Welchol Prescribing Information
Package insert / product label
Generic name: colesevelam hydrochloride
Dosage form: tablet, film coated, oral suspension
Drug class: Bile acid sequestrants
Medically reviewed by Drugs.com. Last updated on Jun 10, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
WELCHOL (colesevelam hydrochloride) tablets, for oral use
WELCHOL (colesevelam hydrochloride) for oral suspension
Initial U.S. Approval: 2000
Indications and Usage for Welchol
WELCHOL is a bile acid sequestrant indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia (1.1).
- reduce LDL-C levels in boys and postmenarchal girls, 10 to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification (1.1).
- improve glycemic control in adults with type 2 diabetes mellitus (1.2).
Limitations of Use (1.3):
- Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis.
- Not studied in Fredrickson Type I, III, IV, and V dyslipidemias
Welchol Dosage and Administration
- Obtain lipid parameters, including serum triglyceride (TG) levels, before starting WELCHOL (2.1).
- The recommended dosage for adults and for boys and postmenarchal girls aged 10 to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage for adults with type 2 diabetes mellitus is 3.75 grams daily. WELCHOL should be taken as follows (2.2, 2.4):
Tablets
Take 6 tablets once daily or 3 tablets twice daily with a meal and liquid.
For Oral Suspension
Take one packet once daily with a meal. To prepare, empty the entire contents of one packet into a glass or cup. Add 1 cup of water, fruit juice, or diet soft drinks. Stir well and drink.
Contraindications
Warnings and Precautions
- Hypertriglyceridemia and Pancreatitis: WELCHOL can increase TG. Hypertriglyceridemia can cause acute pancreatitis. Monitor lipids, including TG. Instruct patients to discontinue WELCHOL and seek prompt medical attention if the symptoms of acute pancreatitis occur (5.1).
- Gastrointestinal Obstruction: Cases of bowel obstruction have occurred. WELCHOL is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction (5.2).
- Vitamin K or Fat-Soluble Vitamin Deficiencies: WELCHOL may decrease absorption of fat-soluble vitamins. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk. Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to WELCHOL (5.3).
- Drug Interactions: Due to the potential for decreased absorption of other drugs that have not been tested for interaction, consider administering at least 4 hours prior to WELCHOL (5.4, 7, 12.3).
- Risks in Patients with Phenylketonuria (PKU): Phenylalanine can be harmful to patients with phenylketonuria. WELCHOL for oral suspension contains 27 mg phenylalanine per 3.75 gram packet (5.5, 11).
Adverse Reactions/Side Effects
In clinical trials, the most common (incidence ≥2% and greater than placebo) adverse reactions with WELCHOL included constipation, dyspepsia, and nausea (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Daiichi Sankyo, Inc. at 1-877-437-7763 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Concomitant use with WELCHOL may decrease the exposure of the following drugs: Drugs with a narrow therapeutic index (e.g., cyclosporine), phenytoin, thyroid hormone replacement therapy, warfarin, oral contraceptives containing ethinyl estradiol and norethindrone, olmesartan medoxomil, and sulfonylureas (glimepiride, glipizide, glyburide). Administer these drugs 4 hours prior to WELCHOL. For patients on warfarin, monitor International Normalized Ratio (INR) frequently during initiation then periodically (7.1).
Concomitant use with WELCHOL may increase the exposure of the following drugs: Metformin extended release. Monitor patients' glycemic control (7.2).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 10/2021
Full Prescribing Information
1. Indications and Usage for Welchol
1.1 Primary Hyperlipidemia
WELCHOL is indicated as an adjunct to diet and exercise to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia.
WELCHOL is indicated to reduce LDL-C levels in boys and postmenarchal girls, 10 to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH) who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification.
2. Welchol Dosage and Administration
2.1 Testing Prior to Initiation of WELCHOL
Obtain lipid parameters, including triglyceride (TG) levels, before starting WELCHOL. WELCHOL is contraindicated in patients with TG levels >500 mg/dL [see Contraindications (4) and Warnings and Precautions (5.1)].
2.2 Recommended Dosage in Primary Hyperlipidemia and Type 2 Diabetes Mellitus
The recommended dosage of WELCHOL for adults and for boys and postmenarchal girls aged 10 to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage of WELCHOL for adults with type 2 diabetes mellitus is 3.75 grams daily. WELCHOL should be taken as follows:
2.3 Important Dosing Information for Primary Hyperlipidemia
WELCHOL can be dosed at the same time as a statin, or WELCHOL and the statin can be dosed apart. Monitor lipid levels within 4 to 6 weeks after initiation of WELCHOL.
2.4 Administration Instructions
Tablets
Take WELCHOL tablets with a meal and liquid. For patients with difficulty swallowing tablets, use WELCHOL for oral suspension [see Warnings and Precautions (5.2)].
For Oral Suspension
To prepare, empty the entire contents of one packet into a glass or cup. Add 1 cup (8 ounces) of water, fruit juice, or diet soft drinks. Stir well and drink. Take WELCHOL oral suspension with meals. Do not take WELCHOL oral suspension in its dry form. Due to tablet size, WELCHOL for oral suspension is recommended for use in the pediatric population.
3. Dosage Forms and Strengths
- Tablets: 625 mg tablets are off-white, oval, film-coated and imprinted with "Sankyo" and "C01" on one side.
- For Oral Suspension: 3.75 gram packet containing a white to pale yellow powder with yellow granules.
4. Contraindications
WELCHOL is contraindicated in patients with:
- Serum TG concentrations >500 mg/dL [see Warnings and Precautions (5.1)]
- History of hypertriglyceridemia-induced pancreatitis [see Warnings and Precautions (5.1)]
- A history of bowel obstruction [see Warnings and Precautions (5.2)]
5. Warnings and Precautions
5.1 Hypertriglyceridemia and Pancreatitis
WELCHOL, like other bile acid sequestrants, can increase serum TG concentrations. Hypertriglyceridemia can cause acute pancreatitis.
WELCHOL had effects on serum TG (median increase 5% compared to placebo) in trials of patients with primary hyperlipidemia.
In trials in patients with type 2 diabetes, greater increases in TG levels occurred when WELCHOL was used as monotherapy (median increase 9.7% compared to placebo) and when WELCHOL was used in combination with pioglitazone (median increase 11% compared to placebo in combination with pioglitazone), sulfonylureas (median increase 18% compared to placebo in combination with sulfonylureas), and insulin (median increase 22% compared to placebo in combination with insulin) [see Adverse Reactions (6.1)].
Obtain lipid parameters, including TG levels, before starting WELCHOL and periodically thereafter. WELCHOL is contraindicated in patients with TG levels >500 mg/dL or patients with a history of hypertriglyceridemia-induced pancreatitis [see Contraindications (4)]. Patients with TG levels greater than 300 mg/dL could have greater increases in serum TG levels with WELCHOL and may require additional TG monitoring. Instruct patients to discontinue WELCHOL and seek prompt medical attention if the symptoms of acute pancreatitis occur (e.g., severe abdominal pain with or without nausea and vomiting). Discontinue WELCHOL if TG levels exceed 500 mg/dL [see Adverse Reactions (6.1)].
5.2 Gastrointestinal Obstruction
Postmarketing cases of bowel obstruction have occurred with WELCHOL [see Adverse Reactions (6.2)]. Because of its constipating effects, WELCHOL is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction. WELCHOL is contraindicated in patients with a history of bowel obstruction [see Contraindications (4)]. Instruct patients to promptly discontinue WELCHOL and seek medical attention if severe abdominal pain or severe constipation occurs.
