Qutenza Prescribing Information
Package insert / product label
Generic name: capsaicin
Dosage form: patch
Drug class: Miscellaneous topical agents
J Code (medical billing code): J7336 (Per 10 cm2)
Medically reviewed by Drugs.com. Last updated on Jun 15, 2023.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
QUTENZA® (capsaicin) 8% patch
Initial U.S. Approval: 2009
Indications and Usage for Qutenza
- •
- Qutenza is a TRPV1 channel agonist indicated for the management of neuropathic pain associated with postherpetic neuralgia (PHN). (1)
Qutenza Dosage and Administration
- •
- Only physicians or health care professionals under the close supervision of a physician are to administer Qutenza. (2.1)
- •
- Do not use Qutenza on broken skin. (2.1)
- •
- Apply Qutenza to the most painful skin areas, using up to four patches. (2.2)
- •
- Apply Qutenza for 60 minutes and repeat every 3 months or as warranted by the return of pain (not more frequently than every three months). (2.2)
- •
- Use only nitrile (not latex) gloves when handling Qutenza and when cleaning treatment areas. (2.1)
- •
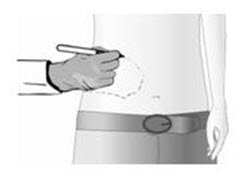
- Before patch application, a physician must identify and mark the painful area, including areas of hypersensitivity and allodynia. (2.3)
- •
- Apply a topical anesthetic before Qutenza application. (2.3)
- •
- Apply Qutenza by placing on the skin while slowly removing the release liner from underneath. (2.3)
- •
- Remove the Qutenza patches by gently and slowly rolling them inward. (2.3)
- •
- After removal of Qutenza, apply Cleansing Gel for one minute and then remove it with a dry wipe. (2.3)
- •
- Treat acute pain during and following the procedure with local cooling and/or analgesics. (5.4)
- •
- Dispose of patches and other treatment materials immediately after use in accordance with local biomedical waste procedures. (2.1)
- •
- The treated area may be sensitive for a few days to heat (e.g., hot showers/baths, direct sunlight, vigorous exercise). (2.3)
Dosage Forms and Strengths
- •
- Qutenza patch contains 8% capsaicin (640 mcg/cm2). Each patch contains a total of 179 mg of capsaicin. (3)
Contraindications
- •
- None
Warnings and Precautions
- •
- Do not use near eyes or mucous membranes. (5.1)
- •
- Inhalation of airborne capsaicin can result in coughing or sneezing. (5.2)
- •
- If irritation of eyes or airway occurs, remove the affected individual from the vicinity of Qutenza and flush the mucous membranes or eyes with water. If skin not intended to be treated comes into contact with Qutenza, apply Cleansing Gel and then wipe off with dry gauze. (5.2, 5.3)
- •
- Transient increases in blood pressure may occur in patients during and shortly after the Qutenza treatment. Monitor blood pressure during and following the treatment procedure. For those patients who require the use of opioids to treat pain during or following the procedure, their ability to perform potentially hazardous activities such as driving or operating machinery may be affected. (5.4, 5.5)
Adverse Reactions/Side Effects
The most common adverse reactions (≥ 5% and greater than control) are application site erythema, application site pain, application site pruritus and application site papules. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Averitas Pharma at 1-877-900-6479 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2018
Full Prescribing Information
1. Indications and Usage for Qutenza
Qutenza is indicated for the management of neuropathic pain associated with postherpetic neuralgia.
2. Qutenza Dosage and Administration
2.1 Special Precautions
- •
- Only physicians or health care professionals under the close supervision of a physician are to administer Qutenza.
- •
- Use only nitrile gloves when handling Qutenza, and when cleaning capsaicin residue from the skin. Do not use latex gloves as they do not provide adequate protection.
- •
- Immediately after use, dispose of used and unused patches, Cleansing Gel and other treatment materials in accordance with the local biomedical waste procedures.
- •
- Use Qutenza only on dry, intact (unbroken) skin.
- •
- Apply the Qutenza patch within 2 hours of opening the pouch.
2.2 Dosing
The recommended dose of Qutenza is a single, 60-minute application of up to four patches.
Treatment with Qutenza may be repeated every three months or as warranted by the return of pain (not more frequently than every three months).
2.3 Instructions for Use
Prepare
Put on nitrile gloves. Inspect the pouch. Do not use if the pouch has been torn or damaged.
