Ciclopirox Topical Suspension Prescribing Information
Package insert / product label
Generic name: ciclopirox olamine
Dosage form: suspension, topical
Drug class: Topical antifungals
Medically reviewed by Drugs.com. Last updated on Dec 12, 2023.
On This Page
Ciclopirox Topical Suspension Description
Ciclopirox Topical Suspension, USP 0.77% is for topical use.
Each ml of Ciclopirox Topical Suspension, USP 0.77% contains 7.70 mg of ciclopirox (as ciclopirox olamine) in a water miscible suspension base consisting of purified water USP, cocamide DEA, octyldodecanol NF, mineral oil USP, stearyl alcohol NF, cetyl alcohol NF, polysorbate 60 NF, myristyl alcohol NF, lactic acid USP, sorbitan monostearate NF, and benzyl alcohol NF (1%) as preservative.
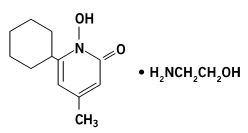
Ciclopirox Topical Suspension, USP 0.77% contains a synthetic, broad spectrum, antifungal agent ciclopirox (as ciclopirox olamine). The chemical name is 6-cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridone, 2-aminoethanol salt.
The CAS Registry Number is 41621-49-2.
Ciclopirox Topical Suspension, USP 0.77% has a pH of 7.
The chemical structure is:
Ciclopirox Topical Suspension - Clinical Pharmacology
Mechanism of Action
Ciclopirox is a hydroxypyridone antifungal agent that acts by chelation of polyvalent cations (Fe3+ or Al3+), resulting in the inhibition of the metal-dependent enzymes that are responsible for the degradation of peroxides within the fungal cell.
Pharmacokinetics
Pharmacokinetic studies in men with radiolabeled ciclopirox solution in polyethylene glycol 400 showed an average of 1.3% absorption of the dose when it was applied topically to 750 cm2 on the back followed by occlusion for 6 hours. The biological half life was 1.7 hours and excretion occurred via the kidney. Two days after application only 0.01% of the dose applied could be found in the urine. Fecal excretion was negligible. Autoradiographic studies with human cadaver skin showed that ciclopirox penetrates into the hair and through the epidermis and hair follicles into the sebaceous glands and dermis, while a portion of the drug remains in the stratum corneum.
In vitro penetration studies in frozen or fresh excised human cadaver and pig skin indicated that the penetration of Ciclopirox Topical Suspension, USP 0.77% is equivalent to that of Ciclopirox Cream, 0.77%. Therapeutic equivalence of cream and suspension formulations also was indicated by studies of experimentally induced guinea pig and human trichophytosis.
Indications and Usage for Ciclopirox Topical Suspension
Ciclopirox Topical Suspension, USP 0.77% is indicated for the topical treatment of the following dermal infections: tinea pedis, tinea cruris, and tinea corporis due to Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, and Microsporum canis; ; cutaneous candidiasis (moniliasis) due to Candida albicans; and tinea (pityriasis) versicolor due to Malassezia furfur.
Contraindications
Ciclopirox Topical Suspension, USP 0.77% is contraindicated in individuals who have shown hypersensitivity to any of its components.
Warnings
PRECAUTIONS
If a reaction suggesting sensitivity or chemical irritation should occur with the use of Ciclopirox Topical Suspension, USP 0.77%, treatment should be discontinued and appropriate therapy instituted.
Information for Patients
The patient should be told to:
- Use the medication for the full treatment time even though signs/symptoms may have improved and notify the physician if there is no improvement after four weeks.
- Inform the physician if the area of application shows signs of increased irritation (redness, itching, burning, blistering, swelling, oozing) indicative of possible sensitization.
- Avoid the use of occlusive wrappings or dressings
Carcinogenesis, Mutagenesis, Impairment of Fertility
A 104-week dermal carcinogenicity study in mice was conducted with ciclopirox cream applied at doses up to 1.93% (100 mg/kg/day or 300 mg/m2/day). No increase in drug related neoplasms was noted when compared to control.