Because of the tablet size, WELCHOL tablets can cause dysphagia or esophageal obstruction. For patients with difficulty swallowing tablets, use WELCHOL for oral suspension.
5.3 Vitamin K or Fat-Soluble Vitamin Deficiencies
WELCHOL may decrease the absorption of fat-soluble vitamins A, D, E, and K. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk when taking WELCHOL.
Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to WELCHOL [see Drug Interactions (7.1)].
5.4 Drug Interactions
WELCHOL reduces gastrointestinal absorption of some drugs. Administer drugs with a known interaction at least 4 hours prior to WELCHOL [see Drug Interactions (7)].
Due to the potential for decreased absorption of other drugs that have not been tested for interaction, especially those with a narrow therapeutic index, consider administering at least 4 hours prior to WELCHOL [see Clinical Pharmacology (12.3)].
5.5 Risks in Patients with Phenylketonuria (PKU)
Phenylalanine can be harmful to patients with PKU. WELCHOL for oral suspension contains phenylalanine, a component of aspartame. Each 3.75 gram packet contains 27 mg of phenylalanine. Before prescribing WELCHOL for oral suspension to a patient with PKU, consider the combined daily amount of phenylalanine from all sources, including WELCHOL for oral suspension.
6. Adverse Reactions/Side Effects
The following important adverse reactions are described below and elsewhere in the labeling:
- Hypertriglyceridemia and Pancreatitis [see Warnings and Precautions (5.1)]
- Gastrointestinal Obstruction [see Warnings and Precautions (5.2)]
- Vitamin K or Fat-Soluble Vitamin Deficiencies [see Warnings and Precautions (5.3)]
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in clinical studies of another drug and may not reflect the rates observed in practice.
Primary Hyperlipidemia
In 7 double-blind, placebo-controlled clinical trials, 807 patients with primary hyperlipidemia (age range 18-86 years, 50% women, 90% Caucasians, 7% Blacks, 2% Hispanics, 1% Asians) and elevated LDL-C were treated with WELCHOL 1.5 g/day to 4.5 g/day from 4 to 24 weeks (total exposure 199 patient-years).
| WELCHOL N=807 | Placebo N=258 |
|
|---|---|---|
| Constipation | 11.0% | 7.0% |
| Dyspepsia | 8.3% | 3.5% |
| Nausea | 4.2% | 3.9% |
| Accidental injury | 3.7% | 2.7% |
| Asthenia | 3.6% | 1.9% |
| Pharyngitis | 3.2% | 1.9% |
| Flu syndrome | 3.2% | 3.1% |
| Rhinitis | 3.2% | 3.1% |
| Myalgia | 2.1% | 0.4% |
Pediatric Patients 10 to 17 Years of Age
In an 8-week double-blind, placebo-controlled study, boys and post-menarchal girls, 10 to 17 years of age, with HeFH (n=194), were treated with WELCHOL tablets (1.9-3.8 g, daily) or placebo tablets.
| WELCHOL N=129 | Placebo N=65 |
|
|---|---|---|
| Nasopharyngitis | 6.2% | 4.6% |
| Headache | 3.9% | 3.1% |
| Fatigue | 3.9% | 1.5% |
| Creatine Phosphokinase Increase | 2.3% | 0.0% |
| Rhinitis | 2.3% | 0.0% |
| Vomiting | 2.3% | 1.5% |
The reported adverse reactions during the additional 18-week open-label treatment period with WELCHOL 3.8 g per day were similar to those during the double-blind period and included headache (7.6%), nasopharyngitis (5.4%), upper respiratory tract infection (4.9%), influenza (3.8%), and nausea (3.8%).
Type 2 Diabetes Mellitus
In 5 add-on combination and 1 monotherapy double-blind, 12- to 26-week, placebo-controlled clinical trials in patients with type 2 diabetes mellitus, 1022 patients were treated with WELCHOL. The mean exposure duration was 20 weeks (total exposure 393 patient-years). Patients were to receive 3.8 grams of WELCHOL per day. The mean age of patients was 55.7 years, 52.8 percent of the population was male and 61.9% were Caucasian, 4.8% were Asian, and 15.9% were Black or African American. At baseline the population had a mean hemoglobin A1c (HbA1c) of 8.2%, and 26% had past medical history suggestive of microvascular complications of diabetes.
Table 3 shows adverse reactions associated with the use of WELCHOL in patients with type 2 diabetes. These adverse reactions were not present at baseline, occurred more commonly on WELCHOL than on placebo, and occurred in at least 2% of patients treated with WELCHOL.
| WELCHOL N=1022 | Placebo N=1010 |
|
|---|---|---|
| Constipation | 6.5% | 2.2% |
| Hypoglycemia | 3.4% | 3.1% |
| Dyspepsia | 2.8% | 1.0% |
| Nausea | 2.6% | 1.6% |
| Hypertension | 2.6% | 1.9% |
| Back Pain | 2.3% | 1.3% |
A total of 5.3% of WELCHOL-treated patients and 3.6% of placebo-treated patients were discontinued from the diabetes trials due to adverse reactions. This difference was driven mostly by gastrointestinal adverse reactions such as abdominal pain and constipation.
One patient in the add-on to sulfonylurea trial discontinued due to body rash and mouth blistering that occurred on the first day of dosing of WELCHOL, which may represent a hypersensitivity reaction to WELCHOL.
Hypertriglyceridemia
Patients with fasting serum TG levels above 500 mg/dL were excluded from the diabetes clinical trials. In the diabetes trials, 1292 (67.7%) patients had baseline fasting serum TG levels less than 200 mg/dL, 426 (22.3%) had baseline fasting serum TG levels between 200 and less than 300 mg/dL, 175 (9.2%) had baseline fasting serum TG levels between 300 and 500 mg/dL, and 16 (0.8%) had fasting serum TG levels greater than or equal to 500 mg/dL. The median baseline fasting TG concentration for the study population was 160 mg/dL; the median post-treatment fasting TG was 180 mg/dL in the WELCHOL group and 162 mg/dL in the placebo group. WELCHOL therapy resulted in a median placebo-corrected increase in serum TG of 9.7% (p=0.03) in the monotherapy study and of 5% (p=0.22), 11% (p<0.001), 18% (p<0.001), and 22% (p<0.001), when added to metformin, pioglitazone, sulfonylureas, and insulin, respectively. In comparison, WELCHOL resulted in a median increase in serum TG of 5% compared to placebo (p=0.42) in a 24-week monotherapy lipid-lowering trial.
Fasting TG concentrations ≥500 mg/dL occurred in 0.9% of WELCHOL-treated patients compared to 0.7% of placebo-treated patients in the diabetes trials. Among these patients, the TG concentrations with WELCHOL (median 606 mg/dL; interquartile range 570-794 mg/dL) were similar to that observed with placebo (median 663 mg/dL; interquartile range 542-984 mg/dL). Five (0.6%) patients on WELCHOL and 3 (0.3%) patients on placebo developed TG elevations ≥1000 mg/dL.
Cardiovascular Adverse Reactions
During the diabetes trials, the incidence of patients with serious adverse reactions involving the cardiovascular system was 2.2% (22/1022) in the WELCHOL group and 1% (10/1010) in the placebo group. These overall rates included disparate events (e.g., myocardial infarction, aortic stenosis, and bradycardia); therefore, the significance of this imbalance is unknown.
