Altafluor: Package Insert / Prescribing Info
Package insert / product label
Generic name: fluorescein sodium and benoxinate hydrochloride
Dosage form: ophthalmic solution
Drug class: Ophthalmic diagnostic agents
Medically reviewed by Drugs.com. Last updated on Dec 4, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
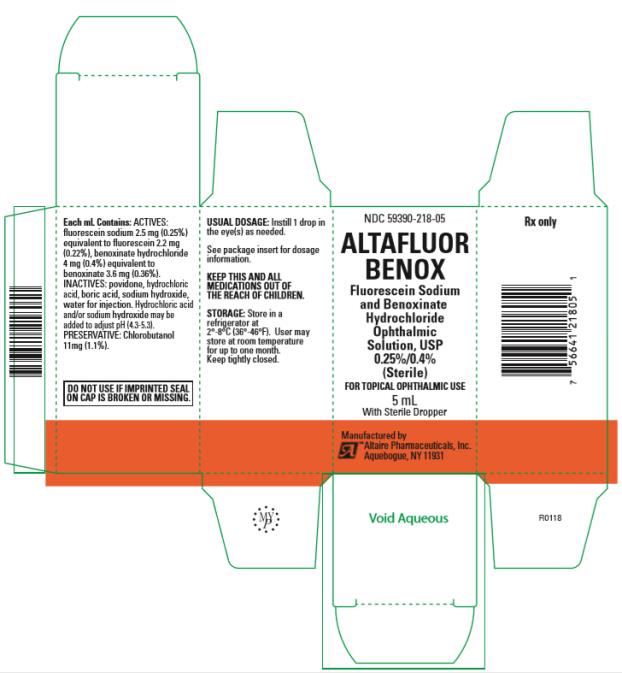
Altafluor Benox (fluorescein sodium and benoxinate hydrochloride ophthalmic solution) 0.25%/0.4% for topical ophthalmic use
Initial U.S. Approval: 2017
Indications and Usage for Altafluor
Altafluor Benox (fluorescein sodium and benoxinate hydrochloride ophthalmic solution) 0.25%/0.4% is a combination disclosing agent and local ester anesthetic indicated for procedures requiring a disclosing agent in combination with a topical ophthalmic anesthetic. (1)
Altafluor Dosage and Administration
Instill 1 to 2 drops topically in the eye as needed to achieve adequate anesthesia. (2)
Dosage Forms and Strengths
Ophthalmic solution containing fluorescein sodium 2.5 mg/mL (0.25%) and benoxinate hydrochloride 4 mg/mL (0.4%). (3)
Contraindications
Known hypersensitivity to any component of this product. (4)
Warnings and Precautions
- Corneal toxicity: Prolonged use or abuse may lead to corneal epithelial toxicity and manifest as epithelial defects which may progress to permanent corneal damage. (5.1)
- Corneal injury: Patients should not touch the eye for approximately 20 minutes after using anesthetic as accidental injuries can occur due to insensitivity of the eye. (5.2)
Adverse Reactions/Side Effects
The most common ocular adverse events are: stinging, burning and conjunctival redness. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Altaire Pharmaceuticals, Inc., at 1-800-258-2471 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2017
Full Prescribing Information
1. Indications and Usage for Altafluor
Altafluor Benox (fluorescein sodium and benoxinate hydrochloride ophthalmic solution) 0.25%/0.4% is indicated for ophthalmic procedures requiring a disclosing agent in combination with a topical ophthalmic anesthetic agent.
2. Altafluor Dosage and Administration
Instill 1 to 2 drops of Altafluor Benox in the eye as needed.
3. Dosage Forms and Strengths
Altafluor Benox is a yellow to orange-red ophthalmic solution containing fluorescein sodium 2.5 mg/mL (0.25%) and benoxinate hydrochloride 4 mg/mL (0.4%).
4. Contraindications
Altafluor Benox is contraindicated in patients with known hypersensitivity to any component of this product.
