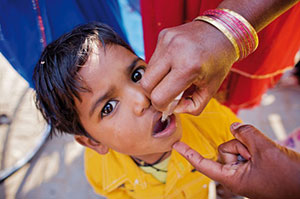
FDA Contributing to Fight Against Polio
On this page:
Few diseases in U.S. history have been as devastating as polio. At the height of the epidemic in 1952, there were nearly 60,000 cases in the United States and more than 3,000 deaths.
The crippling, highly infectious disease is caused by a virus that invades the nervous system and can paralyze a person in a matter of hours. It mainly affects children under 5, but can cause paralysis in unvaccinated adults. The most famous victim of polio was President Franklin D. Roosevelt, who contracted it at age 39, paralyzing him from the waist down.
A successful vaccination program has wiped out polio in the U.S. But the disease still exists in some parts of the world and can easily spread to countries where it has previously been eradicated, requiring continued vaccination of the entire world population.
In the U.S., the Food and Drug Administration’s (FDA) Center for Biologics Evaluation and Research (CBER) regulates vaccines. All vaccines undergo extensive development and rigorous evaluation, and those approved by FDA are continuously monitored for safety and effectiveness.
FDA works with the World Health Organization (WHO) and other partners to make safe and effective vaccines available all over the world.
Unforeseen Challenges
In 1988, when WHO proposed the goal of complete polio eradication, more than 350,000 people were paralyzed by polio in 125 countries on all continents. Compare this with just 223 cases in 2012 in five countries. This is the good news.
However, the bad news is that WHO's original target for eradicating polio was 2000. "Thirteen years later, we're still not there," says Konstantin Chumakov, Ph.D., associate director for research within FDA's Office of Vaccines Research and Review (OVRR).
Chumakov gives two main reasons for this: "We didn’t realize how tough this job would be, and there was a scientific realization that the viruses in the oral vaccine used in the eradication campaign can mutate to virulent forms that circulate in the population."
The vaccine used globally in resource-limited countries is the oral vaccine made from a live virus. It is inexpensive and easy to give—a volunteer worker can place a drop on a child’s tongue.
The vaccine used in the U.S. and other developed countries is made from the killed (inactivated) virus. It is injected into the muscle, and must be given by a health care professional. It cannot mutate and cause polio. But it costs dollars versus pennies per dose.
The inactivated virus vaccine is made from highly virulent strains, says Chumakov. To prevent the virus from being accidentally released into the environment, manufacturers must maintain complex, expensive biosecurity measures. There are only a few manufacturers who supply the killed virus vaccine.
For all these reasons, WHO, Rotary International, the Bill and Melinda Gates Foundation and other organizations support scientists working on the development of new vaccines against poliovirus. "This is critical, says Chumakov. "Without new vaccines, we will not be able to achieve and maintain eradication."
Polio Vaccine Collaborations
Work is under way to create new oral vaccines that are not associated with the risk of mutation into virulent forms.
FDA scientists are an important part of this effort. They assess new virus strains that colleagues at the Centers for Disease Control and Prevention (CDC) propose for the manufacture of improved oral polio vaccines.
"The idea is to change the genetic makeup of the virus in a way that would prevent it from reverting to virulence," says Chumakov. A new, more stable vaccine could be used by WHO for the global eradication program.
Work is also underway to create a new generation of inactivated virus vaccines that are less expensive and produced from non-virulent strains. Cost reduction could be achieved by using adjuvants, substances that help boost the immune response of the vaccinated person, and therefore reduce the dose of the vaccine needed for immunization.
FDA is a part of the steering committee for the Global Access to Adjuvants project under PATH, a global health organization. FDA provides advice on various issues, including adjuvant development, polio eradication, regulatory affairs and manufacturing. Under this project, funded by the Gates Foundation, adjuvants will be evaluated for their potential to safely improve the immune response to inactivated polio vaccines.
The agency also helps PATH and other participating institutions implement an FDA-developed test to determine how well the immune system responds to inactivated vaccines, and to prepare the optimal formulations of the vaccine.
Research by FDA scientists also informs the regulatory process. "FDA is a science-based regulatory agency,” says Chumakov. "You cannot regulate something you don't know about. Biotechnology and vaccine science are evolving very fast. To keep up and to maintain proficiency and knowledge about the development of new technologies, FDA regulatory scientists have to be personally involved in the cutting-edge research."
April 25, 2013
Return to FDA Consumer Articles