Identify
The treatment area (painful area including areas of hypersensitivity and allodynia) must be identified by a physician and marked on the skin.
If necessary, clip hair (do not shave) in and around the identified treatment area to promote patch adherence.
Qutenza can be cut to match the size and the shape of the treatment area.
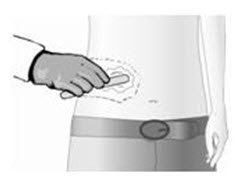
Gently wash the treatment area with mild soap and water and dry thoroughly.
Anesthetize
Pre-treat with a topical anesthetic to reduce discomfort associated with the application of Qutenza.
Apply topical anesthetic to the entire treatment area and surrounding 1 to 2 cm, and keep the local anesthetic in place until the skin is anesthetized prior to the application of Qutenza patch.
Remove the topical anesthetic with a dry wipe. Gently wash the treatment area with mild soap and water and dry thoroughly.
Apply
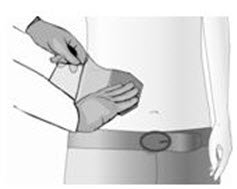
Tear open the pouch along the three dashed lines, remove the Qutenza patch.
Inspect the Qutenza patch and identify the outer surface backing layer with the printing on one side and the capsaicin-containing adhesive on the other side. The adhesive side of the patch is covered by a clear, unprinted, diagonally-cut release liner.
Cut Qutenza before removing the protective release liner.
The diagonal cut in the release liner is to aid in its removal. Peel a small section of the release liner back, and place the adhesive side of the patch on the treatment area.
While you slowly peel back the release liner from under the patch with one hand, use your other hand to smooth the patch down on to the skin.
Once Qutenza is applied, leave in place for 60 minutes.
To ensure Qutenza maintains contact with the treatment area, a dressing, such as rolled gauze, may be used.
Instruct the patient not to touch the patch or treatment area.
Remove
Remove Qutenza patches by gently and slowly rolling them inward.
Cleanse
After removal of Qutenza, generously apply Cleansing Gel to the treatment area and leave on for at least one minute. Remove Cleansing Gel with a dry wipe and gently wash the area with mild soap and water and dry thoroughly.
Dispose of all treatment materials as described. [see Dosage and Administration (2.1)]
Inform the patient that the treated area may be sensitive for a few days to heat (e.g., hot showers/baths, direct sunlight, vigorous exercise).
3. Dosage Forms and Strengths
Qutenza patch contains 8% capsaicin (640 mcg/cm2). Each patch contains a total of 179 mg of capsaicin.
5. Warnings and Precautions
5.1 Eye and Mucous Membrane Exposure
Do not apply Qutenza to the face or scalp to avoid risk of exposure to the eyes or mucous membranes.
5.2 Aerosolization of Capsaicin
Aerosolization of capsaicin can occur upon rapid removal of Qutenza patches. Therefore, remove Qutenza patches gently and slowly by rolling the adhesive side inward [see Dosage and Administration (2.3)].
If irritation of eyes or airways occurs, remove the affected individual from the vicinity of Qutenza. Flush eyes and mucous membranes with cool water.
Inhalation of airborne capsaicin can result in coughing or sneezing. Provide supportive medical care if shortness of breath develops.
5.3 Unintended Skin Exposure
If skin not intended to be treated comes in contact with Qutenza, apply Cleansing Gel for one minute and wipe off with dry gauze. After the Cleansing Gel has been wiped off, wash the area with soap and water.
5.4 Application Associated Pain
Even following use of a local anesthetic prior to administration of Qutenza, patients may experience substantial procedural pain. Prepare to treat acute pain during and following the application procedure with local cooling (such as an ice pack) and/or appropriate analgesic medication, such as opioids. Opioids may affect the ability to perform potentially hazardous activities such as driving or operating machinery.
5.5 Increase in Blood Pressure
In clinical trials, increases in blood pressure occurred during or shortly after exposure to Qutenza. The changes averaged less than 10 mm Hg, although some patients had greater increases and these changes lasted for approximately two hours after patch removal. Increases in blood pressure were unrelated to the pretreatment blood pressure but were related to treatment-related increases in pain. Monitor blood pressure periodically during the treatment and provide adequate support for treatment related pain.
