Anticoagulant Citrate Phosphate Dextrose Adenine Solution: Package Insert / Prescribing Info
Package insert / product label
Generic name: dextrose monohydrate, trisodium citrate dihydrate, anhydrous citric acid, sodium phosphate, monobasic, monohydrate and adenine
Dosage form: solution
Fenwal Blood-Pack Units
Rx only
Using Anticoagulant Citrate Phosphate Dextrose Adenine Solution, USP (CPDA-1) and Fenwal HighFlo Needle
Contains Y Sampling Site for the collection of unanticoagulated whole blood samples for laboratory testing.
Instructions for Use
Collection Procedure:
Use aseptic technique
Note: Nominal tubing dimensions of product are 0.118" inner diameter x 0.025" wall thickness.
Note: If the Y-Sampling Site is not used, donor samples may be collected using an alternate method following standard procedures.
Precautions:
• Do not use unless the solution is clear.
• Before beginning procedure, obtain one access device for each Blood-Pack unit with Y-Sampling Site to be processed.
1. Identify Blood-Pack unit using appropriate donor identification system.
2. Adjust donor scale to desired collection weight and position primary container on the donor scale as far as possible below donor arm.
3. Clamp donor tubing between Fenwal HighFlo1 needle and primary container. This step may be performed prior to step 1 or 2.
4. Following blood center procedures, apply pressure to donor's arm and disinfect site of venipuncture.
5. Remove needle cover per instructions below:
a) Holding the hub and cover near the tamper-evident seal, twist cover and hub in opposite directions to break seal.
b) Remove needle cover, being careful not to drag the cover across the needle point.
6. Following blood center procedures, perform venipuncture, appropriately secure donor needle and/or tubing and release clamp on donor tubing.
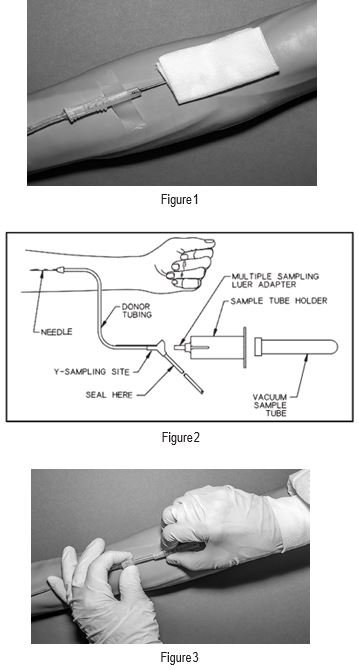
7. When good blood flow is established, stabilize the front of the needle guard to arm with tape. (see Figure 1)
8. Mix blood and anticoagulant in primary container at several intervals during collection and immediately after collection.
9. Collect the appropriate volume based on Blood-Pack unit used.
Note: The volume of anticoagulant is sufficient for the blood collection indicated on Blood-Pack unit ± 10%.
10. Release the pressure on the donor's arm as appropriate.
Precaution: Do not proceed with the remaining steps until the entire whole blood unit is collected.
11. To avoid possible contamination of the whole blood unit, before filling whole blood sample tubes, hermetically seal the donor tubing near the
Y-Sampling Site on the side leading to the primary container using
a metal clip or appropriate alternate method.
Precaution: Complete steps 12 - 20 within approximately 4 minutes after sealing the donor tubing to avoid possible clot formation in the tubing.
12. To collect samples, insert the access device by pushing firmly into the Y-Sampling Site until the membrane seal is penetrated (see Figure 2).
Note: If the access device is assembled such that the outer barrel is screwed onto the Luer, make sure to rotate clockwise upon insertion to avoid barrel detaching from Luer.
13. Open the cap on the access device (if applicable).
14. Directly align the vacuum sample tube with the internal needle in the access device. Insert vacuum sample tube into device until the stopper is punctured.
15. Allow vacuum sample tube to fill with blood then remove from the access device.
16. Repeat steps 14 and 15 until the desired number of vacuum sample tubes have been filled.
Notes:
• If the access device needs to be replaced, clamp the tubing between the needle and the Y-Sampling Site. Then, grasp base of Sampling Site with one hand and pull the access device out with the other hand. Firmly insert the new access device. Remove clamp and continue sampling.
• If the access device is assembled such that the outer barrel is screwed onto the Luer, make sure to rotate clockwise upon removal to avoid barrel detaching from Luer.
• The access device can only be replaced one time.
Precaution: When replacing the access device, be careful to avoid contact with any blood droplets on the Luer or Sampling Site. Discard used access device appropriately.
