M 86 Pill - orange oval, 15mm
Pill with imprint M 86 is Orange, Oval and has been identified as Captopril and Hydrochlorothiazide 50 mg / 25 mg. It is supplied by Mylan Pharmaceuticals Inc.
Captopril/hydrochlorothiazide is used in the treatment of Heart Failure; High Blood Pressure and belongs to the drug class ACE inhibitors with thiazides. There is positive evidence of human fetal risk during pregnancy. Captopril/hydrochlorothiazide 50 mg / 25 mg is not a controlled substance under the Controlled Substances Act (CSA).
Images for M 86
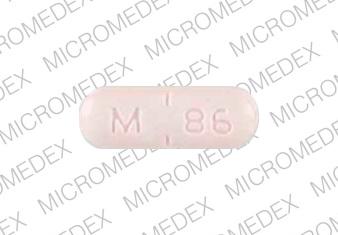
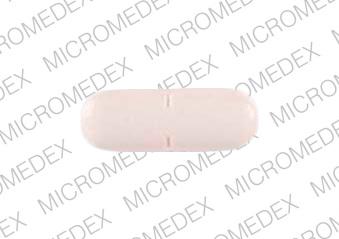

Captopril and Hydrochlorothiazide
- Imprint
- M 86
- Strength
- 50 mg / 25 mg
- Color
- Orange
- Size
- 15.00 mm
- Shape
- Oval
- Availability
- Prescription only
- Drug Class
- ACE inhibitors with thiazides
- Pregnancy Category
- D - Positive evidence of risk
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Mylan Pharmaceuticals Inc.
- Inactive Ingredients
-
lactose anhydrous,
silicon dioxide,
croscarmellose sodium,
magnesium stearate,
microcrystalline cellulose,
corn starch,
sodium lauryl sulfate,
FD&C Yellow No. 6
Note: Inactive ingredients may vary.
Labelers / Repackagers
| NDC Code | Labeler / Repackager |
|---|---|
| 00378-0086 (Discontinued) | Mylan Pharmaceuticals Inc. |
| 54868-4062 (Discontinued) | Physicians Total Care Inc. (repackager) |
Related images for "M 86"
More about captopril / hydrochlorothiazide
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (1)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: ACE inhibitors with thiazides
- En español
Patient resources
- Hydrochlorothiazide and captopril drug information
- Captopril and hydrochlorothiazide (Advanced Reading)
Other brands
Capozide 25/15, Capozide 25/25, Capozide 50/15, Capozide 50/25
Professional resources
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.