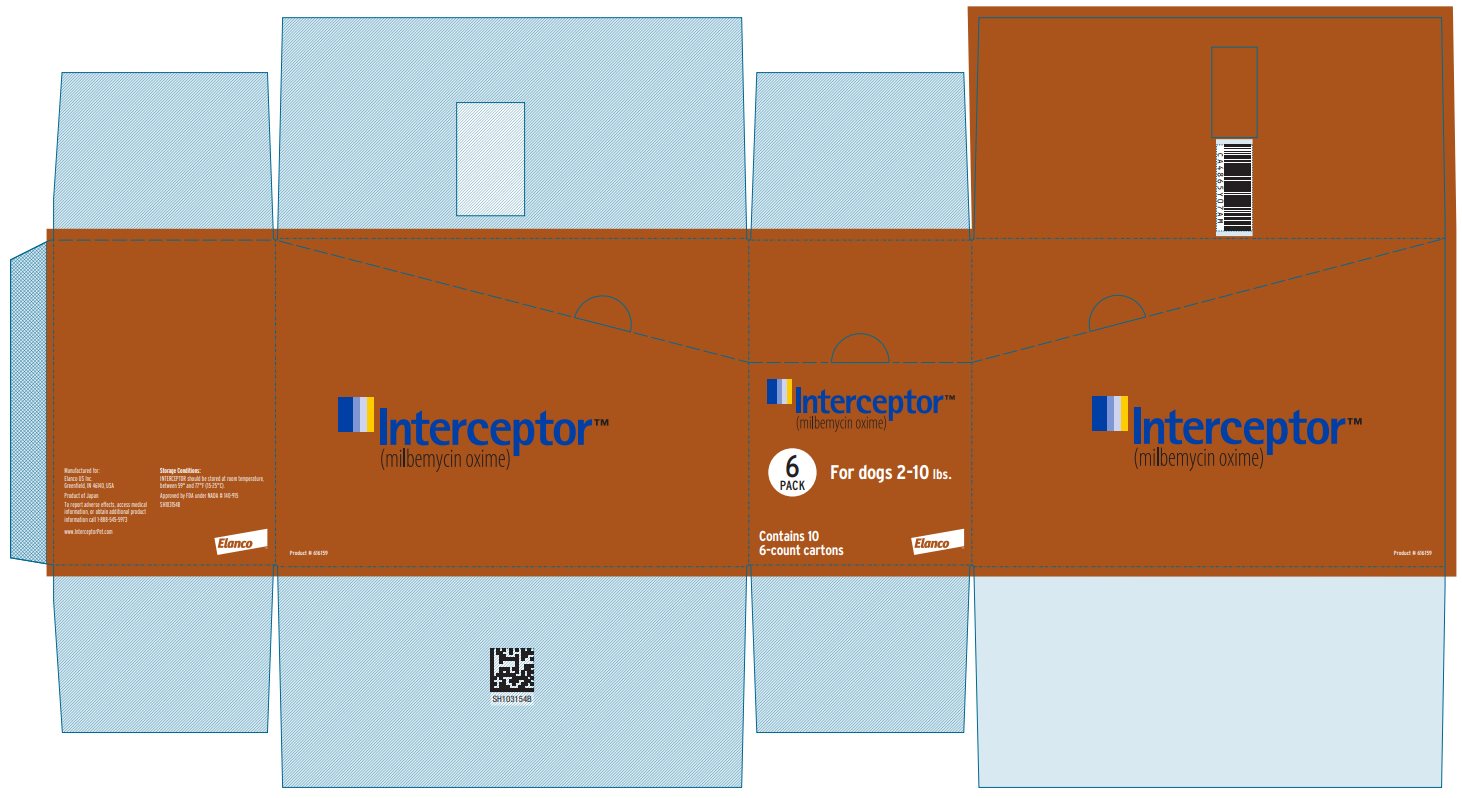
Principal Display Panel - Interceptor 2.3 mg Box Label
Interceptor™
(milbemycin oxime)
6
PACK
For dogs 2-10 lbs.
Contains 10
6-count cartons
Elanco™
Principal Display Panel - Interceptor 2.3 mg Carton Label
Interceptor™
(milbemycin oxime)
BROAD SPECTRUM
PARASITICIDE
Contains once-a-month flavored
tablets to prevent heartworm disease,
control adult hookworm infection,
and remove and control adult
roundworms and whipworms in
dogs and puppies.
6
PACK
For dogs 2-10 lbs.
Net contents: 6 tablets. 2.3 mg each.
Product #616159
Approved by FDA under NADA # 140-915
Keep this and all drugs out of the reach of children.