17. Release remaining pressure on donor's arm.
18. If desired, apply clamp to donor tubing between needle and Y-Sampling Site.
19. Withdrawal of Needle (see Figure 3)
Precaution: The needle guard must be held stationary while the needle is withdrawn into it.
a) Place folded sterile gauze over puncture site and hold in place with finger tip without exerting pressure.
b) Hold sides of the needle guard near the front, between the index finger and thumb. Pull the hub back smoothly until the needle is completely enclosed and securely locked into the needle guard.
c) Confirm the needle is completely enclosed and securely locked into the needle guard.
20. Strip blood from donor tubing into primary container, mix and allow the tubing to refill; repeat once. Seal at X marks on donor tubing to provide numbered aliquots of anticoagulated blood for typing or crossmatching.
Note: Step 21 may be performed prior to step 20 if desired.
21. Remove and discard the Y-Sampling Site and the needle into an appropriate biohazardous waste container following established procedures.
22. Store suspended CPDA-1 Whole Blood/Red Blood Cells between 1 and 6°C.
23. Infuse CPDA-1 Whole Blood/Red Blood Cells within 35 days of collection.
Store at Controlled Room Temperature. Protect from freezing. Avoid excessive heat.
Definition of "Controlled Room Temperature":
"A temperature maintained thermostatically that encompasses the usual and customary working environment of 20° to 25°C (68° to 77°F); that results in a mean kinetic temperature calculated to be not more than 25°C; and that allows for excursions between 15°C and 30°C (59° and 86°F) that are experienced in pharmacies, hospitals, and warehouses. Provided the mean kinetic temperature remains in the allowed range, transient spikes up to 40°C are permitted as long as they do not exceed 24 hours ... The mean kinetic temperature is a calculated value that may be used as an isothermal storage temperature that simulates the non isothermal effects of storage temperature variations."
Reference: United States Pharmacopeia, General Notices. United States Pharmacopeial Convention, Inc. 12601 Twinbrook Parkway, Rockville, MD.
1 Van der Meer, P.F., & de Korte, D. “Increase of blood donation speed by optimizing the needle-to-tubing connection: an application of donation software.” Vox Sanguinis 2009, 97: 21-25
Manufacturer
Fresenius Kabi AG
61346 Bad Homburg / Germany
www.fresenius-kabi.com
1-800-933-6925
47-23-13-513 REV: A
© 2019 Fresenius Kabi AG. All rights reserved
PACKAGE/LABEL DISPLAY PANEL
Code 4R3611
12 Units
FRESENIUS KABI
Fenwal Blood-Pack Units
Single
Anticoagulant Citrate Phosphate Dextrose Adenine Solution, USP (CPDA-1)
For the Collection and Processing of 450 mL Blood
Y-Sampling Site, 16 ga. Ultra Thin Wall Fenwal HighFlo Needle
Rx only
Each unit consists of a primary container with 63 mL of CPDA-1 solution containing 2 g Dextrose (monohydrate) USP, 1.66 g Sodium Citrate (dihydrate) USP, 188 mg Citric Acid (anhydrous) USP, 140 mg Monobasic Sodium Phosphate (monohydrate) USP and 17.3 mg Adenine USP. pH may have been adjusted with sodium hydroxide.
Sterile, non-pyrogenic fluid path.
See instructions for use.
Single use only.
Store at Controlled Room Temperature (refer to direction insert). Protect from freezing. Avoid excessive heat.
- Open pouch by tearing across at notch.
- Direct handling of product surfaces prior to extended storage in the foil pouch, may result in mold growth.
- Unused units in open foil pouch may be kept up to 60 days by folding and securing open end of foil pouch to prevent possible loss of moisture, provided:
-
- •
- Units are not removed from foil pouch, or
- •
- Unused units removed from foil pouch are returned to the foil pouch within 12 hours. Units may be removed from the pouch and returned only once.
- Units removed from the foil pouch (that are not returned to the pouch within 12 hours) must be used within 4 days (96 hours). Units out of the foil pouch for longer than 96 hours must be discarded.
Manufacturer
Fresenius Kabi AG
61346 Bad Homburg / Germany
www.fresenius-kabi.com
Made in US
| CPDA-1
anticoagulant citrate phosphate dextrose adenine (cpda-1) solution solution |
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
|
|||||||||||||||||||||
| Labeler - Fenwal, Inc. (794519020) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Fenwal International, Inc. | 091164590 | MANUFACTURE(0942-9394) | |