6.2 Post-marketing Experience
The following additional adverse reactions have been identified during post-approval use of WELCHOL. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse Reactions Resulting from Drug Interactions [see Drug Interactions (7)]: Increased seizure activity or decreased phenytoin levels in patients receiving phenytoin, reduced International Normalized Ratio (INR) in patients receiving warfarin therapy, and elevated thyroid-stimulating hormone (TSH) in patients receiving thyroid hormone replacement therapy
Gastrointestinal: Bowel obstruction (in patients with a history of bowel obstruction or resection), dysphagia or esophageal obstruction (occasionally requiring medical intervention), fecal impaction, pancreatitis, abdominal distension, exacerbation of hemorrhoids, and increased transaminases
Laboratory Abnormalities: Hypertriglyceridemia
7. Drug Interactions
7.1 WELCHOL Drug Interactions that Decrease the Exposure of the Concomitant Medication
Table 4 includes a list of drugs that decrease exposure of the concomitant medication when administered concomitantly with WELCHOL and instructions for preventing or managing them.
| Drugs with a Narrow Therapeutic Index | |
| Clinical Impact: | Concomitant use with WELCHOL may decrease the exposure of the narrow therapeutic index drug. In vivo drug interactions studies showed a decrease in exposure of cyclosporine when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer the narrow therapeutic index drug at least 4 hours prior to WELCHOL. Monitor drug levels when appropriate. |
| Examples: | Cyclosporine |
| Phenytoin | |
| Clinical Impact: | There have been postmarketing reports of increased seizure activity or decreased phenytoin levels in patients receiving phenytoin [see Adverse Reactions (6.2)]. |
| Intervention: | Administer phenytoin 4 hours prior to WELCHOL. |
| Thyroid Hormone Replacement Therapy | |
| Clinical Impact: | In vivo drug interactions studies showed a decrease in exposure of levothyroxine when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. There have been postmarketing reports of elevated thyroid-stimulating hormone (TSH) in patients receiving thyroid hormone replacement therapy [see Adverse Reactions (6.2)]. |
| Intervention: | Administer thyroid hormone replacement therapy 4 hours prior to WELCHOL. |
| Warfarin | |
| Clinical Impact: | There have been postmarketing reports of reduced INR in patients receiving warfarin therapy [see Adverse Reactions (6.2)]. |
| Intervention: | Monitor INR frequently during WELCHOL initiation then periodically thereafter. |
| Oral Contraceptives Containing Ethinyl Estradiol and Norethindrone | |
| Clinical Impact: | In vivo drug interactions studies showed a decrease in exposure of ethinyl estradiol and norethindrone when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer oral contraceptives containing ethinyl estradiol and norethindrone 4 hours prior to WELCHOL. |
| Olmesartan Medoxomil | |
| Clinical Impact: | In vivo drug interactions studies showed a decrease in olmesartan medoxomil when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer olmesartan medoxomil 4 hours prior to WELCHOL. |
| Sulfonylureas | |
| Clinical Impact: | In vivo drug interactions studies showed a decrease in sulfonylureas when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer sulfonylureas 4 hours prior to WELCHOL. |
| Examples: | Glimepiride, glipizide, and glyburide |
| Oral Vitamin Supplements | |
| Clinical Impact: | WELCHOL may decrease the absorption of fat-soluble vitamins A, D, E, and K [see Warnings and Precautions (5.3)]. |
| Intervention: | Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to WELCHOL. |
7.2 WELCHOL Drug Interactions that Increase the Exposure of the Concomitant Medication
| Metformin Extended Release (ER) | |
| Clinical Impact: | In vivo drug interactions studies showed an increase in metformin extended release (ER) when coadministered with WELCHOL [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients' glycemic control. |
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
WELCHOL is not absorbed systemically following oral administration, and maternal use is not expected to result in fetal exposure to the drug. Limited available data on the use of WELCHOL are insufficient to determine a drug-associated risk of major congenital malformations or miscarriage. In animal reproduction studies, no evidence of either maternal or fetal toxicity was found in rats or rabbits exposed to colesevelam hydrochloride during the period of fetal organogenesis at 8 and 5 times, respectively, the maximum recommended human dose (MRHD) of 3.75 g/day, based on body surface area (mg/m2). No adverse effects on offspring survival and development were observed in rats administered 5 times the MRHD (see Data). WELCHOL may decrease the absorption of fat-soluble vitamins [see Warnings and Precautions (5.3)]. There are no data available on the effect of colesevelam hydrochloride on the absorption of fat-soluble vitamins in pregnant women. If the patient becomes pregnant while taking WELCHOL, the patient should be advised of the lack of known clinical benefit with continued use during pregnancy.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
There are no adequate and well-controlled studies of colesevelam hydrochloride use in pregnant women. In the postmarketing setting there have been infrequent reports of pregnancy with use of WELCHOL and a causal association with congenital anomalies has not been established.
Animal Data
In pregnant rats given dietary doses of 0.3, 1.0, 3.0 g/kg/day colesevelam hydrochloride from gestation days 7 through 17, no teratogenic effects were observed. Exposures at 3.0 g/kg/day were 8 times the human exposure at 3.75 g/day MRHD, based on body surface area (mg/m2).
In pregnant rabbits given oral gavage doses of 0.1, 0.5, 1.0 g/kg/day colesevelam hydrochloride from gestation days 6 through 18, no teratogenic effects were observed. Exposures at 1.0 g/kg/day were 5 times the human exposure at 3.75 g/day MRHD, based on body surface area (mg/m2).
In pregnant rats given oral gavage doses of 0.1, 0.3, 1.0 g/kg/day colesevelam hydrochloride from gestation day 6 through lactation day 21 (weaning), no adverse effects on survival and development were observed. Exposures at 1.0 g/kg/day were 5 times the human exposure at 3.75 g/day MRHD, based on body surface area (mg/m2).
8.3 Females and Males of Reproductive Potential
Contraception
Use of WELCHOL may reduce the efficacy of oral contraceptives. Advise patients to take oral contraceptives at least 4 hours prior to taking WELCHOL [see Drug Interactions (7)].
8.4 Pediatric Use
Primary Hyperlipidemia
The safety and effectiveness of WELCHOL to reduce LDL-C levels in boys and postmenarchal girls 10 to 17 years of age with HeFH who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification have been established. Use of WELCHOL for this indication is supported by a study in 129 WELCHOL-treated pediatric patients aged 10 to 17 years with HeFH [see Clinical Studies (14.1)]. Adverse reactions commonly observed in pediatric patients compared to placebo, but not in adults, included headache (3.9%), creatine phosphokinase increase (2.3%), and vomiting (2.3%) [see Adverse Reactions (6.1)]. There were no significant effects on fat-soluble vitamin levels or clotting factors in the adolescent boys or girls relative to placebo.
Due to WELCHOL tablet size, WELCHOL for oral suspension is recommended for use in the pediatric population [see Dosage and Administration (2.2, 2.4)]. The safety and effectiveness of WELCHOL in pediatric patients with HeFH less than 10 years of age or in premenarchal females have not been established.
Type 2 Diabetes Mellitus
The safety and effectiveness of WELCHOL to improve glycemic control in pediatric patients with type 2 diabetes mellitus have not been established. Effectiveness was not demonstrated in a 6-month, adequate and well-controlled study conducted in 141 WELCHOL-treated pediatric patients aged 10 to 17 years with type 2 diabetes mellitus.