Caution: Federal (USA) law restricts this drug to use
by or on the order of a licensed veterinarian.
Elanco™
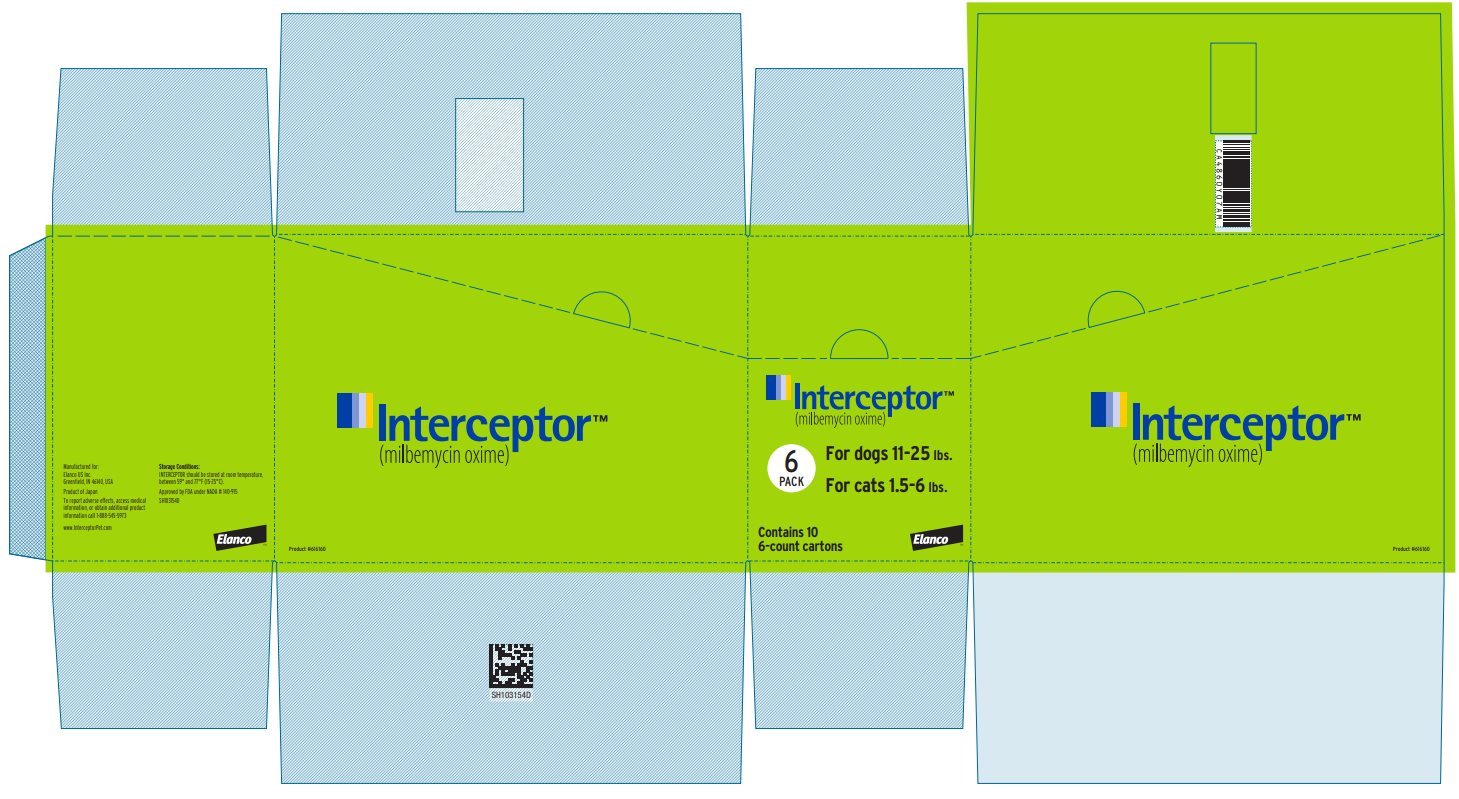
Principal Display Panel - Interceptor 5.75 mg Box Label
Interceptor™
(milbemycin oxime)
6
PACK
For dogs 11-25 lbs.
For cats 1.5-6 lbs.
Contains 10
6-count cartons
Elanco™
Principal Display Panel - Interceptor 5.75 mg Carton Label
Interceptor™
(milbemycin oxime)
BROAD SPECTRUM
PARASITICIDE
Contains once-a-month flavored
tablets to prevent heartworm disease,
control adult hookworm infection,
and remove and control adult
roundworms and whipworms in
dogs and puppies, and to prevent
heartworm disease and remove
adult hookworms and
roundworms in cats
and kittens.