Patients with unstable or poorly controlled hypertension, a recent history of cardiovascular or cerebrovascular events may be at an increased risk of adverse cardiovascular effects. Consider these factors prior to initiating Qutenza treatment.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are discussed elsewhere in the labeling:
Application-Associated Pain [see Warnings and Precautions (5.4)]
Increase in Blood Pressure [see Warnings and Precautions (5.5)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of other drugs and may not reflect the rates observed in clinical practice.
Across all controlled and uncontrolled trials, more than 1,600 patients have received Qutenza. A total of 394 patients received more than one treatment application and 274 patients were followed for 48 weeks or longer.
In controlled clinical studies, 98% of patients completed ≥ 90% of the intended patch application duration. Among patients treated with Qutenza, 1% discontinued prematurely due to an adverse event.
Controlled Clinical Studies
Common Adverse Reactions
Adverse reactions occurring in ≥ 5% of patients in the Qutenza group and at an incidence greater than in the control group were application site erythema, application site pain, application site pruritus and application site papules.
Table 1 summarizes all adverse reactions, regardless of causality, occurring in ≥ 1% of patients with postherpetic neuralgia in the Qutenza group for which the incidence was greater than in the control group. The majority of application site reactions were transient and self-limited. Transient increases in pain were commonly observed on the day of treatment in patients treated with Qutenza. Pain increases occurring during patch application usually began to resolve after patch removal. On average, pain scores returned to baseline by the end of the treatment day and then remained at or below baseline levels. A majority of Qutenza-treated patients in clinical studies had adverse reactions with a maximum intensity of “mild” or “moderate”.
|
Body System |
Qutenza |
Control |
|
General Disorders and Administration Site Conditions | ||
|
Application Site Erythema |
63 |
54 |
|
Application Site Pain |
42 |
21 |
|
Application Site Pruritus |
6 |
4 |
|
Application Site Papules |
6 |
3 |
|
Application Site Edema |
4 |
1 |
|
Application Site Swelling |
2 |
1 |
|
Application Site Dryness |
2 |
1 |
|
Infections and Infestations | ||
|
Nasopharyngitis |
4 |
2 |
|
Bronchitis |
2 |
1 |
|
Sinusitis |
3 |
1 |
|
Gastrointestinal Disorders | ||
|
Nausea |
5 |
2 |
|
Vomiting |
3 |
1 |
|
Skin and Subcutaneous Tissue Disorder | ||
|
Pruritus |
2 |
< 1 |
|
Vascular Disorders | ||
|
Hypertension |
2 |
1 |
Other Adverse Reactions Observed During the Clinical Studies of Qutenza
General Disorders and Administration Site Conditions: Application site urticaria, Application site paresthesia, Application site dermatitis, Application site hyperesthesia, Application site excoriation, Application site warmth, Application site anesthesia, Application site bruising, Application site inflammation, Application site exfoliation, Peripheral edema
Nervous System Disorders: Headache, Burning sensation, Peripheral sensory neuropathy, Dizziness, Dysgeusia, Hyperesthesia, Hypoesthesia
Respiratory, Thoracic and Mediastinal Disorders: Cough, Throat irritation
Skin and Subcutaneous Tissue Disorders: Abnormal skin odor
7. Drug Interactions
No clinical drug interaction studies have been performed.
Data from in vitro cytochrome P450 inhibition and induction studies show that capsaicin does not inhibit or induce liver cytochrome P450 enzymes at concentrations which far exceed those measured in blood samples. Therefore, interactions with systemic medicinal products are unlikely.
8. Use In Specific Populations
8.1 Pregnancy
Teratogenic effects: Pregnancy Category B
There are no adequate and well-controlled studies evaluating Qutenza in pregnant women.
There was no evidence of fetal teratogenicity in embryofetal developmental toxicological studies conducted in pregnant rats and rabbits in which Qutenza patches (rats) or liquid (rabbits) were applied once daily for a 3 hour duration during the period of fetal organogenesis up to doses corresponding to an 11-fold (rat, 32 mg capsaicin patch/day) and 37-fold (rabbit, 260 mg capsaicin/day) margin over the maximum recommended human dose [MRHD] based on a Cmax exposure comparison.
