Sodium Sulfacetamide and Sulfur Cream Prescribing Information
Package insert / product label
Dosage form: cream
Drug class: Topical antibiotics
Medically reviewed by Drugs.com. Last updated on Dec 18, 2023.
On This Page
Sodium Sulfacetamide and Sulfur Cream Description
Each gram contains 100 mg of sodium sulfacetamide and 50 mg of colloidal sulfur in a vehicle consisting of: butylated hydroxytoluene, cetyl alcohol, citric acid, disodium EDTA, glyceryl stearate SE, mineral oil, PEG-100 stearate, phenoxyethanol, propylene glycol, purified water, sodium thiosulfate, stearyl alcohol, triacetin, xanthan gum.
Sodium sulfacetamide is a sulfonamide with antibacterial activity while sulfur acts as a keratolytic agent. Sodium sulfacetamide is C 8H 9N 2NaO 3S·H 2O with molecular weight of 254.24. Chemically, sodium sulfacetamid is N-[(4-aminophenyl) sulfonyl]-acetamide, monosodium salt, monohydrate. The structural formula is:
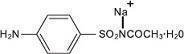
Sodium sulfacetamide is an odorless, white, crystalline powder with a bitter taste. It is freely soluble in water, sparingly soluble in alcohol, while practically insoluble in benzene, in chloroform and in ether.
Sodium Sulfacetamide and Sulfur Cream - Clinical Pharmacology
Sodium sulfacetamide exerts a bacteriostatic effect against sulfonamide sensitive Gram-positive and Gram-negative microorganisms commonly isolated from secondary cutaneous pyogenic infections. It acts by restricting the synthesis of folic acid required by bacteria for growth, by its competition with para-aminobenzoic acid. There is no clinical data available on the degree and rate of systemic absorption of this product when applied to the skin or scalp. However, significant absorption of sodium sulfacetamide through the skin has been reported.
The following in vitro data is available, but the clinical significance is unknown. Organisms that show susceptibility to sodium sulfacetamide are: Streptococci, Staphylococci, E. coli, Klebsiella pneumoniae, Pseudomonas pyocyanea, Salmonella species, Proteus vulgaris, Nocardia and Actinomyces.
The exact mode of action of sulfur in the treatment of acne is unknown, but it has been reported that it inhibits the growth of Propionibacterium acnes and the formation of free fatty acids.
Related/similar drugs
prednisone, doxycycline, metronidazole topical, clindamycin topical, dexamethasone, hydrocortisone topical, erythromycin topical
Indications and Usage for Sodium Sulfacetamide and Sulfur Cream
This product is indicated for use in the topical control of acne vulgaris, acne rosacea and seborrheic dermatitis.
Contraindications
This product is contraindicated in persons with known or suspected hypersensitivity to any of the ingredients of the product. This product is not to be used by patients with kidney disease.
Warnings
Sulfonamides are known to cause Stevens-Johnson syndrome in hypersensitive individuals. Stevens-Johnson syndrome also has been reported following the use of sodium sulfacetamide topically. Cases of drug-induced systemic lupus erythematosus from topical sulfacetamide also have been reported. In one of these cases, there was a fatal outcome.
KEEP OUT OF THE REACH OF CHILDREN.
FOR EXTERNAL USE ONLY. NOT FOR OPHTHALMIC USE.
General:
Nonsusceptible organisms, including fungi, may proliferate with the use of this preparation.
Although rare, sensitivity to sodium sulfacetamide may occur. Therefore, caution and careful supervision should be observed when prescribing this drug for patients who may be prone to hypersensitivity to topical sulfonamides. If the use of this product produces signs of hypersensitivity or other untoward reactions, discontinue use of the preparation. Patients should be carefully observed for possible local irritation or sensitization during long-term therapy. Systemic toxic reactions such as agranulocytosis, acute hemolytic anemia, purpura hemorrhagica, drug fever, jaundice and contact dermatitis indicate hypersensitivity to sulfonamides. Particular caution should be employed if areas of denuded or abraded skin are involved. Systemic absorption of topical sulfonamides is greater following application to large, infected, abraded, denuded, or severely burned areas. Under these circumstances, any of the adverse effects produced by the systemic administration of these agents could potentially occur, and appropriate observations and laboratory determinations should be performed.