6
PACK
For dogs 11-25 lbs.
For cats 1.5-6 lbs
Net contents: 6 tablets. 5.75 mg each.
Product #616160
Approved by FDA under NADA # 140-915
Keep this and all drugs out of the reach of children.
Caution: Federal (USA) law restricts this drug to use
by or on the order of a licensed veterinarian.
Elanco™
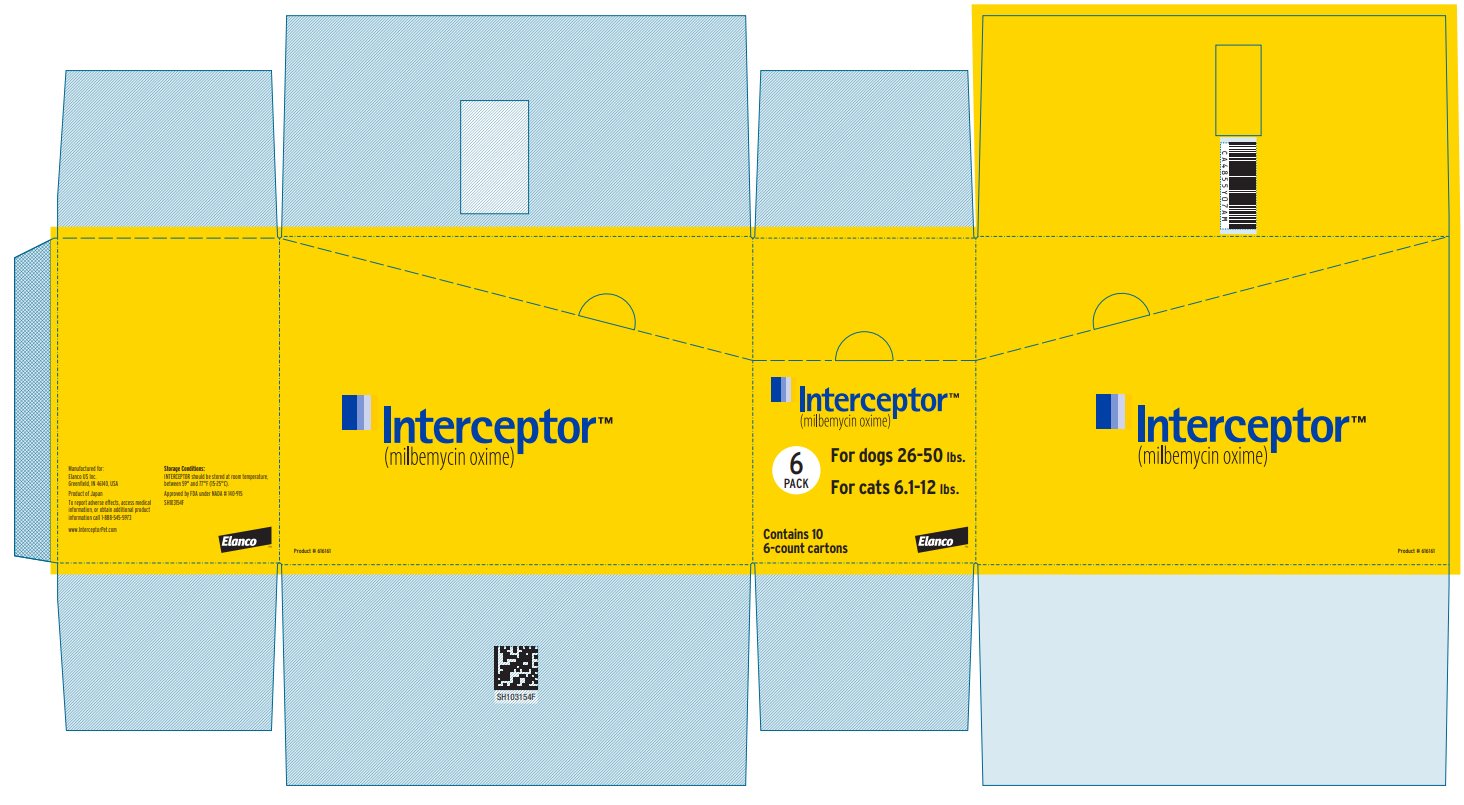
Principal Display Panel - Interceptor 11.5 mg Box Label
Interceptor™
(milbemycin oxime)
6
PACK
For dogs 26-50 lbs.
For cats 6.1-12 lbs.
