Verucide Physician Formula
Dosage form: liquid
Ingredients: SALICYLIC ACID 17mL in 10mL
Labeler: Blaine Labs Inc.
NDC code: 63347-700
Medically reviewed by Drugs.com. Last updated on Mar 10, 2025.
Salicylic Acid 17%
Alcohol 60%
Water, SDA Alcohol 40B, Carcia Papaya Fruit Extract, Hydroxypropyl-cellulose, Artemisia Scoparia Oil, FD C Brown 1
Wart Removal
Skin Cleanser
For the removal of common warts (easily recognized by the rough cauliflower-like appearance of the surface) For the removal of plantar warts that are located on the bottom of the foot.
Wash Affected area with one or two drops of the Pre-Treatment Solution and scrub with a brush. Rinse and dry thoroughly. Dab the wart removal solution directly onto the wart and allow to dry. Repeat steps once daily until the wart is gone.
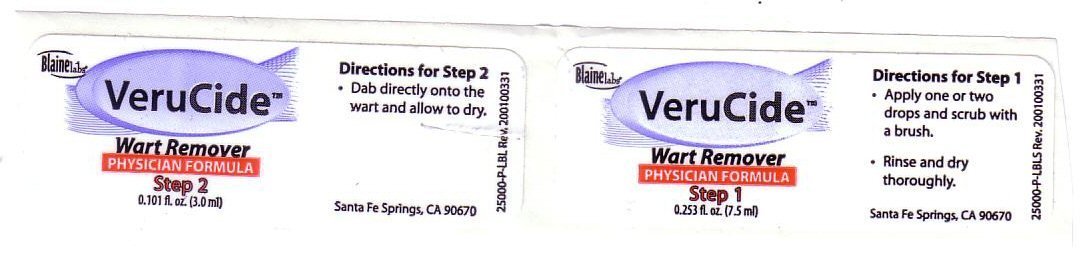
Directions for Step 1 Apply one or two drops and scrub with a brush Rinse and dry thoroughly
Directions for Step 2 Dab directly onto the wart and allow to dry.
Keep Out Of Reach Of Children
Avoid contact with the eyes. If eye contact does occur, rinse thoroughly with water for 15 minutes. For external use only. Avoid inhaling vapors. Extremely flammable. Keep away from fire or flame. Replace bottle caps tightly and store at room temperature away from heat. Do not use on irritated skin, moles, any areas that is infected or reddened, birthmarks, genital warts, warts with hair growing from them, warts on the face, warts on mucous membranes (such as inside the mouth, nose, anus, genitals, or lips)
Stop and ask a Doctor if discomfort persists. If swallowed get medical help or contact a poison control center right away. Ask a doctor before use if you have diabetes or poor blood circulation.
Questions or comments Call 800-307-8818 Monday through Friday 8:00 am to 5:00 pm (Pacific Time) Please recycle. This package is made from recyclable materials. Proudly made in the USA.

| VERUCIDE PHYSICIAN FORMULA
salicyllic acid liquid |
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
|
|||||||||||||||||||
| Labeler - Blaine Labs Inc. (017314571) |
| Registrant - Blaine Labs Inc. (017314571) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Blaine Labs Inc. | 017314571 | manufacture | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