8.5 Geriatric Use
Primary Hyperlipidemia
Of the 1350 patients enrolled in the hyperlipidemia clinical studies, 349 (26%) were ≥65 years old, and 58 (4%) were ≥75 years old. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Type 2 Diabetes Mellitus
Of the 2048 patients enrolled in the six diabetes studies, 397 (19%) were ≥65 years old, and 36 (2%) were ≥75 years old. In these trials, WELCHOL 3.8 g/day or placebo was added onto background anti-diabetic therapy. No overall differences in safety or effectiveness were observed between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
Type 2 Diabetes Mellitus
Of the 2048 patients enrolled in the six diabetes studies, 807 (39%) had mild renal insufficiency (creatinine clearance [CrCl] 50-<80 mL/min), 61 (3%) had moderate renal insufficiency (CrCl 30-<50 mL/min), and none had severe renal insufficiency (CrCl <30 mL/min), as estimated from baseline serum creatinine using the Modification of Diet in Renal Disease (MDRD) equation. No overall differences in safety or effectiveness were observed between patients with CrCl <50 mL/min (n=53) and those with a CrCl ≥50 mL/min (n=1075) in the add-on to metformin, sulfonylureas, and insulin diabetes studies. In the monotherapy study and add-on to pioglitazone study, only 3 and 5 patients, respectively, had moderate renal insufficiency.
10. Overdosage
WELCHOL is not absorbed and the risk of systemic toxicity is low. Excessive doses of WELCHOL may cause more severe local gastrointestinal effects (e.g., constipation).
11. Welchol Description
WELCHOL (colesevelam hydrochloride) is a non-absorbed, polymeric, lipid-lowering and glucose-lowering agent for oral administration. Colesevelam hydrochloride is a high-capacity bile acid-binding molecule.
Colesevelam hydrochloride is poly(allylamine hydrochloride) cross-linked with epichlorohydrin and alkylated with 1-bromodecane and (6-bromohexyl)-trimethylammonium bromide. The chemical name (IUPAC) of colesevelam hydrochloride is allylamine polymer with 1-chloro-2,3-epoxypropane, [6-(allylamino)-hexyl]trimethylammonium chloride and N-allyldecylamine, hydrochloride. The chemical structure of colesevelam hydrochloride is represented by the following formula:
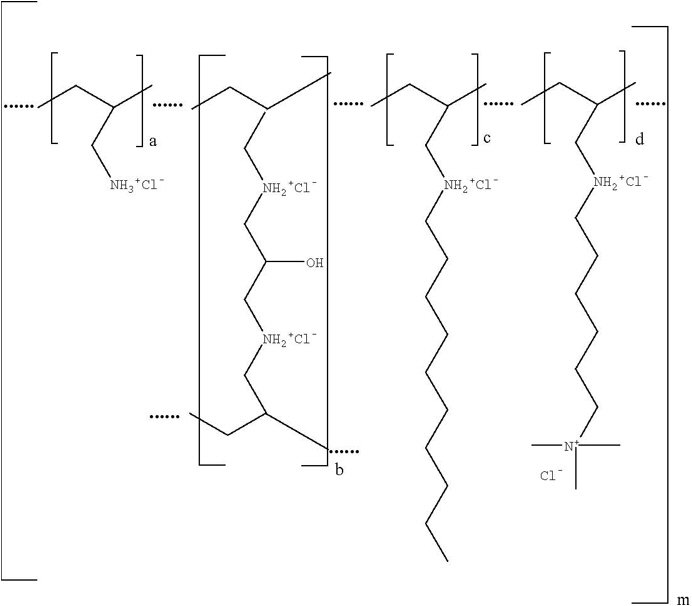
wherein (a) represents allyl amine monomer units that have not been alkylated by either of the 1-bromodecane or (6-bromohexyl)-trimethylammonium bromide alkylating agents or cross-linked by epichlorohydrin; (b) represents allyl amine units that have undergone cross-linking with epichlorohydrin; (c) represents allyl amine units that have been alkylated with a decyl group; (d) represents allyl amine units that have been alkylated with a (6-trimethylammonium) hexyl group, and m represents a number ≥100 to indicate an extended polymer network. A small amount of the amines are dialkylated and are not depicted in the formula above. No regular order of the groups is implied by the structure; cross-linking and alkylation are expected to occur randomly along the polymer chains. A large amount of the amines are protonated. The polymer is depicted in the hydrochloride form; a small amount of the halides are bromide. Colesevelam hydrochloride is hydrophilic and insoluble in water.
WELCHOL tablets are off-white, oval, film-coated, solid tablets each containing 625 mg colesevelam hydrochloride. In addition, each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, silicon dioxide, HPMC (hydroxypropyl methylcellulose), and acetylated monoglyceride. The tablets are imprinted using a water-soluble black ink (<5 calories per 6 tablets).
WELCHOL for oral suspension is a citrus-flavored, white to pale yellow powder containing yellow granules packaged in a packet containing 3.75 gram colesevelam hydrochloride. In addition, each packet contains the following inactive ingredients: lemon flavor, orange flavor, propylene glycol alginate, simethicone, aspartame, citric acid, medium chain triglycerides, and magnesium trisilicate (<5 calories per 3.75 gram single-dose packet). PHENYLKETONURICS: WELCHOL for oral suspension contains 27 mg phenylalanine per 3.75 gram dose.
12. Welchol - Clinical Pharmacology
12.1 Mechanism of Action
Primary Hyperlipidemia: Colesevelam hydrochloride, the active pharmaceutical ingredient in WELCHOL, is a non-absorbed, lipid-lowering polymer that binds bile acids in the intestine, impeding their reabsorption. As the bile acid pool becomes depleted, the hepatic enzyme, cholesterol 7-α-hydroxylase, is upregulated, which increases the conversion of cholesterol to bile acids. This causes an increased demand for cholesterol in the liver cells, resulting in the dual effect of increasing transcription and activity of the cholesterol biosynthetic enzyme, HMG-CoA reductase, and increasing the number of hepatic LDL receptors. These compensatory effects result in increased clearance of LDL-C from the blood, resulting in decreased serum LDL-C levels. Serum TG levels may increase or remain unchanged.
12.2 Pharmacodynamics
A maximum therapeutic response to the lipid-lowering effects of WELCHOL was achieved within 2 weeks and was maintained during long-term therapy. In the diabetes clinical studies, a therapeutic response to WELCHOL, as reflected by a reduction in HbA1c, was initially noted following 4-6 weeks of treatment and reached maximal or near-maximal effect after 12-18 weeks of treatment.
12.3 Pharmacokinetics
Absorption
Colesevelam hydrochloride is a hydrophilic, water-insoluble polymer that is not hydrolyzed by digestive enzymes and is not absorbed.
Distribution
Colesevelam hydrochloride is not absorbed, and therefore, its distribution is limited to the gastrointestinal tract.
