Anadrol: Package Insert / Prescribing Info
Package insert / product label
Generic name: oxymetholone
Dosage form: tablet
Drug class: Androgens and anabolic steroids
Medically reviewed by Drugs.com. Last updated on Nov 18, 2024.
On This Page
Peliosis hepatitis, a condition in which liver and sometimes splenic tissue is replaced with blood-filled cysts, has been reported in patients receiving androgenic anabolic steroid therapy. These cysts are sometimes present with minimal hepatic dysfunction, but at other times they have been associated with liver failure. They are often not recognized until life-threatening liver failure or intra-abdominal hemorrhage develops. Withdrawal of drug usually results in complete disappearance of lesions.
Liver cell tumors are also reported. Most often these tumors are benign and androgen-dependent, but fatal malignant tumors have been reported. Withdrawal of drug often results in regression or cessation of progression of the tumor. However, hepatic tumors associated with androgens or anabolic steroids are much more vascular than other hepatic tumors and may be silent until life-threatening intra-abdominal hemorrhage develops.
Blood lipid changes that are known to be associated with increased risk of atherosclerosis are seen in patients treated with androgens and anabolic steroids. These changes include decreased high density lipoprotein and sometimes increased low density lipoprotein. The changes may be very marked and could have a serious impact on the risk of atherosclerosis and coronary artery disease.
Anadrol Description
Anadrol®-50 (oxymetholone) Tablets for oral administration each contain 50 mg of the steroid oxymetholone, a potent anabolic and androgenic drug.
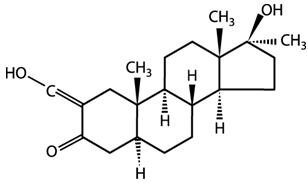
The chemical name for oxymetholone is 17β-hydroxy-2-(hydroxymethylene)-17-methyl-5α-androstan-3-one. The structural formula is:
Inactive Ingredients- lactose, magnesium stearate, povidone, starch
Anadrol - Clinical Pharmacology
Anabolic steroids are synthetic derivatives of testosterone. Nitrogen balance is improved with anabolic agents but only when there is sufficient intake of calories and protein. Whether this positive nitrogen balance is of primary benefit in the utilization of protein-building dietary substances has not been established. Oxymetholone enhances the production and urinary excretion of erythropoietin in patients with anemias due to bone marrow failure and often stimulates erythropoiesis in anemias due to deficient red cell production.
Certain clinical effects and adverse reactions demonstrate the androgenic properties of this class of drugs. Complete dissociation of anabolic and androgenic effects has not been achieved. The actions of anabolic steroids are therefore similar to those of male sex hormones with the possibility of causing serious disturbances of growth and sexual development if given to young children. They suppress the gonadotropic functions of the pituitary and may exert a direct effect upon the testes.
Indications and Usage for Anadrol
Anadrol®-50 Tablets is indicated in the treatment of anemias caused by deficient red cell production. Acquired aplastic anemia, congenital aplastic anemia, myelofibrosis and the hypoplastic anemias due to the administration of myelotoxic drugs often respond. Anadrol®-50 Tablets should not replace other supportive measures such as transfusion, correction of iron, folic acid, vitamin B12 or pyridoxine deficiency, antibacterial therapy and the appropriate use of corticosteroids.
Contraindications
- 1.
- Carcinoma of the prostate or breast in male patients.
- 2.
- Carcinoma of the breast in females with hypercalcemia; androgenic anabolic steroids may stimulate osteolytic resorption of bones.
- 3.
- Oxymetholone can cause fetal harm when administered to pregnant women. It is contraindicated in women who are or may become pregnant. If the patient becomes pregnant while taking the drug, she should be apprised of the potential hazard to the fetus.
- 4.
- Nephrosis or the nephrotic phase of nephritis.
- 5.
- Hypersensitivity to the drug.
- 6.
- Severe hepatic dysfunction.
Warnings
The following conditions have been reported in patients receiving androgenic anabolic steroids as a general class of drugs:
Cholestatic hepatitis and jaundice occur with 17-alpha-alkylated androgens at relatively low doses. Clinical jaundice may be painless, with or without pruritus. It may also be associated with acute hepatic enlargement and right upper-quadrant pain, which has been mistaken for acute (surgical) obstruction of the bile duct. Drug-induced jaundice is usually reversible when the medication is discontinued. Continued therapy has been associated with hepatic coma and death. Because of the hepatoxicity associated with oxymetholone administration, periodic liver function tests are recommended.
In patients with breast cancer, anabolic steroid therapy may cause hypercalcemia by stimulating osteolysis. In this case, the drug should be discontinued.
Edema with or without congestive heart failure may be a serious complication in patients with pre-existing cardiac, renal or hepatic disease. Concomitant administration with adrenal steroids or ACTH may add to the edema. This is generally controllable with appropriate diuretic and/or digitalis therapy.
Geriatric male patients treated with androgenic anabolic steroids may be at an increased risk for the development of prostate hypertrophy and prostatic carcinoma.
Anabolic steroids have not been shown to enhance athletic ability.