The following in vitro genotoxicity tests have been conducted with ciclopirox: evaluation of gene mutation in the Ames Salmonella and E. coli assays (negative); chromosome aberration assays in V79 Chinese hamster lung fibroblast cells, with and without metabolic activation (positive); chromosome aberration assays in V79 Chinese hamster lung fibroblast cells in the presence of supplemental Fe3+, with and without metabolic activation (negative); gene mutation assays in the HGPRT-test with V79 Chinese hamster lung fibroblast cells (negative); and a primary DNA damage assay (i.e., unscheduled DNA synthesis assay in A549 human cells) (negative). An in vitro cell transformation assay in BALB/c 3T3 cells was negative for cell transformation. In an in vivo Chinese hamster bone marrow cytogenetic assay, ciclopirox was negative for chromosome aberrations at a dosage of 5000 mg/kg body weight.
A combined oral fertility and embryofetal developmental study was conducted in rats with ciclopirox olamine. No effect on fertility or reproductive performance was noted at the highest dose tested of 3.85 mg/kg/day ciclopirox (approximately 1.2 times the maximum recommended human dose based on body surface area comparisons).
Pregnancy
Teratogenic Effects: Pregnancy Category B
There are no adequate or well-controlled studies in pregnant women. Therefore, Ciclopirox Topical Suspension, USP 0.77% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Oral embryofetal developmental studies were conducted in mice, rats, rabbits and monkeys. Ciclopirox or ciclopirox olamine was orally administered during the period of organogenesis. No maternal toxicity, embryotoxicity or teratogenicity were noted at the highest doses of 77, 125, 80 and 38.5 mg/kg/day ciclopirox in mice, rats, rabbits and monkeys, respectively (approximately 11, 37, 51 and 24 times the maximum recommended human dose based on body surface area comparisons, respectively).
Dermal embryofetal developmental studies were conducted in rats and rabbits with ciclopirox olamine dissolved in PEG 400. Ciclopirox olamine was topically administered during the period of organogenesis. No maternal toxicity, embryotoxicity or teratogenicity were noted at the highest doses of 92 mg/kg/day and 77 mg/kg/day ciclopirox in rats and rabbits, respectively (approximately 27 and 49 times the maximum recommended human dose based on body surface area comparisons, respectively).
Adverse Reactions/Side Effects
In the controlled clinical trial with 89 patients using Ciclopirox Topical Suspension, USP 0.77% and 89 patients using the vehicle, the incidence of adverse reactions was low. Those considered possibly related to treatment or occurring in more than one patient were pruritus, which occurred in two patients using ciclopirox suspension and one patient using the suspension vehicle, and burning, which occurred in one patient using ciclopirox suspension.
Ciclopirox Topical Suspension Dosage and Administration
Gently massage Ciclopirox Topical Suspension, USP 0.77% into the affected and surrounding skin areas twice daily, in the morning and evening. Clinical improvement with relief of pruritus and other symptoms usually occurs within the first week of treatment. If a patient shows no clinical improvement after four weeks of treatment with Ciclopirox Topical Suspension, USP 0.77%, the diagnosis should be redetermined. Patients with tinea versicolor usually exhibit clinical and mycological clearing after two weeks of treatment.
How is Ciclopirox Topical Suspension supplied
Ciclopirox Topical Suspension, USP 0.77% is supplied in
30 mL bottles (NDC 69315-309-30)
60 mL bottles (NDC 69315-309-60)
Bottle space provided to allow for vigorous shaking before each use.
Store between 5º - 25ºC (41º - 77ºF).
To report SUSPECTED ADVERSE REACTIONS, call 1-866-306-4256 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Manufactured for: Leading Pharma, LLC
3 Oak Road, Fairfield, NJ 07004-2402 USA
www.leadingpharma.com
Manufactured by: Teligent Pharma, Inc., Buena, NJ 08310
Iss. 09/18
| CICLOPIROX OLAMINE
ciclopirox olamine suspension |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Leading Pharma, LLC (079575060) |
| Registrant - Medimetriks Pharamceuticals, Inc (019903816) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Teligent Pharma, Inc. | 011036910 | MANUFACTURE(69315-309) , PACK(69315-309) | |
Frequently asked questions
More about ciclopirox topical
- Compare alternatives
- Pricing & coupons
- Reviews (53)
- Side effects
- Dosage information
- During pregnancy
- Drug class: topical antifungals
- Breastfeeding
- En español
Patient resources
Professional resources
- Ciclopirox monograph
- Ciclopirox (FDA)
- Ciclopirox Gel (FDA)
- Ciclopirox Nail Lacquer (FDA)
- Ciclopirox Shampoo (FDA)
- Ciclopirox Topical Solution (FDA)




