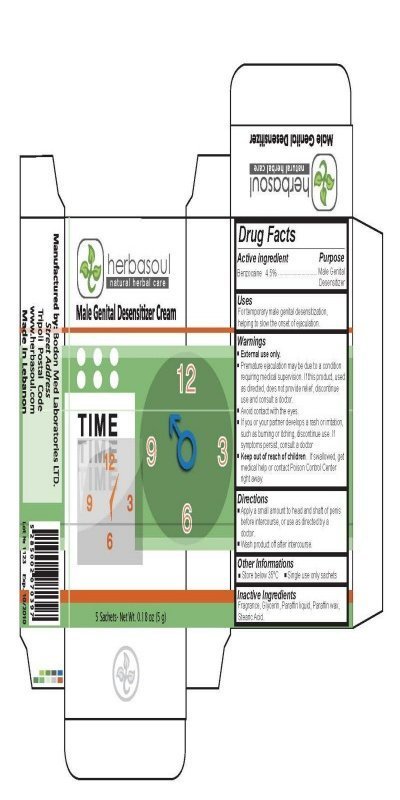
Herbasoul Male Genital Desensitizer Cream
Dosage form: cream
Ingredients: BENZOCAINE 4.5g in 100g
Labeler: Bodon Med Laboratories Ltd.
NDC code: 50664-100
Medically reviewed by Drugs.com. Last updated on Feb 13, 2025.
Active Ingredient Purpose
Benzocaine 4.5 percent .......................................................................Male Genital
Desensitizer
Uses
For temporary male genital desensitization, helping to slow the onset of ejaculation.
Warnings
External use only.
Premature ejaculation may be due to a condition requiring medical supervision.
If this product, used as directed, does not provide relief, discontinue use. If symptoms persist,consult a doctor.
Keep out of reach of children.
If swallowed, get medical help or contact Poison Control Center right away.
Directions
Apply a small amount to head and shaft of penis before intercourse,
or use as directed by a doctor.
Wash product off after intercourse.
Other Informations
Store below 35 celius
Single use only sachets
Inactive Ingredients
Fragrance, Glycerin, Paraffin Liquid, Paraffin wax, Stearic Acid.
Manufactured By: Bodon Med Laboratories LTD.
Street Address
Tripoli Postal Code
www.herbasoul.com
Made in Lebanon
This unit not labeled for retail sale
Herbasoul
Natural Herbal Care
Male Genital Desensitizer Cream
Net Wt. 0.036 oz (1 g)

| HERBASOUL MALE GENITAL DESENSITIZER CREAM
benzocaine cream |
|||||||||||||
|
|||||||||||||
|
|||||||||||||
|
|||||||||||||
|
|||||||||||||
|
|||||||||||||
| Labeler - Bodon Med Laboratories Ltd. (557643207) |
| Registrant - Bodon Med Laboratories Ltd. (557643207) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Bodon Med Laboratories Ltd. | 557643207 | manufacture | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
