October 17, 2023
IMPORTANT PRESCRIBING INFORMATION
Subject:
Temporary Importation of Steri-Med Pharma, Inc.'s Erythromycin Ophthalmic Ointment 5mg/g to Address Drug Shortage
Dear Healthcare Provider,
In order to address the current drug shortage of Erythromycin Ophthalmic Ointment, Fera Pharmaceuticals, LLC (Fera), in conjunction with Steri-Med Pharma, Inc. (Steri-Med), is coordinating with the U.S. FDA Drug Shortage Staff to increase its availability in the U.S. market by temporary importation of non-FDA approved product from Canada. Effective immediately and during this temporary period, Fera will distribute the following product:
Table 1: Imported Product Information
|
|
| Product Name and Description
| Size
| Drug Identification Number (DIN)
| National Drug Code (NDC)
|
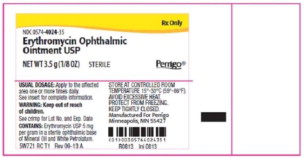
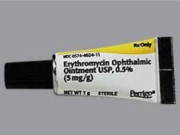
Erythromycin Ophthalmic Ointment USP, 5 mg/g
| 1 g Tube
| 02326663
| 48102-057-11

|
| Lot No.
| Expiry
| |
| 21M02*
| Oct 2024
| | The linear barcode on the imported product may not register accurately on U.S. scanning systems.
A linear NDC barcode is provided above to assist with input by U.S. scanning system.
|
| 21M03*
| Oct 2024
| |
| 21M04*
| Oct 2024
| |
| 21M06*
| Oct 2024
| |
| 21N02*
| Nov 2024
| |
| 22A05*
| Dec 2024
| |
| 22C01*
| Feb 2025
| |
| 22C02*
| Feb 2025
| |
| 22C04
| Feb 2025
| |
| 22C06
| Feb 2025
| |
| 22E01
| Mar 2025
| |
| 22E03
| Mar 2025
| |
|
| | |
This supply of Erythromycin Ophthalmic Ointment is approved and marketed in Canada, under DIN 02326663, and manufactured by Steri-Med (previously known as Sterigen), a facility licensed by Health Canada. At this time, no other entity except Steri-Med or its distributor Fera is authorized by the FDA to import or distribute Steri-Med's Erythromycin Ophthalmic Ointment in the U.S.
It is important to note the following:
- The strength and qualitative composition of the imported drug product are the same as the FDA- approved drug product. Both also meet the USP monograph specification.
-
The linear barcode on the imported product label may not register accurately on U.S. scanning systems. Institutions should manually input the product information into their systems to confirm that barcode systems do not provide incorrect information when the product is scanned. Alternative procedures should be followed to assure that the correct drug product is being used and administered to individual patients. The linear barcode is provided in the Table 1 above and Appendix 1 to assist with input of product information in the institutional setting.
In addition, the carton of the imported product does not include a product identifier as required under the Drug Supply Chain Security Act (DSCSA). Specifically, each package does not include the NDC, unique serial number, lot number, and expiration date in both human-readable and a two-dimensional data matrix barcode. Additionally, the imported product may not be accompanied with DSCSA product tracing documentation (transaction information, transaction history, and transaction statement).
- The drug product being distributed by Fera Pharmaceuticals, LLC is approved in Canada. The Prescribing Information (PI) and Patient Medication Information (Medication Guide) reviewed and approved by Health Canada differ from the full prescribing information for the current U.S. FDA Reference Standard (RS). The U.S. FDA label does not contain a Medication Guide.
In particular, the FDA label provides more detailed information regarding DESCRIPTION, CLINICAL PHARMACOLOGY, USAGE during pregnancy and in infants, and in PRECAUTIONS.
Please refer to the package insert for the U.S. FDA-approved Erythromycin Ophthalmic Ointment USP, 5 mg/g for full prescribing information.
Table 3. Comparison Between the FDA-approved vs. Steri-Med Prescribing Information
PACKAGE INSERT
SECTION | FDA-Approved RS Label | Steri-Med Label |
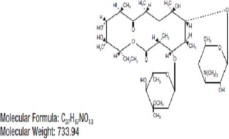
| DESCRIPTION | Erythromycin Ophthalmic Ointment belongs to the macrolide group of antibiotics. It is basic and readily forms a salt when combined with an acid. The base, as crystals or powder, is slightly soluble in water, moderately soluble in ether, and readily soluble in alcohol or chloroform. Erythromycin ((3R*,4S*,5S*,6R*,7R*,9R*,11R*,12R*,13S*,14R*)- 4-[(2,6-dideoxy-3-C-methyl-3-0-methyl-α-L-ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy-3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-ß-D-xylo-hexopyranosyl]oxy]oxacyclotetradecane-2,10-dione) is antibiotic produced from a strain of Streptomyces erythraeus. It has the following structural formula:
Each gram contains Erythromycin USP 5 mg in a sterile ophthalmic base of mineral oil and white petrolatum.