In a peri- and post-natal reproduction toxicology study, pregnant female rats were treated with Qutenza patches at doses up to 32 mg capsaicin patch/rat/day applied once daily for a 3 hours duration during gestation and lactation (from gestation day 7 through day 28 postpartum). Analyses of milk samples on day 14 of the lactation period demonstrated measurable levels of capsaicin in the dam's milk at all dose levels. There were no effects on survival, growth, learning and memory tests (passive avoidance and water maze), sexual maturation, mating, pregnancy, and fetal development in the offspring of mothers treated with capsaicin up to 32 mg capsaicin patch/rat/day (corresponding to an 11-fold margin over the MRHD based on Cmax exposure).
8.3 Nursing Mothers
There are no adequate and well-controlled studies in nursing women. Studies in rat have demonstrated capsaicin is excreted into breast milk of this species. It is unknown whether capsaicin is excreted in human breast milk. Because Qutenza is administered as a single 60-minute application and capsaicin is rapidly cleared from the bloodstream [see Clinical Pharmacology (12.3)], mothers can reduce infant exposure by not breast-feeding after treatment on the day of treatment.
8.4 Pediatric Use
The safety and effectiveness of Qutenza in patients younger than 18 years of age have not been studied.
8.5 Geriatric Use
In controlled clinical studies of Qutenza in neuropathic pain associated with postherpetic neuralgia, 75% of patients were 65 years and older and 43% of patients were 75 years and older.
Safety and effectiveness were similar in geriatric patients and younger patients. No dose adjustments are required in geriatric patients.
10. Overdosage
There is no clinical experience with Qutenza overdose in humans.
There is no specific antidote for overdose with capsaicin. In case of suspected overdose, remove patches gently, apply Cleansing Gel for one minute, wipe off with dry gauze and gently wash the area with soap and water. Use supportive measures and treat symptoms as clinically warranted.
11. Qutenza Description
Qutenza (capsaicin) 8% patch contains capsaicin in a localized dermal delivery system. The capsaicin in Qutenza is a synthetic equivalent of the naturally occurring compound found in chili peppers. Capsaicin is soluble in alcohol, acetone, and ethyl acetate and very slightly soluble in water.
Qutenza is a single-use patch stored in a foil pouch. Each Qutenza patch is 14 cm x 20 cm (280 cm2) and consists of a polyester backing film coated with a drug-containing silicone adhesive mixture, and covered with a removable polyester release liner.
The backing film is imprinted with “capsaicin 8%”. Each Qutenza patch contains a total of 179 mg of capsaicin (8% in adhesive, 80 mg per gram of adhesive) or 640 micrograms (mcg) of capsaicin per square cm of patch.
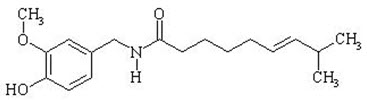
The empirical formula is C18H27NO3, with a molecular weight of 305.42. The chemical compound capsaicin [(E)-8-methyl-N-vanillyl-6-nonenamide] is an activating ligand for transient receptor potential vanilloid 1 receptor (TRPV1) and it has the following structure:
The patch contains the following inactive ingredients: diethylene glycol monoethyl ether, dimethicone, ethyl cellulose, polyester film, silicone adhesive and white ink.
Qutenza is supplied with a Cleansing Gel which is used to remove residual capsaicin from the skin after treatment. Cleansing Gel consists of the following ingredients: butylated hydroxyanisole, carbomer copolymer, edetate disodium, polyethylene glycol, purified water, and sodium hydroxide.
12. Qutenza - Clinical Pharmacology
12.1 Mechanism of Action
Capsaicin is an agonist for the transient receptor potential vanilloid 1 receptor (TRPV1), which is an ion channel-receptor complex expressed on nociceptive nerve fibers in the skin. Topical administration of capsaicin causes an initial enhanced stimulation of the TRPV1-expressing cutaneous nociceptors that may be associated with painful sensations. This is followed by pain relief thought to be mediated by a reduction in TRPV1-expressing nociceptive nerve endings [see Clinical Pharmacology (12.2)]. Over the course of several months, there may be a gradual re-emergence of painful neuropathy thought to be due to TRPV1 nerve fiber reinnervation of the treated area.
12.2 Pharmacodynamics
Two studies evaluated the pharmacodynamic effects of Qutenza on sensory function and epidermal nerve fiber (ENF) density in healthy volunteers. Consistent with the known pharmacodynamic effects of capsaicin on TRPV1-expressing nociceptive nerve endings, reduced ENF density and minor changes in cutaneous nociceptive function (heat detection and sharp sensation) were noted one week after exposure to Qutenza. ENF density reduction and sensory changes were fully reversible.