5. Warnings and Precautions
6. Adverse Reactions/Side Effects
The following ocular adverse reactions are described elsewhere in the labeling:
- Corneal Toxicity [see Warnings and Precautions (5.1)]
- Corneal Injury due to Insensitivity [see Warnings and Precautions (5.2)]
The following adverse reactions have been identified following use of fluorescein sodium and benoxinate hydrochloride ophthalmic solution 0.25% / 0.4%: ocular hyperemia, burning, stinging, eye irritation, blurred vision and punctate keratitis. Because these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
There are no available data on the use of Altafluor Benox in pregnant women to inform any drug associated risk. Adequate animal reproduction studies have not been conducted with fluorescein sodium and/or benoxinate hydrochloride. Altafluor Benox should be given to a pregnant woman only if clearly needed.
8.2 Lactation
Risk Summary
There are no data on the presence of fluorescein sodium or benoxinate hydrochloride in human milk after ocular administration of Altafluor Benox, the effects on the breastfed infant, or the effects on milk production.
The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for Altafluor Benox and any potential adverse effects on the breastfed infant from Altafluor Benox.
11. Altafluor Description
Altafluor Benox (fluorescein sodium and benoxinate hydrochloride ophthalmic solution) 0.25%/0.4% is a sterile disclosing agent in combination with a short-acting ester anesthetic for topical ophthalmic use.
Fluorescein sodium is represented by the following structural formula:
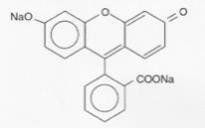
C20H10Na2O5 Mol. Wt. 376.27
Chemical Name: Spiro [isobenzofuran-1 (3H),9’-9[9H] xanthene]-3-one, 3’,6’ dihydroxy, disodium salt.
Benoxinate hydrochloride is represented by the following structural formula:
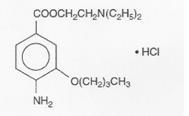
C17H28N2O3 • HCl Mol. Wt. 344.88
Chemical Name: 2-(Diethylamino) etyl 4-amino-3-butoxybenzoate monohydrochloride.
Each mL contains: Actives: fluorescein sodium 2.5 mg (0.25%) equivalent to fluorescein 2.2 mg (0.22%), benoxinate hydrochloride 4 mg (0.4%) equivalent to benoxinate 3.6 mg (0.36%); Inactives: povidone, hydrochloric acid, boric acid, sodium hydroxide, water for injection. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (4.3 – 5.3). Preservative: chlorobutanol 11mg (1.1%).
12. Altafluor - Clinical Pharmacology
This product is the combination of a disclosing agent with a rapidly acting ester anesthetic of short duration.
14. Clinical Studies
Controlled clinical studies in adults and pediatric patients have demonstrated that topical administration of fluorescein sodium and benoxinate hydrochloride ophthalmic solution 0.25%/0.4% enables visualization and corneal anesthesia sufficient to enable applanation tonometry, tear fluid dynamics evaluation and short conjunctival and corneal procedures. Maximal corneal anesthesia usually occurs in about 5-45 seconds and lasts about 20 minutes after single administration.
16. How is Altafluor supplied
Altafluor Benox (fluorescein sodium and benoxinate hydrochloride ophthalmic solution) 0.25%/0.4% is a sterile, yellow to orange-red solution supplied in a 5 mL glass bottle with a sterilized dropper.
NDC #59390-218-05
Storage: Store in refrigerator at 2° to 8°C (36° to 46°F). After opening, Altafluor Benox can be stored up to one month if stored at room temperature or until the expiration date on the bottle if stored in refrigerated conditions. Keep tightly closed.
17. Patient Counseling Information
Accidental Injury Precaution
Advise patients not to touch their eyes for approximately 20 minutes after application. Their eyes will be insensitive due to the effect of the anesthetic, and care should be taken to avoid accidental injuries.
Rev. 12/2017
Manufactured by:
ALTAIRE Pharmaceuticals, Inc.
Aquebogue, NY 11931
| ALTAFLUOR
fluorescein sodium and benoxinate hydrochloride solution |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - Altaire Pharmaceuticals Inc. (786790378) |