Elimination
Drug Interaction Studies
Drug interactions between colesevelam and concomitantly administered drugs were screened through in vitro studies and confirmed in in vivo studies. In vitro studies demonstrated that cephalexin, metformin, and ciprofloxacin had negligible binding to colesevelam hydrochloride. Therefore, an in vivo pharmacokinetic interaction of WELCHOL with these drugs is unlikely. WELCHOL was found to have no significant effect on the bioavailability of aspirin, atenolol, digoxin, enalapril, fenofibrate, lovastatin, metoprolol, phenytoin, pioglitazone, quinidine, rosiglitazone, sitagliptin, valproic acid, and warfarin. The results of additional in vivo drug interactions of WELCHOL are presented in Table 6.
| Drug | Dose | Co-administered | 1 hr prior to WELCHOL | 4 hrs prior to WELCHOL | |||
|---|---|---|---|---|---|---|---|
| AUC0-∞ | Cmax | AUC0-∞ | Cmax | AUC0-∞ | Cmax | ||
| N/A − not available | |||||||
| Cyclosporine | 200 mg | -34% | -44% | N/A | N/A | N/A | N/A |
| Ethinyl Estradiol† | 0.035 mg | -24% | -24% | -18% | -1% | -12% | 0% |
| Glimepiride | 4 mg | -18% | -8% | N/A | N/A | -6% | 3% |
| Glipizide | 20 mg | -12% | -13% | N/A | N/A | -4% | 0% |
| Glyburide | 3 mg | -32% | -47% | -20% | -15% | -7% | 4% |
| Levothyroxine | 600 µg | -22% | -33% | 6% | -2% | 1% | 8% |
| Metformin ER | 1500 mg | 44% | 8% | N/A | N/A | N/A | N/A |
| Norethindrone† | 1 mg | -1% | -20% | 5% | -3% | 6% | 7% |
| Olmesartan Medoxomil | 40 mg | -39% | -28% | N/A | N/A | -15% | -4% |
| Repaglinide | 2 mg | -7% | -19% | -6% | -1% | N/A | N/A |
| Verapamil Sustained Release | 240 mg | -31% | -11% | N/A | N/A | N/A | N/A |
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
A 104-week carcinogenicity study with colesevelam hydrochloride was conducted in CD-1 mice, at oral dietary doses up to 3 g/kg/day. This dose was approximately 50 times the maximum recommended human dose of 4.5 g/day, based on body weight, mg/kg. There were no significant drug-induced tumor findings in male or female mice. In a 104-week carcinogenicity study with colesevelam hydrochloride in Harlan Sprague-Dawley rats, a statistically significant increase in the incidence of pancreatic acinar cell adenoma was seen in male rats at doses >1.2 g/kg/day (approximately 20 times the maximum human dose, based on body weight, mg/kg) (trend test only). A statistically significant increase in thyroid C-cell adenoma was seen in female rats at 2.4 g/kg/day (approximately 40 times the maximum human dose, based on body weight, mg/kg).
Mutagenesis
Colesevelam hydrochloride and 4 degradants present in the drug substance have been evaluated for mutagenicity in the Ames test and a mammalian chromosomal aberration test. The 4 degradants and an extract of the parent compound did not exhibit genetic toxicity in an in vitro bacterial mutagenesis assay in S. typhimurium and E. coli (Ames assay) with or without rat liver metabolic activation. An extract of the parent compound was positive in the Chinese Hamster Ovary (CHO) cell chromosomal aberration assay in the presence of metabolic activation and negative in the absence of metabolic activation. The results of the CHO cell chromosomal aberration assay with 2 of the 4 degradants, decylamine HCl and aminohexyltrimethyl ammonium chloride HCl, were equivocal in the absence of metabolic activation and negative in the presence of metabolic activation. The other 2 degradants, didecylamine HCl and 6-decylamino-hexyltrimethyl ammonium chloride HCl, were negative in the presence and absence of metabolic activation.
13.2 Animal Toxicology and/or Pharmacology
Reproductive Toxicology Studies
Reproduction studies have been performed in rats and rabbits at doses up to 3 g/kg/day and 1 g/kg/day, respectively (approximately 50 and 17 times the maximum human dose, based on body weight, mg/kg) and have revealed no evidence of harm to the fetus due to colesevelam hydrochloride.
14. Clinical Studies
14.1 Primary Hyperlipidemia
WELCHOL reduces total cholesterol (TC), LDL-C, apolipoprotein B (Apo B), and non-high-density lipoprotein cholesterol (non-HDL-C) when administered alone or in combination with a statin in patients with primary hyperlipidemia.
Approximately 1600 patients were studied in 9 clinical trials with treatment durations ranging from 4 to 50 weeks. With the exception of one open-label, uncontrolled, long-term extension study, all studies were multicenter, randomized, double-blind, and placebo-controlled. A maximum therapeutic response to WELCHOL was achieved within 2 weeks and was maintained during long-term therapy.
Monotherapy
In a study in patients with LDL-C between 130 mg/dL and 220 mg/dL (mean 158 mg/dL), WELCHOL was given for 24 weeks in divided doses with the morning and evening meals.
As shown in Table 7, the mean LDL-C reductions were 15% and 18% at the 3.8 g and 4.5 g doses. The respective mean TC reductions were 7% and 10%. The mean Apo B reductions were 12% in both treatment groups. WELCHOL at both doses increased HDL-C by 3%. Increases in TG of 9-10% were observed at both WELCHOL doses, but the changes were not statistically different from placebo.
| Grams/Day | N | TC | LDL-C | Apo B | HDL-C* | Non-HDL-C | TG* |
|---|---|---|---|---|---|---|---|
| Placebo | 88 | +1 | 0 | 0 | −1 | +1 | +5 |
| 3.8 g (6 tablets) | 95 | −7† | −15† | −12† | +3† | −10† | +10 |
| 4.5 g (7 tablets) | 94 | −10† | −18† | −12† | +3 | −13† | +9 |
In a study in 98 patients with LDL-C between 145 mg/dL and 250 mg/dL (mean 169 mg/dL), WELCHOL 3.8 g was given for 6 weeks as a single dose with breakfast, as a single dose with dinner, or as divided doses with breakfast and dinner. The mean LDL-C reductions were 18%, 15%, and 18% for the 3 dosing regimens, respectively. The reductions with these 3 regimens were not statistically different from one another.
Combination Therapy
Co-administration of WELCHOL and a statin (atorvastatin, lovastatin, or simvastatin) in 3 clinical studies demonstrated an additive reduction of LDL-C. The mean baseline LDL-C was 184 mg/dL in the atorvastatin study (range 156-236 mg/dL), 171 mg/dL in the lovastatin study (range 115-247 mg/dL), and 188 mg/dL in the simvastatin study (range 148-352 mg/dL). As demonstrated in Table 8, WELCHOL doses of 2.3 g to 3.8 g resulted in an additional 8% to 16% reduction in LDL-C above that seen with the statin alone.
| Dose/Day | N | TC | LDL-C | Apo B | HDL-C* | Non-HDL-C | TG* |
|---|---|---|---|---|---|---|---|
| Atorvastatin Trial (4-week) | |||||||
| Placebo | 19 | +4 | +3 | −3 | +4 | +4 | +10 |
| Atorvastatin 10 mg | 18 | −27† | −38† | −32† | +8 | −35† | −24† |
| WELCHOL 3.8 g/Atorvastatin 10 mg | 18 | −31† | −48† | −38† | +11 | −40† | −1 |
| Atorvastatin 80 mg | 20 | −39† | −53† | −46† | +6 | −50† | −33† |
| Simvastatin Trial (6-week) | |||||||
| Placebo | 33 | −2 | −4 | −4† | −3 | −2 | +6† |
| Simvastatin 10 mg | 35 | −19† | −26† | −20† | +3† | −24† | −17† |
| WELCHOL 3.8 g/Simvastatin 10 mg | 34 | −28† | −42† | −33† | +10† | −37† | −12† |
| Simvastatin 20 mg | 39 | −23† | −34† | −26† | +7† | −30† | −12† |
| WELCHOL 2.3 g/Simvastatin 20 mg | 37 | −29† | −42† | −32† | +4† | −37† | −12† |
| Lovastatin Trial (4-week) | |||||||
| Placebo | 26 | +1 | 0 | 0 | +1 | +1 | +1 |
| Lovastatin 10 mg | 26 | −14† | −22† | −16† | +5 | −19† | 0 |
| WELCHOL 2.3 g/Lovastatin 10 mg Together | 27 | −21† | −34† | −24† | +4 | −27† | −1 |
| WELCHOL 2.3 g/Lovastatin 10 mg Apart | 23 | −21† | −32† | −24† | +2 | −28† | −2 |
In all 3 studies, the LDL-C reduction achieved with the combination of WELCHOL and any given dose of statin therapy was statistically superior to that achieved with WELCHOL or that dose of the statin alone. The LDL-C reduction with atorvastatin 80 mg was not statistically significantly different from the combination of WELCHOL 3.8 g and atorvastatin 10 mg.