Contains 10
6-count cartons
Elanco™
Principal Display Panel - Interceptor 11.5 mg Carton Label
Interceptor™
(milbemycin oxime)
BROAD SPECTRUM
PARASITICIDE
Contains once-a-month flavored
tablets to prevent heartworm disease,
control adult hookworm infection,
and remove and control adult
roundworms and whipworms in
dogs and puppies, and to prevent
heartworm disease and remove
adult hookworms and
roundworms in cats
and kittens.
6
PACK
For dogs 26-50 lbs.
For cats 6.1-12 lbs.
Net contents: 6 tablets. 11.5 mg each.
Product #616161
Approved by FDA under NADA # 140-915
Keep this and all drugs out of the reach of children.
Caution: Federal (USA) law restricts this drug to use
by or on the order of a licensed veterinarian.
Elanco™
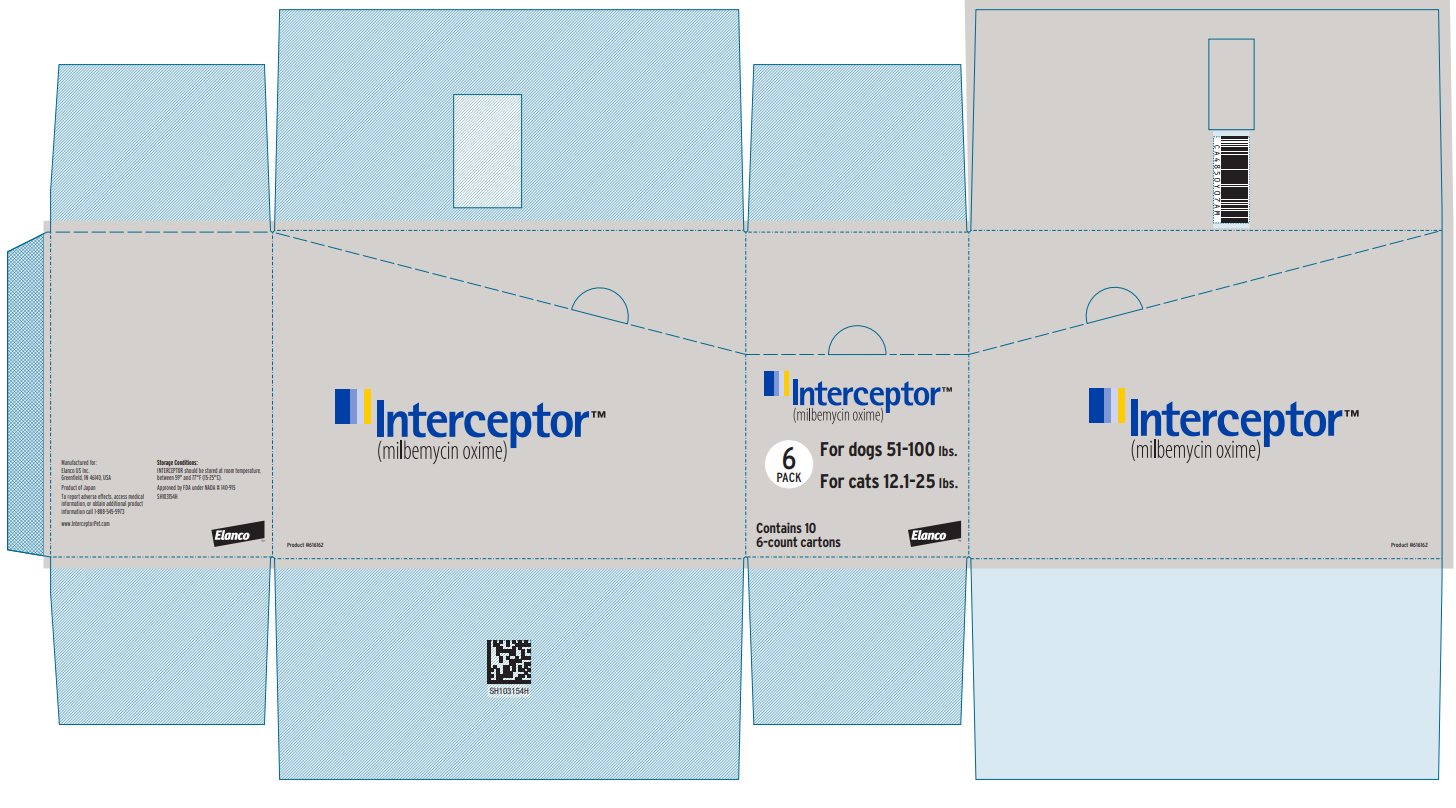
Principal Display Panel - Interceptor 23 mg Box Label
Interceptor™
(milbemycin oxime)
6
PACK
For dogs 51-100 lbs.
