X 10 Pill - yellow & white round, 8mm
Generic Name: alfuzosin
Pill with imprint X 10 is Yellow & White, Round and has been identified as Uroxatral 10 mg. It is supplied by Sanofi-Aventis.
Uroxatral is used in the treatment of Benign Prostatic Hyperplasia and belongs to the drug class alpha-adrenoreceptor antagonists. There is no proven risk in humans during pregnancy. Uroxatral 10 mg is not a controlled substance under the Controlled Substances Act (CSA).
Images for X 10

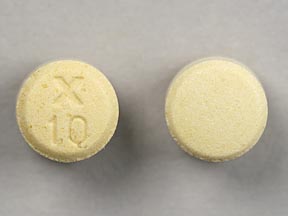


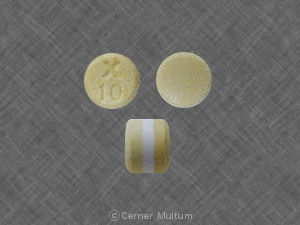
Uroxatral
- Generic Name
- alfuzosin
- Imprint
- X 10
- Strength
- 10 mg
- Color
- Yellow & White
- Size
- 8.00 mm
- Shape
- Round
- Availability
- Prescription only
- Drug Class
- Alpha-adrenoreceptor antagonists
- Pregnancy Category
- B - No proven risk in humans
- CSA Schedule
- Not a controlled drug
- Labeler / Supplier
- Sanofi-Aventis
- National Drug Code (NDC)
- 00024-4200 (Discontinued)
- Inactive Ingredients
-
silicon dioxide,
ethylcellulose (100 mPa.s),
hydrogenated castor oil,
hypromelloses,
magnesium stearate,
mannitol,
microcrystalline cellulose,
povidone,
ferric oxide yellow
Note: Inactive ingredients may vary.
Related images for "X 10"
More about Uroxatral (alfuzosin)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (21)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- Drug class: alpha-adrenoreceptor antagonists
- En español
Patient resources
Professional resources
Related treatment guides
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.