Pediatric Therapy
The safety and efficacy of WELCHOL in pediatric patients were evaluated in an 8-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study followed by an open-label phase, in 194 boys and postmenarchal girls 10-17 years of age (mean age 14.1 years) with HeFH, taking a stable dose of an FDA-approved statin (with LDL-C >130 mg/dL) or naïve to lipid-lowering therapy (with LDL-C >160 mg/dL). This study had 3 periods: a single-blind, placebo stabilization period; an 8-week, randomized, double-blind, parallel-group, placebo-controlled treatment period; and an 18-week, open-label treatment period. Forty-seven (24%) patients were taking statins and 147 (76%) patients were statin-naïve at screening. The mean baseline LDL-C at Day 1 was approximately 199 mg/dL.
During the double-blind treatment period, patients were assigned randomly to treatment: WELCHOL 3.8 g/day (n=64), WELCHOL 1.9 g/day (n=65), or placebo (n=65). In total, 186 patients completed the double-blind treatment period. After 8 weeks of treatment, WELCHOL 3.8 g/day significantly decreased plasma levels of LDL-C, non-HDL-C, TC, and Apo B and significantly increased HDL-C. A moderate, non-statistically significant increase in TG was observed versus placebo (Table 9).
| Treatment Difference | TC | LDL-C | Apo B | HDL-C | Non-HDL-C | TG* |
|---|---|---|---|---|---|---|
| (N=128) | (N=128) | (N=124) | (N=128) | (N=128) | (N=128) | |
| Values represent LS mean. Only patients with values at both study baseline and endpoint are included in this table. Study baseline was defined as the last value measured before or on Day 1 prior to the first dose of randomized study medication. | ||||||
| Results were based on the ITT population with LOCF. | ||||||
| WELCHOL 3.8 g vs Placebo | -7† | -13† | -8† | +6† | −11† | +5 |
During the open-label treatment period patients were treated with WELCHOL 3.8 g/day. In total, 173 (89%) patients completed 26 weeks of treatment. Results at Week 26 were consistent with those at Week 8.
14.2 Type 2 Diabetes Mellitus
WELCHOL has been studied as monotherapy and in combination with metformin, pioglitazone, sulfonylureas, and insulin. In these studies, WELCHOL and placebo were administered either as 3 tablets twice daily with lunch and dinner or as 6 tablets with dinner alone.
Monotherapy
The efficacy of WELCHOL 3.8 g/day as anti-diabetes monotherapy was evaluated in a randomized double-blind, placebo-controlled trial involving 357 patients (176 WELCHOL and 181 placebo) with type 2 diabetes mellitus who were treatment-naïve or had not received antihyperglycemic medication within 3 months prior to the start of the study. Statin use at baseline was reported in 13% of the WELCHOL-treated patients and 16% of the placebo-treated patients.
WELCHOL resulted in a statistically significant reduction in HbA1c of 0.27% compared to placebo (Table 10).
The mean baseline LDL-C was 121 mg/dL in the monotherapy trial. WELCHOL treatment resulted in a placebo-corrected 11% reduction in LDL-C. WELCHOL treatment also reduced serum TC, ApoB, and non-HDL-C (Table 11). The mean change in body weight was -0.6 kg for WELCHOL and -0.7 kg for placebo treatment groups.
| WELCHOL 3.8 g/day | Placebo | |
|---|---|---|
| FPG = fasting plasma glucose | ||
| HbA1c (%), Mean | ||
| N | 175 | 169 |
| Baseline | 8.25 | 8.17 |
| Change from baseline* | -0.26 | 0.01 |
| Treatment difference (p-value) | -0.27 (p=0.013) | |
| FPG (mg/dL), Mean | ||
| N | 172 | 166 |
| Baseline | 172 | 168 |
| Change from baseline* | -4.6 | 5.7 |
| Treatment difference (p-value) | -10.3 (p=0.037†) | |
| Dose/Day | N* | TC | LDL-C | Apo B | HDL-C | Non-HDL-C | TG† |
|---|---|---|---|---|---|---|---|
|
|||||||
| WELCHOL 3.8 g | 162 | -3.3‡ | -10.0‡ | -5.6‡ | 1.7 | -4.4‡ | 15.5 |
| Placebo | 160 | 1.8 | 1.2 | 0.9 | -0.1 | 3.0 | 5.8 |
Add-on Combination Therapy
The efficacy of WELCHOL 3.8 g/day in patients with type 2 diabetes mellitus was evaluated in 5 double-blind, placebo-controlled add-on therapy trials involving a total of 1691 patients with baseline HbA1c 7.5-9.5%. Patients were enrolled and maintained on their pre-existing, stable, background anti-diabetic regimen. Statin use at baseline was reported in 41% of the WELCHOL-treated patients and 48% of the placebo-treated patients.
In 3 add-on combination therapy trials (metformin, sulfonylurea and insulin), treatment with WELCHOL resulted in a statistically significant reduction in HbA1c of 0.5% compared to placebo. Similar placebo-corrected reductions in HbA1c occurred in patients who received WELCHOL in combination with metformin, sulfonylurea, or insulin monotherapy or combinations of these therapies with other anti-diabetic agents. In the pioglitazone trial, treatment with WELCHOL resulted in a statistically significant reduction in HbA1c of 0.32% compared to placebo. In the metformin, pioglitazone, and sulfonylurea trials, treatment with WELCHOL also resulted in statistically significant reductions in FPG of at least 14 mg/dL compared to placebo.
WELCHOL had consistent effects on HbA1c across subgroups of age, gender, race, body mass index, and baseline HbA1c. WELCHOL's effects on HbA1c were also similar for the two dosing regimens (3 tablets with lunch and with dinner or 6 tablets with dinner alone).
The mean baseline LDL-C was 104 mg/dL in the metformin study (range 32-214 mg/dL), 107 mg/dL in the pioglitazone study (range 48-263 mg/dL), 106 mg/dL in the sulfonylurea study (range 41-264 mg/dL), 102 mg/dL in the insulin study (range 35-204 mg/dL). In these trials, WELCHOL treatment was associated with a 12% to 16% reduction in LDL-C levels. The percentage decreases in LDL-C were of similar magnitude to those observed in patients with primary hyperlipidemia. WELCHOL treatment was associated with statistically significant increases in TG levels in the studies of patients on insulin, patients on a sulfonylurea, and patients on pioglitazone but not in the study of patients on metformin. The clinical significance of these increases is unknown. WELCHOL is contraindicated in patients with TG levels >500 mg/dL [see Contraindications (4)], and periodic monitoring of lipid parameters including TG is recommended [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)].
Body weight did not significantly increase from baseline with WELCHOL therapy, compared with placebo, in any of the add-on combination diabetes studies.