For cats 12.1-25 lbs.
Contains 10
6-count cartons
Elanco™
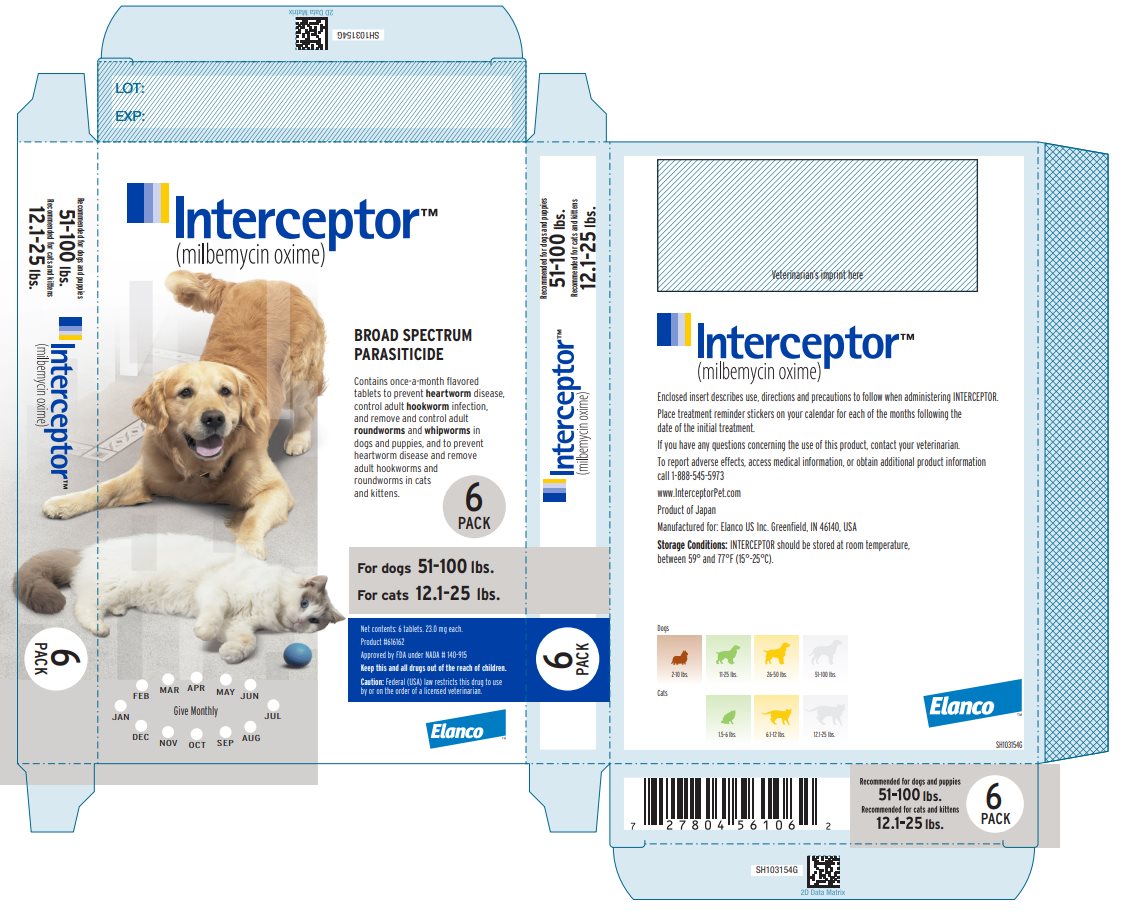
Principal Display Panel - Interceptor 23 mg Carton Label
Interceptor™
(milbemycin oxime)
BROAD SPECTRUM
PARASITICIDE
Contains once-a-month flavored
tablets to prevent heartworm disease,
control adult hookworm infection,
and remove and control adult
roundworms and whipworms in
dogs and puppies, and to prevent
heartworm disease and remove
adult hookworms and
roundworms in cats
and kittens.
6
PACK
For dogs 51-100 lbs.
For cats 12.1-25 lbs.
Net contents: 6 tablets. 23.0 mg each.
Product #616162
Approved by FDA under NADA # 140-915
Keep this and all drugs out of the reach of children.
Caution: Federal (USA) law restricts this drug to use
by or on the order of a licensed veterinarian.
Elanco™
| INTERCEPTOR FLAVOR TABS
milbemycin oxime tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| INTERCEPTOR FLAVOR TABS
milbemycin oxime tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| INTERCEPTOR FLAVOR TABS
milbemycin oxime tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| INTERCEPTOR FLAVOR TABS
milbemycin oxime tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Elanco US Inc. (966985624) |