12.3 Pharmacokinetics
Pharmacokinetic data in humans showed transient, low (< 5 ng/mL) systemic exposure to capsaicin in about one third of PHN patients following 60-minute applications of Qutenza. The highest plasma concentration of capsaicin detected was 4.6 ng/mL and occurred immediately after Qutenza removal. Most quantifiable levels were observed at the time of Qutenza removal and were below the limit of quantitation 3 to 6 hours after Qutenza removal. No detectable levels of metabolites were observed in any subject.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Adequate carcinogenicity studies have not been conducted with Qutenza or capsaicin.
Mutagenesis
Capsaicin was not mutagenic in the Ames, mouse micronucleus and chromosomal aberration in human peripheral blood lymphocytes assays. As with other catechol-containing compounds (e.g., dopamine), capsaicin showed a weak mutagenic response in the mouse lymphoma assay.
Impairment of Fertility
A fertility and reproductive toxicology study was conducted in rats with exposure to Qutenza patches daily for 3 hours/day beginning 28 days before cohabitation, through cohabitation and continuing through the day before sacrifice (approximately 49 days of treatment). The results revealed a statistically significant reduction in the number and percent of motile sperm. Sperm motility obtained from the vas deferens was reduced in all capsaicin treatment groups (16, 24 and 32 mg capsaicin patch/rat/day). Though a “no effect” level was not determined, dose levels used in the study correspond to a 13- to 28-fold exposure margin over the mean Cmax associated with the maximal human recommended dose. Sperm counts were reduced in the vas deferens or cauda epididymis in the 24 and 32 mg capsaicin patch/rat/day dose groups (79 and 69%, respectively) compared to the placebo patch treated control group; however, these reductions did not adversely affect fertility. As this animal model has a large excess of sperm generating capacity relative to the threshold necessary for fertilization, the lack of an effect on fertility in this species is of unknown significance for human risk assessment.
14. Clinical Studies
14.1 Postherpetic Neuralgia
The efficacy of Qutenza, was established in two 12-week, double-blind, randomized, dose-controlled, multicenter studies. These studies enrolled patients with postherpetic neuralgia (PHN) persisting for at least 6 months following healing of herpes zoster rash and a baseline score of 3-9 on an 11-point Numerical Pain Rating Scale (NPRS) ranging from 0 (no pain) to 10 (worst possible pain). Qutenza and a control patch were each applied as a single 60-minute application. The control used in these studies looked similar to Qutenza but contained a low concentration of the active ingredient, capsaicin (3.2 mcg/cm2, 0.04% w/w) to retain blinding regarding the known application site reactions of capsaicin (such as burning and erythema). The baseline mean pain scores across the 2 studies was approximately 6.0. Patients who entered the study on stable doses of pain-control medications were required to keep dosing stable throughout the duration of the study. Approximately half of the patients were taking concomitant medications including anticonvulsants, non-SSRI antidepressants, or opioids for their PHN at study entry. Prior to study patch application a topical anesthetic was applied to the treatment area for 60 minutes. Patients were permitted to use local cooling and additional analgesic medications for treatment-related discomfort as needed through Day 5. Patients recorded their pain daily in a diary.
PHN Study 1: In this 12-week study, the Qutenza group demonstrated a greater reduction in pain compared to the Control group during the primary assessment at Week 8. The percent change in average pain from baseline to Week 8 was -18% (±2%) for the low-dose control and -29% (±2%) for Qutenza.
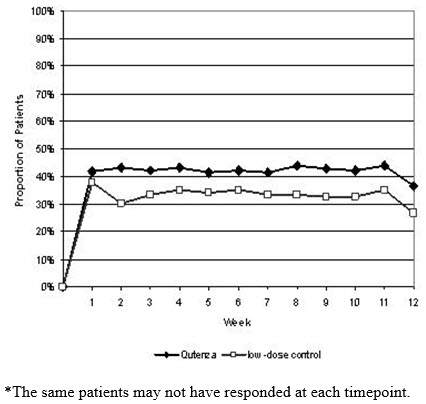
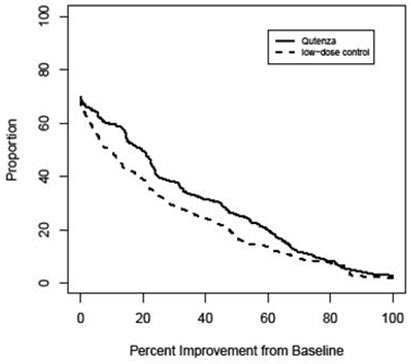
For various degrees of improvement in pain from baseline to study endpoint, Figure 2 shows the fraction of patients achieving that degree of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study through Week 12 or who showed no improvement at Week 12 were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study. The proportion of patients experiencing ≥ 30% reduction in pain intensity from baseline for each week through Week 12 is shown in Figure 3.