Add-on Combination Therapy with Metformin
WELCHOL 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 316 patients already receiving treatment with metformin alone (N=159) or metformin in combination with other oral agents (N=157). A total of 60% of these patients were receiving ≥1,500 mg/day of metformin. In combination with metformin, WELCHOL resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 12). WELCHOL also reduced TC, LDL-C, Apo B, and non-HDL-C (Table 13). The mean percent change in serum LDL-C levels with WELCHOL compared to placebo was -16% among statin users and statin non-users; the median percent change in serum TG levels with WELCHOL compared to placebo was -2% among statin users and 10% among statin non-users. The mean change in body weight was -0.5 kg for WELCHOL and -0.3 kg for placebo.
| Total Patient Population | Metformin Alone | Metformin in Combination with Other Oral Anti-diabetic Agents | ||||
|---|---|---|---|---|---|---|
| WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | |
|
||||||
| HbA1c (%), Mean | ||||||
| N | 148 | 152 | 79 | 76 | 69 | 76 |
| Baseline | 8.1 | 8.1 | 8.2 | 8.2 | 8.1 | 8.0 |
| Change from baseline* | -0.4 | 0.2 | -0.4 | 0.0 | -0.4 | 0.3 |
| Treatment difference (p-value) | -0.5 (p<0.001) | -0.5 (p=0.002) | -0.6 (p<0.001) | |||
| FPG (mg/dL), Mean | ||||||
| N | 149 | 152 | 79 | 76 | 70 | 76 |
| Baseline | 178 | 174 | 184 | 180 | 171 | 168 |
| Change from baseline* | -3 | 11 | -7 | 8 | 0 | 13 |
| Treatment difference (p-value) | -14 (p=0.01) | -14 (p=0.07) | -14 (p=0.10) | |||
| Dose/Day | N* | TC | LDL-C | Apo B | HDL-C | Non-HDL-C | TG† |
|---|---|---|---|---|---|---|---|
|
|||||||
| Total Patient Population | |||||||
| WELCHOL 3.8 g | 125 | -4‡ | -12‡ | -4‡ | 1 | -6‡ | 12 |
| Placebo | 126 | 3 | 4 | 4 | 0 | 5 | 7 |
| Metformin Alone | |||||||
| WELCHOL 3.8 g | 66 | -3 | -9 | -2 | 1 | -4 | 15 |
| Placebo | 61 | 2 | 0 | 1 | -2 | 4 | 8 |
| Metformin in Combination with Other Oral Anti-diabetic Agents | |||||||
| WELCHOL 3.8 g | 59 | -6‡ | -15‡ | -6‡ | 1 | -7‡ | 8 |
| Placebo | 65 | 4 | 7 | 7 | 2 | 6 | 5 |
Add-on Combination Therapy with Pioglitazone
WELCHOL 3.8 g/day or placebo was added to background anti-diabetic therapy in a 24-week trial of 562 patients already receiving treatment with pioglitazone alone (N=51) or pioglitazone in combination with other oral agents (N=511). Of these, most were on dual therapy with metformin (N=298) or triple therapy with metformin and a sulfonylurea (N=139). In combination with pioglitazone-based therapy, WELCHOL resulted in statistically significant reductions in HbA1c and FPG compared to placebo (Table 14). WELCHOL also reduced TC, LDL-C, Apo B, and non-HDL-C but increased serum TG (Table 15). The mean change in body weight was 0.8 kg for WELCHOL and 0.4 kg for placebo.
| WELCHOL 3.8 g/day | Placebo | |
|---|---|---|
|
||
| HbA1c (%), Mean | ||
| N | 271 | 276 |
| Baseline | 8.2 | 8.1 |
| Change from baseline* | -0.34 | -0.02 |
| Treatment difference (p-value) | -0.32 (0.0001) | |
| FPG (mg/dL), Mean | ||
| N | 268 | 270 |
| Baseline | 155 | 157 |
| Change from baseline* | -4.8 | +9.9 |
| Treatment difference (p-value) | -14.7 (<0.0001) | |
Add-on Combination Therapy with Sulfonylurea
WELCHOL 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 460 patients already treated with sulfonylurea alone (N=156) or sulfonylurea in combination with other oral agents (N=304). A total of 72% of these patients were receiving at least half-maximal doses of sulfonylurea therapy. In combination with a sulfonylurea, WELCHOL resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 16). WELCHOL also reduced TC, LDL-C, Apo B, and non-HDL-C, but increased serum TG (Table 17). The mean percent change in serum LDL-C levels with WELCHOL compared to placebo was -18% among statin users and -15% among statin non-users; the median percent increase in serum TG with WELCHOL compared to placebo was 29% among statin users and 9% among statin non-users. The mean change in body weight was 0.0 kg for WELCHOL and -0.4 kg for placebo.
| Total Patient Population | Sulfonylurea Alone | Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | ||||
|---|---|---|---|---|---|---|
| WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | |
|
||||||
| HbA1c (%), Mean | ||||||
| n | 218 | 218 | 69 | 80 | 149 | 138 |
| Baseline | 8.2 | 8.3 | 8.2 | 8.4 | 8.2 | 8.3 |
| Change from baseline* | -0.3 | 0.2 | -0.3 | 0.5 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p<0.001) | -0.8 (p<0.001) | -0.4 (p<0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 218 | 217 | 70 | 80 | 148 | 137 |
| Baseline | 177 | 181 | 181 | 186 | 175 | 178 |
| Change from baseline* | -4 | 10 | 3 | 15 | -11 | 4 |
| Treatment difference (p-value) | -14 (p=0.009) | -12 (p=0.18) | -14 (p=0.03) | |||
| Dose/Day | N* | TC | LDL-C | Apo B | HDL-C | Non-HDL-C | TG† |
|---|---|---|---|---|---|---|---|
|
|||||||
| Total Patient Population | |||||||
| WELCHOL 3.8 g | 186 | -5‡ | -16‡ | -6‡ | 1 | -6‡ | 20‡ |
| Placebo | 193 | 0 | 1 | 1 | 0 | 1 | 1 |
| Sulfonylurea Alone | |||||||
| WELCHOL 3.8 g | 57 | -5 | -14‡ | -5 | -1 | -6 | 17 |
| Placebo | 68 | 0 | 1 | 1 | 1 | 0 | -1 |
| Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | |||||||
| WELCHOL 3.8 g | 129 | -5 | -18‡ | -7‡ | 1 | -6 | 21‡ |
| Placebo | 125 | 0 | 0 | 1 | 0 | 1 | 2 |
Add-on Combination Therapy with Insulin
WELCHOL 3.8 g/day or placebo was added to background anti-diabetic therapy in a 16-week trial of 287 patients already treated with insulin alone (N=116) or insulin in combination with oral agents (N=171). At baseline, the median daily insulin dose was 70 units in the WELCHOL group and 65 units in the placebo group. In combination with insulin, WELCHOL resulted in a statistically significant placebo-corrected reduction in HbA1c (Table 18). WELCHOL also reduced LDL-C and Apo B, but increased serum TG (Table 19). The mean percent change in serum LDL-C levels with WELCHOL compared to placebo was -13% among statin users and statin non-users; the median percent increase in serum TG levels with WELCHOL compared to placebo was 24% among statin users and 17% among statin non-users. The mean change in body weight was 0.6 kg for WELCHOL and 0.2 kg for placebo.