The object of this therapy is to achieve desquamation without irritation, but sodium sulfacetamide and sulfur can cause reddening and scaling of the epidermis. These side effects are not unusual in the treatment of acne vulgaris, but patients should be cautioned about the possibility.
Information for Patients:
Patients should discontinue the use of this product if the condition becomes worse or if a rash develops in the area being treated or elsewhere. The use of this product also should be discontinued promptly, and the physician notified if any arthritis, fever, or sores in the mouth develop. Avoid contact with eyes, lips and mucous membranes.
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Long-term animal studies for carcinogenic potential have not been performed on this product to date. Studies on reproduction and fertility also have not been performed. Chromosomal nondisjunction has been reported in the yeast, Saccharomyces cerevisiae, following application of sodium sulfacetamide. The significance of this finding to the topical use of sodium sulfacetamide in the human is unknown.
Pregnancy:
Category C. Animal reproduction studies have not been conducted with this product. It is also not known whether this product can affect reproduction capacity or cause fetal harm when administered to a pregnant woman. This product should be used by a pregnant woman only if clearly needed or when potential benefits outweigh potential hazards to the fetus.
Adverse Reactions/Side Effects
Reports of irritation and hypersensitivity to sodium sulfacetamide are uncommon. The following adverse reactions, reported after administration of sterile ophthalmic sodium sulfacetamide, are noteworthy: instances of Stevens-Johnson syndrome and instances of local hypersensitivity which progressed to a syndrome resembling systemic lupus erythematosus; in one case a fatal outcome was reported (see WARNINGS).
Overdosage
The oral LD 50 of sulfacetamide in mice is 16.5 g/kg. In the event of overdosage, emergency treatment should be started immediately.
Manifestations: Overdosage may cause nausea and vomiting. Large oral overdosage may cause hematuria, crystalluria and renal shutdown due to the precipitation of sulfa crystals in the renal tubules and the urinary tract. For treatment, contact your local Poison Control Center or your doctor.
Sodium Sulfacetamide and Sulfur Cream Dosage and Administration
Cleanse affected areas. Apply a thin layer to the affected areas with light massaging, 1 to 3 times daily or as directed by a physician.
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). Brief exposure to temperatures up to 40°C (104°F) may be tolerated provided the mean kinetic temperature does not exceed 25°C (77°F); however, such exposure should be minimized.
NOTICE:
Protect from freezing and excessive heat. The product may tend to darken slightly on storage. Slight discoloration does not impair the efficacy or safety of the product. Keep bottle tightly closed.
Occasionally, a slight discoloration of fabric may occur when an excessive amount of the product is used and comes in contact with white fabrics. This discoloration, however, presents no problem, as it is readily removed by ordinary laundering without bleaches.
How is Sodium Sulfacetamide and Sulfur Cream supplied
This product is supplied in the following sizes:
1 oz. (28 g) bottles, NDC 58657-468-01
2 oz. (57 g) bottles, NDC 58657-468-02
To report a serious adverse event or obtain product information, call (877) 250-3427.
Manufactured in the U.S.A. for
Method Pharmaceuticals, LLC
Southlake, Texas 76092
| SODIUM SULFACETAMIDE 10% AND SULFUR 5% EMOLLIENT CREAM
sodium sulfacetamide 10% and sulfur 5% emollient cream cream |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Method Pharmaceuticals, LLC (060216698) |
More about sulfacetamide sodium/sulfur topical
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (64)
- Side effects
- Dosage information
- Drug class: topical antibiotics
Patient resources
Professional resources
- Sodium Sulfacetamide Sulfur Cleansing Cloths prescribing information
- Sodium Sulfacetamide and Sulfur Cleanser (FDA)
- Sodium Sulfacetamide and Sulfur Foam (FDA)
- Sodium Sulfacetamide and Sulfur Lotion (FDA)
- Sodium Sulfacetamide and Sulfur Suspension (FDA)
Other brands
Plexion, Sulfacleanse 8/4, Plexion Cleansing Cloths, Sumadan, ... +10 more