| THERAPEUTIC CLASSIFICATION
Antibiotic
|
CLINICAL
PHARMACOLOGY | Microbiology
Erythromycin inhibits protein synthesis without affecting nucleic acid synthesis. Erythromycin is usually active against the following organisms in vitro and in clinical infections:
Streptococcus pyogenes (group A ß-hemolytic) Alpha-hemolytic streptococci (viridans group)
Staphylococcus aureus, including penicillinase-producing strains (methicillin-resistant staphylococci are uniformly resistant to erythromycin)
Streptococcus pneumonia
Mycoplasma pneumoniae (Eaton Agent, PPLO)
Haemophilus influenzae (not all strains of this organism are susceptible at the erythromycin concentrations ordinarily achieved)
Treponema pallidum
Corynebacterium diphtheriae
Neisseria gonorrhoeae
Chlamydia trachomatis
| Erythromycin inhibits protein synthesis by binding to the 50S ribosomal subunit within the microorganism. It is usually bacteriostatic but may be bactericidal depending on the sensitivity and number of organisms and the concentrations of the drug. Its spectrum of activity is similar to that of penicillin G. Resistance to erythromycin of some strains of H. influenza and Staphylococci has been demonstrated.
|
| INDICATIONS AND USAGE | For the treatment of superficial ocular infections involving the conjunctiva and/or cornea caused by organisms susceptible to erythromycin.
For prophylaxis of ophthalmia neonatorum due to N. gonorrhoeae or C. trachomatis. The effectiveness of erythromycin in the prevention of ophthalmia caused by penicillinase-producing N. gonorrheae is not established.
For infants born to mothers with clinically apparent gonorrhea, intravenous or intramuscular injections of aqueous crystalline penicillin G should be given; a single dose of 50,000 units for term infants or 20,000 units for infants of low birth weight. Topical prophylaxis alone is inadequate for these infants.
| For the treatment of superficial ocular infections involving the conjunctiva and/or cornea caused by organisms susceptible to erythromycin.
For prophylaxis of ophthalmic neonatorum due to N. gonorrhoeae or C. trachomatis. The Canadian Pediatric Society, the Centers for Diseases Control and the Committee on Drugs (US), the Committee of Fetus and Newborn and the Committee on Infectious Diseases of the American Academy of Pediatrics recommend 1% silver nitrate solution in single dose ampoules or single use tubes of an ophthalmic ointment containing 0.5% erythromycin or 1% tetracycline as effective and acceptable regimens for prophylaxis of gonococcal ophthalmia neonatorum. Erythromycin ophthalmic ointment has also been effective for prevention of neonatal conjunctivitis due to C. trachomatis, a condition that may develop one to several weeks after delivery in infants of mothers whose birth canal harbor the organism.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ERYTHROMYCIN ophthalmic ointment and other antibacterial drugs, ERYTHROMYCIN ophthalmic ointment should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
|
CONTRAINDICATIONS:
| This drug is contraindicated in patients with a history of hypersensitivity to erythromycin.
| Erythromycin and its derivatives should not be used in patients with known hypersensitivity to these drugs or any ingredient in the formulations or with infections that are resistant to the drug (primarily certain Staphylococci organisms).
|
| PRECAUTIONS | General:
The use of antimicrobial agents may be associated with the overgrowth of nonsusceptible organisms including fungi; in such a case, antibiotic administration should be stopped and appropriate measures taken.
Information for Patients:
Avoid contaminating the applicator tip with material from the eye, fingers, or other source.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Two year oral studies conducted in rats with erythromycin did not provide evidence of tumorigenicity. Mutagenicity studies have not been conducted.
No evidence of impaired fertility that appeared related to erythromycin was reported in animal studies.
Pregnancy:
Teratogenic effects
Pregnancy Category B.
Reproduction studies have been performed in rats, mice, and rabbits using erythromycin and its various salts and esters, at doses that were several multiples of the usual human dose. No evidence of harm to the fetus that appeared related to erythromycin was reported in these studies. There are, however, no adequate and well controlled studies in pregnant women. Because animal reproductive studies are not always predictive of human response, the erythromycins should be used during pregnancy only if clearly needed.
Nursing Mothers:
Caution should be exercised when erythromycin is administered to a nursing woman.