| Total Patient Population | Insulin Alone | Insulin in Combination with Oral Anti-diabetic Agents | ||||
|---|---|---|---|---|---|---|
| WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | WELCHOL 3.8 g/day | Placebo | |
|
||||||
| HbA1c (%), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 8.3 | 8.2 | 8.2 | 8.3 | 8.3 | 8.2 |
| Change from baseline* | -0.4 | 0.1 | -0.4 | 0.2 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p<0.001) | -0.6 (p<0.001) | -0.4 (p<0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 165 | 151 | 165 | 163 | 165 | 143 |
| Change from baseline* | 2 | 16 | 8 | 17 | -4 | 14 |
| Treatment difference (p-value) | -15 (p=0.08) | -9 (p=0.51) | -18 (p=0.09) | |||
| Dose/Day | N* | TC | LDL-C | Apo B | HDL-C | Non-HDL-C | TG† |
|---|---|---|---|---|---|---|---|
|
|||||||
| Total Patient Cohort | |||||||
| WELCHOL 3.8 g | 129 | -3 | -12‡ | -4 | -1 | -3 | 23‡ |
| Placebo | 121 | 1 | 1 | 1 | 0 | 1 | 0 |
| Insulin Alone | |||||||
| WELCHOL 3.8 g | 46 | -3 | -12 | -5 | 0 | -3 | 19 |
| Placebo | 48 | 2 | 4 | 2 | 3 | 2 | -2 |
| Insulin in Combination with Oral Anti-diabetic Agents | |||||||
| WELCHOL 3.8 g | 83 | -4 | -13 | -4 | -1 | -3 | 25‡ |
| Placebo | 73 | -1 | -3 | 0 | -1 | -1 | 2 |
16. How is Welchol supplied
WELCHOL 625 mg tablets are supplied as off-white, solid tablets imprinted with the word "Sankyo" and "C01" on one side and are available as follows:
- Bottles of 180 – NDC 65597-701-18
WELCHOL 3.75 gram packets for oral suspension contain a white to pale yellow powder containing yellow granules and are available as follows:
- Cartons of 30 packets – NDC 65597-902-30
17. Patient Counseling Information
Hypertriglyceridemia and Pancreatitis
Inform patients that WELCHOL may increase their serum triglycerides which can lead to hypertriglyceridemia and pancreatitis. Instruct patients to discontinue WELCHOL and seek prompt medical attention if the symptoms of acute pancreatitis occur (e.g., severe abdominal pain with or without nausea and vomiting) [see Warnings and Precautions (5.1)].
Gastrointestinal
Inform patients that WELCHOL may cause bowel obstruction. Instruct patients to promptly discontinue WELCHOL and seek medical attention if severe abdominal pain or severe constipation occurs [see Warnings and Precautions (5.2)].
Drug and Vitamin Interactions
Advise patients that WELCHOL has drug interactions, and WELCHOL may decrease the absorption of fat-soluble vitamins A, D, E, and K. Instruct patients to take oral vitamins at least 4 hours prior to WELCHOL. Instruct patients to inform their physician about all the drugs and vitamins that they are prescribed or take over the counter [see Warnings and Precautions (5.3) and Drug Interactions (7)].
Hypertriglyceridemia and Cardiovascular Disease
Inform patients that WELCHOL may increase serum triglycerides and that the long-term effect of hypertriglyceridemia on the risk of coronary artery disease is uncertain [see Warnings and Precautions (5.1)].
Administration [see Dosage and Administration (2.2, 2.4)]:
Tablets
Advise patients to take WELCHOL tablets with a meal and liquid. Inform patients that WELCHOL tablets can be taken as 6 tablets once daily or 3 tablets twice daily.
For Oral Suspension
Instruct patients to empty the entire contents of one packet into a glass or cup and add 1 cup (8 ounces) of water, fruit juice, or diet soft drinks. Stir well and drink. Advise patients to take WELCHOL oral suspension with meals. Advise patient to not take WELCHOL oral suspension in its dry form.
Females of Reproductive Potential
Advise females of reproductive potential that WELCHOL may reduce the effectiveness of oral contraceptives, and to take oral contraceptives at least 4 hours before taking WELCHOL [see Drug Interactions (7.1) and Use in Specific Populations (8.3)].
PRINCIPAL DISPLAY PANEL - 3.75 g Packet Label
TEAR HERE
No cutting required
Sugar-Free
This packet is contained within the
CARTON NDC 65597-902-30
Welchol™
(colesevelam HCl)
For Oral Suspension
3.75 g
Citrus Flavor
Single-Dose Packet
Each packet contains 3.75 grams of
colesevelam hydrochloride.
Rx only
Keep Out of Reach of Children.
Package Not Child Resistant.

PRINCIPAL DISPLAY PANEL - 3.75 g Packet Carton
Keep Out of Reach of Children.
Package Not Child Resistant.
NDC 65597-902-30
30 packets
Welchol™
(colesevelam HCl)
For Oral Suspension
3.75 g
Each packet contains 3.75 grams of colesevelam hydrochloride.
Rx only
Dosing and use: see package insert.

PRINCIPAL DISPLAY PANEL - 3.75 g Bar Package Carton - 209-30
NDC 65587-209-30
Welchol®
(colesevelam HCl)
Chewable Bar
3.75 g
30 CHEWABLE BARS
RX ONLY
EACH BAR CONTAINS 3.75 g
OF COLESEVELAM HYDROCHLORIDE
CHILD-RESISTANT WRAPPERS
CONTAINS MEDICATION
KEEP OUT OF REACH OF CHILDREN
DOSING AND USE:
SEE PACKAGE INSERT
NOTE TO PHARMACISTS:
DO NOT BREAK BOX. DISPENSE AS 1 BOX.
CHOCOLATE FLAVOR

PRINCIPAL DISPLAY PANEL - 3.75 g Bar Package Carton - 210-30
NDC 65587-210-30
Welchol®
(colesevelam HCl)
Chewable Bar
3.75 g
30 CHEWABLE BARS
RX ONLY
EACH BAR CONTAINS 3.75 g
OF COLESEVELAM HYDROCHLORIDE
CHILD-RESISTANT WRAPPERS
CONTAINS MEDICATION
KEEP OUT OF REACH OF CHILDREN
DOSING AND USE:
SEE PACKAGE INSERT
NOTE TO PHARMACISTS:
DO NOT BREAK BOX. DISPENSE AS 1 BOX.
STRAWBERRY FLAVOR

PRINCIPAL DISPLAY PANEL - 3.75 g Bar Package Carton - 208-30
NDC 65587-208-30
Welchol®
(colesevelam HCl)
Chewable Bar
3.75 g
30 CHEWABLE BARS
RX ONLY
EACH BAR CONTAINS 3.75 g
OF COLESEVELAM HYDROCHLORIDE
CHILD-RESISTANT WRAPPERS
CONTAINS MEDICATION
KEEP OUT OF REACH OF CHILDREN
DOSING AND USE:
SEE PACKAGE INSERT
NOTE TO PHARMACISTS:
DO NOT BREAK BOX. DISPENSE AS 1 BOX.
CARAMEL FLAVOR

| WELCHOL
colesevelam hydrochloride tablet, film coated |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| WELCHOL
colesevelam hydrochloride for suspension |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WELCHOL
colesevelam hydrochloride for suspension |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| WELCHOL
colesevelam hydrochloride bar, chewable |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| WELCHOL
colesevelam hydrochloride bar, chewable |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| WELCHOL
colesevelam hydrochloride bar, chewable |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| Labeler - Daiichi Sankyo Inc. (068605067) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Powdersize, LLC | 080120056 | PARTICLE SIZE REDUCTION(65597-701, 65597-902) | |
More about Welchol (colesevelam)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (43)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- Drug class: bile acid sequestrants
- Breastfeeding
- En español