Precautions
Concurrent dosing of an anabolic steroid and warfarin may result in unexpectedly large increases in the INR or prothrombin time (PT). When an anabolic steroid is prescribed to a patient being treated with warfarin, doses of warfarin may need to be decreased significantly to maintain the desirable INR level and diminish the risk of potentially serious bleeding. (See PRECAUTIONS-Drug Interactions.)
General:
Women should be observed for signs of virilization (deepening of the voice, hirsutism, acne and clitoromegaly). To prevent irreversible change, drug therapy must be discontinued when mild virilism is first detected. Such virilization is usual following androgenic anabolic steroid use at high doses. Some virilizing changes in women are irreversible even after prompt discontinuance of therapy and are not prevented by concomitant use of estrogens. Menstrual irregularities, including amenorrhea, may also occur.
The insulin or oral hypoglycemic dosage may need adjustment in diabetic patients who receive anabolic steroids.
Anabolic steroids may cause suppression of clotting factors II, V, VII and X, and an increase in prothrombin time.
Information for the patient:
The health care provider should instruct patients to report immediately any use of warfarin and any bleeding.
The health care provider should instruct patients to report any of the following effects of androgens.
Adult or Adolescent Males: Too frequent or persistent erections of the penis, appearance or aggravation of acne.
Laboratory Tests:
Women with disseminated breast carcinoma should have frequent determination of urine and serum calcium levels during the course of androgenic anabolic steroid therapy (see WARNINGS).
Because of the hepatotoxicity associated with the use of 17-alpha-alkylated androgens, liver function tests should be obtained periodically.
Periodic (every 6 months) x-ray examinations of bone age should be made during treatment of prepubertal patients to determine the rate of bone maturation and the effects of androgenic anabolic steroid therapy on the epiphyseal centers.
Anabolic steroids have been reported to lower the level of high-density lipoproteins and raise the level of low-density lipoproteins. These changes usually revert to normal on discontinuation of treatment. Increased low-density lipoproteins and decreased high-density lipoproteins are considered cardiovascular risk factors. Serum lipids and high-density lipoprotein cholesterol should be determined periodically.
Hemoglobin and hematocrit should be checked periodically for polycythemia in patients who are receiving high doses of anabolics.
Because iron deficiency anemia has been observed in some patients treated with oxymetholone, periodic determination of the serum iron and iron binding capacity is recommended. If iron deficiency is detected, it should be appropriately treated with supplementary iron.
Oxymetholone has been shown to decrease 17-ketosteroid excretion.
Drug Interactions:
Warfarin: Clinically significant pharmacokinetic and pharmacodynamic interactions between anabolic steroids and warfarin have been reported in healthy volunteers. When anabolic steroid therapy is initiated in a patient already receiving treatment with warfarin, the INR (international normalized ratio) or prothrombin time (PT) should be monitored closely and the dose of warfarin adjusted as necessary until a stable target INR or PT has been achieved. Furthermore, in patients receiving both ANADROL®-50 Tablets and warfarin, careful monitoring of the INR or PT and adjustment of the warfarin dosage, if indicated, are recommended when the ANADROL®-50 dose is changed or discontinued. Patients should be closely monitored for signs and symptoms of occult bleeding.
Anticoagulants: Anabolic steroids may increase sensitivity to oral anticoagulants. Dosage of the anticoagulant may have to be decreased in order to maintain the desired prothrombin time. Patients receiving oral anticoagulant therapy require close monitoring, especially when anabolic steroids are started or stopped.
Drug/Laboratory Test Interferences:
Therapy with androgenic anabolic steroids may decrease levels of thyroxine-binding globulin resulting in decreased total T4 serum levels and increased resin uptake of T3 and T4. Free thyroid hormone levels remain unchanged and there is no clinical evidence of thyroid dysfunction. Altered tests usually persist for 2 to 3 weeks after stopping anabolic therapy.
Anabolic steroids may cause an increase in prothrombin time.
Anabolic steroids have been shown to alter fasting blood sugar and glucose tolerance tests.
Carcinogenesis, Mutagenesis, Impairment of Fertility
A two-year carcinogenicity study in rats given oxymetholone orally was conducted under the auspices of the US National Toxicology Program (NTP). A wide spectrum of neoplastic and non-neoplastic effects was observed. In male rats, no effects were classified as neoplastic in response to doses up to 150 mg/kg/day (5 times therapeutic exposures with 5 mg/kg based on body surface area). Female rats given 30 mg/kg/day (1 fold the maximum recommended clinical dose of 5 mg/kg/day based on the body surface area) had increased incidences of lung alveolar/bronchiolar adenoma and adenoma or carcinoma combined. At 100 mg/kg/day (about 3 fold the maximum recommended clinical dose of 5 mg/kg/day based on BSA), female rats had increased incidences of hepatocellular adenoma and adenoma or carcinoma combined; the combined incidence of squamous cell carcinoma and carcinoma of the sweat glands also was increased.
Human data: There are rare reports of hepatocellular carcinoma in patients receiving long-term therapy with androgens in high doses. Withdrawal of the drugs did not lead to regression of the tumors in all cases.