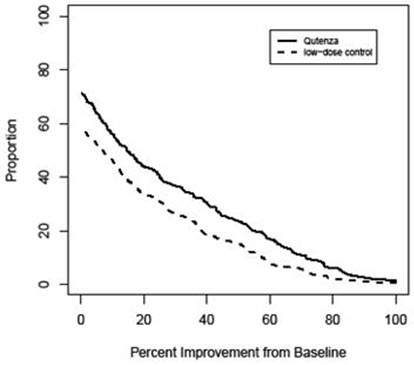
Figure 2: Patients Achieving Various Percentages of Reduction in Pain Intensity at Week 12 – Study 1
PHN Study 2: In this 12-week study the Qutenza group demonstrated a greater reduction in pain compared to the Control group during the primary assessment at Week 8. The percent change in average pain from baseline to Week 8 was -26% (±2%) for the low-dose control and -33% (±2%) for Qutenza.
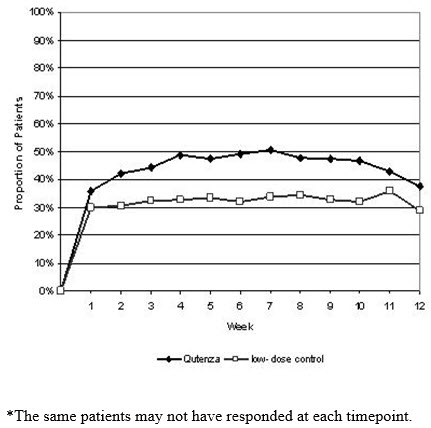
For various degrees of improvement in pain from baseline to study endpoint, Figure 4 shows the fraction of patients achieving that degree of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study through Week 12 or who showed no improvement at Week 12 were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study. The proportion of patients achieving ≥ 30% reduction in pain intensity from baseline for each week through Week 12 is shown in Figure 5.
16. How is Qutenza supplied
16.1 How Supplied
Qutenza (capsaicin) 8% patch is a single-use patch stored in a sealed pouch (NDC 72512-920-00).
Each individual patch is printed with “capsaicin 8%”.
Cleansing Gel is provided in a 50 g tube.
Qutenza is available in the following presentations:
Carton of 1 patch and 50 g tube of Cleansing Gel
(NDC 72512-928-01).
Carton of 2 patches and 50 g tube of Cleansing Gel
(NDC 72512-929-01).
16.2 Storage
Store carton between 20° to 25°C (68° to 77°F). Excursions between 15°C and 30°C (59°F and 86°F) are allowed.
Keep the patch in the sealed pouch until immediately before use.
16.3 Handling and Disposal
Qutenza contains capsaicin capable of producing severe irritation of eyes, skin, respiratory tract and mucous membranes. Do not dispense Qutenza to patients for self-administration. It is critical that health care professionals who administer Qutenza have completely familiarized themselves with proper dosing, handling, and disposal procedures before handling Qutenza to avoid accidental or inadvertent capsaicin exposure to themselves or others [see Dosage and Administration (2)].
- •
- Do not touch Qutenza, treatment areas, and all used supplies or other materials placed in contact with the treatment area without wearing nitrile gloves.
- •
- Wear nitrile gloves at all times while handling Qutenza and cleaning treatment areas. Do NOT use latex gloves.
- •
- Do not hold Qutenza near eyes or mucous membranes.
- •
- Immediately after use, dispose of used and unused patches, patch clippings, unused Cleansing Gel and associated treatment supplies in accordance with local biomedical waste procedures.
17. Patient Counseling Information
- •
- Inform patients that exposure of the skin to Qutenza may result in transient erythema and burning sensation. Instruct patients not to touch the patch and that if they accidentally touch the Qutenza patch it may burn and/or sting.