Pediatric Use:
See INDICATIONS AND USAGE and DOSAGE AND ADMINISTRATION. | WARNINGS AND PRECAUTIONS
Susceptibility/Resistance
Prescribing ERYTHROMYCIN ophthalmic ointment in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and risks the development of drug-resistant bacteria.
The rare possibility of superinfection caused by overgrowth of non-susceptible bacteria or fungi should be kept in mind during prolonged or repeated therapy, especially when other antibacterial agents are simultaneously employed. In such instances the drug should be withdrawn and appropriate treatment instituted if necessary.
|
| ADVERSE REACTIONS | The most frequently reported adverse reactions are minor ocular irritations, redness, and hypersensitivity reactions.
To report SUSPECTED ADVERSE REACTIONS, contact Perrigo at
1-866-634-9120 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
| ADVERSE EFFECTS
Serious allergic reactions to erythromycin have been extremely infrequent. Mild allergic reactions, such as urticaria and morbilliform skin rashes have occurred. Should a patient demonstrate signs of hypersensitivity, administer appropriate countermeasures such as epinephrine, corticosteroid and antihistamines, and withdraw the antibiotic.
|
| DOSAGE AND ADMINISTRATION | In the treatment of superficial ocular infections, a ribbon approximately 1 cm in length of Erythromycin Ophthalmic Ointment should be applied directly to the infected structure up to 6 times daily, depending on the severity of the infection.
For prophylaxis of neonatal gonococcal or chlamydial conjunctivitis, a ribbon of ointment approximately 1 cm in length should be instilled into each lower conjunctival sac. The ointment should not be flushed from the eye following instillation. A new tube should be used for each infant.
| In the treatment of external ocular infections, apply the ointment directly to the infected structure one or more times daily, depending on the severity of the infection.
For prophylaxis neonatal gonococcal or chlamydial conjunctivitis, a ribbon of ointment approximately 0.5 to 1 cm in length should be instilled into each conjunctival sac. The ointment should not be flushed from the eye following installation. A new tube should be used for each infant. Infants born by cesarean section as well as those delivered by vaginal route should receive prophylaxis.
|
To order, please call (414) 434-6604. For additional questions about the information contained in this letter, please contact Fera at (516) 277-1449.
Healthcare providers and patients are encouraged to report adverse events in patients using imported Erythromycin Ophthalmic Ointment to Fera at (414) 434-6604.
Adverse events or quality problems experienced with the use of this product may also be reported to the FDA's MedWatch Adverse Event Reporting Program either online, by regular mail, or by fax:
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
-
Regular mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178 (1-800-332-0178).
Sincerely,
Michelle Kim, PharmD.
Sr. Director of Regulatory Affairs
Appendix 1: Linear Barcode for U.S. NDC Scanning
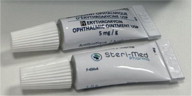
| Product Name
| Erythromycin Ophthalmic Ointment USP, 5 mg/g
|
| Manufacturer
| Steri-Med Pharma, Inc. (formerly Sterigen)
|
| Size
| 1 g Tube
|
| NDC
| 48102-057-11
|
| Linear Barcode
|  |
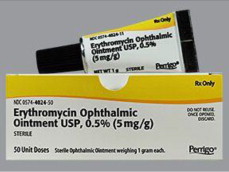
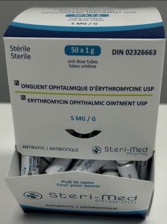
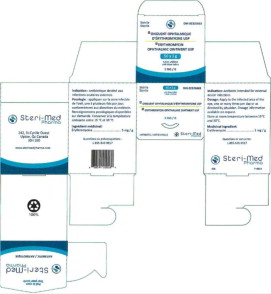
Principal Display Panel - 5 mg Carton Label (Steri-Med)
Sterile
Sterile
50 x 1g
unit dose tubes
tubes unidose
DIN 02326663
Pr ONGUENT OPHTALMIQUE D'ERYTHROMYCINE USP
Pr ERYTHROMYCIN OPHTHALMIC OINTMENT USP
5 MG/G
ANTIBIOTIC / ANTIBIOTIQUE
Steri-Med
Pharma
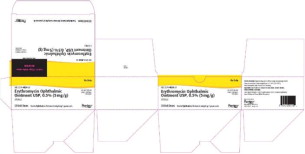
Principal Display Panel - 5 mg Carton Label (Sterigen)
STERILE/STERILE
50 x 1g
unit dose tubes
tubes unidose
DIN 02326663
Pr Erythromycin Ophthalmic Ointment USP
Pr Onguent Ophtalmique d'Erythromycine USP
5 mg/g
ANTIBIOTIC / ANTIBIOTIQUE
sterigen