Geriatric patients treated with androgens may be at an increased risk of developing prostatic hypertrophy and prostatic carcinoma although conclusive evidence to support this concept is lacking.
In studies conducted under the auspices of the US National Toxicology Program, no evidence of genotoxicity was found using standard assays for mutagenicity, chromosomal aberrations, or induction of micronuclei in erythrocytes.
Impairment of fertility was not tested directly in animal species. However, as noted below under ADVERSE REACTIONS, oligospermia in males and amenorrhea in females are potential adverse effects of treatment with ANADROL®-50 Tablets. Therefore, impairment of fertility is a possible outcome of treatment with ANADROL®-50 Tablets.
Nursing Mothers:
It is not known whether anabolics are excreted in human milk. Because of the potential for serious adverse reactions in nursed infants from anabolics, women who take oxymetholone should not nurse.
Pediatric Use:
Anabolic/androgenic steroids should be used very cautiously in children and only by specialists who are aware of their effects on bone maturation.
Anabolic agents may accelerate epiphyseal maturation more rapidly than linear growth in children, and the effect may continue for 6 months after the drug has been stopped. Therefore, therapy should be monitored by x-ray studies at 6-month intervals in order to avoid the risk of compromising the adult height.
Geriatric use
Clinical studies of ANADROL®-50 Tablets did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Adverse Reactions/Side Effects
Hepatic: Cholestatic jaundice with, rarely, hepatic necrosis and death. Hepatocellular neoplasms and peliosis hepatis have been reported in association with long-term androgenic anabolic steroid therapy (see WARNINGS).
Genitourinary System:
In Men:
Prepubertal: Phallic enlargement and increased frequency of erections.
Postpubertal: Inhibition of testicular function, testicular atrophy and oligospermia, impotence, chronic priapism, epididymitis, bladder irritability and decrease in seminal volume.
In Women: Clitoral enlargement, menstrual irregularities.
In Both Sexes: Increased or decreased libido.
CNS: Excitation, insomnia.
Gastrointestinal: Nausea, vomiting, diarrhea.
Hematologic: Bleeding in patients on concomitant anticoagulant therapy, iron-deficiency anemia.
Leukemia has been observed in patients with aplastic anemia treated with oxymetholone. The role, if any, of oxymetholone is unclear because malignant transformation has been seen in patients with blood dyscrasias and leukemia has been reported in patients with aplastic anemia who have not been treated with oxymetholone.
Breast: Gynecomastia.
Larynx: Deepening of the voice in women.
Hair: Hirsutism and male-pattern baldness in women, male-pattern of hair loss in postpubertal males.
Skin: Acne (especially in women and prepubertal boys).
Skeletal: Premature closure of epiphyses in children (see PRECAUTIONS, Pediatric Use), muscle cramps.
Body as a Whole: Chills.
Fluid and Electrolytes: Edema, retention of serum electrolytes (sodium, chloride, potassium, phosphate, calcium).
Metabolic/Endocrine: Decreased glucose tolerance (see PRECAUTIONS), increased serum levels of low-density lipoproteins and decreased levels of high-density lipoproteins (see PRECAUTIONS, Laboratory Tests), increased creatine and creatinine excretion, increased serum levels of creatinine phosphokinase (CPK). Reversible changes in liver function tests also occur, including increased Bromsulphalein (BSP) retention and increase in serum bilirubin, glutamic-oxaloacetic transaminase (SGOT) and alkaline phosphatase.
Related/similar drugs
Drug Abuse and Dependence
Controlled Substance:
ANADROL®-50 Tablets is considered to be a controlled substance and is listed in Schedule III.
Anadrol Dosage and Administration
The recommended daily dose in children and adults is 1-5 mg/kg of body weight per day. The usual effective dose is 1-2 mg/kg/day but higher doses may be required, and the dose should be individualized. Response is not often immediate, and a minimum trial of three to six months should be given. Following remission, some patients may be maintained without the drug; others may be maintained on an established lower daily dosage. A continued maintenance dose is usually necessary in patients with congenital aplastic anemia.
How is Anadrol supplied
ANADROL®-50 (oxymetholone) Tablets is supplied in bottles of 100 (NDC 68220-055-10) white scored tablets embossed with 0055 and ALAVEN. Keep out of reach of children. Dispense in a tight, light-resistant container.
Storage and Handling
Store at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP].
Rx Only
Manufactured for
ALAVEN Pharmaceutical
Marietta, GA 30067
Anadrol®-50 is a registered trademark of Alaven™ Pharmaceutical
Address medical inquiries to:
Alaven Pharmaceutical LLC
2260 Northwest Parkway, Suite A
Marietta, GA 30067
Or call toll free
1-888-317-0001
ALAVEN™ PHARMACEUTICAL
055 Rev 12/06
©2006, ALAVEN Pharmaceutical
| ANADROL-50
oxymetholone tablet |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Alaven Pharmaceutical LLC (140210829) |
More about Anadrol-50 (oxymetholone)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: androgens and anabolic steroids