- •
- Instruct patients that if irritation of eyes or airways occurs, or if any of the side effects become severe, to notify their doctor immediately.
- •
- Inform patients that the treated area may be sensitive to heat (e.g., hot showers/bath, direct sunlight, vigorous exercise) for a few days following treatment.
- •
- Inform patients that they may be given medication to treat acute pain during and after the Qutenza application procedure. Some of these medications, such as opioids, may affect the ability to perform potentially hazardous activities such as driving or operating machinery.
- •
- Inform patients that as a result of treatment-related increases in pain, small transient increases in blood pressure may occur during and shortly after Qutenza treatment and that blood pressure will be monitored during the treatment procedure. Instruct patients to inform the physician if they have experienced any recent cardiovascular event
- •
- Instruct patients to notify their physician if they are pregnant or breast feeding.
Manufactured for Averitas Pharma, Inc., Morristown, NJ 07960, USA
by Lohmann Therapie-Systeme AG (LTS), Andernach, Germany
Qutenza® is a registered trademark of Averitas Pharma, Inc.
Revised: October 2018
72512-QZ-03
PRINCIPAL DISPLAY PANEL - Carton Label (1 Patch)
NDC 72512-928-01
1 Patch
One single-use patch (179 mg capsaicin)
One single-use 50 g tube of Cleansing Gel
Qutenza®
(capsaicin) 8% patch
Rx only. For topical use only.
Dosage/Instructions for Use: See Full Prescribing Information.
Only a healthcare professional should open the Qutenza® carton or pouch.
Use of nitrile gloves is required when handling Qutenza®. Do not use latex gloves. Qutenza® contains capsaicin and
may cause irritation.
Qutenza® should be administered by a physician or healthcare professional under the close supervision of a physician.
AveritasPharma
Manufactured for Averitas Pharma, Inc, Morristown, NJ 07960, USA by Lohmann Therapie-Systeme AG (LTS) Andernach, Germany © Averitas Pharma, Inc. 2018
For questions about Qutenza® call 1-877-900-6479 Rev. October 2018
Store carton between 20°C and 25°C (68°F and 77°F).
Excursions between 15°C and 30°C (59°F and 86°F) are allowed.
Keep Qutenza® in the sealed pouch until immediately before use.
Store out of the reach of children and pets.
Inactive Ingredients:
Patch: diethylene glycol monoethyl ether, dimethicone, ethyl cellulose, polyester film, silicone adhesive, and white ink
Cleansing Gel: butylated hydroxyanisole, carbomer copolymer, edetate disodium, polyethylene glycol, purified water, and sodium hydroxide
PRINCIPAL DISPLAY PANEL - Carton Label (2 Patches)
NDC 72512-929-01
2 Patches
Two single-use patches (179 mg capsaicin)
One single-use 50 g tube of Cleansing Gel
Qutenza®
(capsaicin) 8% patch
Rx only. For topical use only.
Dosage/Instructions for Use: See Full Prescribing Information.
Only a healthcare professional should open the Qutenza® carton or pouch.
Use of nitrile gloves is required when handling Qutenza®. Do not use latex gloves. Qutenza® contains capsaicin and
may cause irritation.
Qutenza® should be administered by a physician or healthcare professional under the close supervision of a physician.
AveritasPharma
Manufactured for Averitas Pharma, Inc, Morristown, NJ 07960, USA by Lohmann Therapie-Systeme AG (LTS) Andernach, Germany © Averitas Pharma, Inc. 2018
For questions about Qutenza® call 1-877-900-6479 Rev. October 2018
Store carton between 20°C and 25°C (68°F and 77°F).
Excursions between 15°C and 30°C (59°F and 86°F) are allowed.
Keep Qutenza® in the sealed pouch until immediately before use.
Store out of the reach of children and pets.
Inactive Ingredients:
Patch: diethylene glycol monoethyl ether, dimethicone, ethyl cellulose, polyester film, silicone adhesive, and white ink
Cleansing Gel: butylated hydroxyanisole, carbomer copolymer, edetate disodium, polyethylene glycol, purified water, and sodium hydroxide
| QUTENZA
capsaicin kit |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| QUTENZA
capsaicin kit |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Averitas Pharma Inc (081313982) |
Frequently asked questions
More about Qutenza (capsaicin topical)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (6)
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: miscellaneous topical agents
- Breastfeeding
- En español