Sodium Fluoride F 18 Injection: Package Insert / Prescribing Info
Package insert / product label
Dosage form: injection
Drug class: Diagnostic radiopharmaceuticals
Medically reviewed by Drugs.com. Last updated on Apr 8, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- References
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
SODIUM FLUORIDE F 18 INJECTION, USP
For Intravenous Use
Initial U.S. Approval: XXXX
Indications and Usage for Sodium Fluoride F 18 Injection
Sodium Fluoride F 18 Injection, USP is a radioactive diagnostic agent for positron emission tomography (PET) indicated for imaging of bone to define areas of altered osteogenic activity (1).
Sodium Fluoride F 18 Injection Dosage and Administration
- Sodium Fluoride F18 Injection emits radiation and must be handled with appropriate safety measures (2.1).
- Administer 300-450 MBq (8 to 12 mCi) as an intravenous injection in adults (2.4).
- Administer approximately 2.1 MBq/kg in children with a minimum of 19 MBq (0.5 mCi) and a maximum of 148 MBq (4 mCi) as an intravenous injection (2.5).
- Imaging can begin 1–2 hours after administration; optimally at one hour post administration (2.7).
- Encourage patients to void immediately prior to imaging the lumbar spine and bony pelvis (2.7).
Dosage Forms and Strengths
Multiple-dose vial containing 370 - 7,400 MBq/mL (10 - 200 mCi/mL) of no-carrier-added sodium fluoride F18 at the end of synthesis (EOS) reference time in aqueous 0.9% sodium chloride solution (3). Sodium Fluoride F 18 Injection is a clear, colorless, sterile, pyrogen-free and preservative-free solution for intravenous administration.
Contraindications
None (4).
Warnings and Precautions
- Allergic Reactions: As with any injectable drug product, allergic reactions and anaphylaxis may occur. Emergency resuscitation equipment and personnel should be immediately available. (5.1).
- Cancer Risk: Sodium Fluoride F 18 Injection may increase the risk of cancer. Use the smallest dose necessary for imaging and ensure safe handling to protect the patient and health care worker (5.2).
(5).
Adverse Reactions/Side Effects
No adverse reactions have been reported for Sodium Fluoride F 18 Injection based on a review of the published literature, publicly available reference sources, and adverse drug reaction reporting systems (6).
To report SUSPECTED ADVERSE REACTIONS, contact SOFIE Co. at 1-800-207-1865 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Use In Specific Populations
- Pregnancy: No human or animal data. Any radiopharmaceutical, including Sodium Fluoride F18 injection, may cause fetal harm. Use only if clearly needed (8.1)
- Nursing: A decision should be made whether to interrupt nursing after Sodium Fluoride F 18 Injection administration or not to administer Sodium Fluoride F 18 Injection taking into consideration the importance of the drug to the mother. (8.3).
- Pediatrics: Children are more sensitive to radiation and may be at higher risk of cancer from Sodium Fluoride F18 injection (8.4).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 5/2018
Full Prescribing Information
1. Indications and Usage for Sodium Fluoride F 18 Injection
Sodium Fluoride F 18 Injection, USP is indicated for diagnostic positron emission tomography (PET) imaging of bone to define areas of altered osteogenic activity.
2. Sodium Fluoride F 18 Injection Dosage and Administration
2.1 Radiation Safety - Drug Handling
- Wear waterproof gloves and effective shielding when handling Sodium Fluoride F 18 Injection. Use appropriate safety measures, including shielding, consistent with proper patient management to avoid unnecessary radiation exposure to the patient, occupational workers, clinical personnel, and other persons.
- Radiopharmaceuticals should be used by or under the control of physicians who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
- Use aseptic technique to maintain sterility during all operations involved in the manipulation and administration of Sodium Fluoride F 18 Injection.
- The dose of Sodium Fluoride F 18 Injection should be minimized consistent with the objectives of the procedure, and the nature of the radiation detection devices employed.
- The final dose for the patient should be calculated using proper decay factors from the time of End of Synthesis (EOS), and measured by a suitable radioactivity calibration system before administration [see Description (11.2)].
2.2 Radiation Safety - Patient Preparation
- To minimize the radiation-absorbed dose to the bladder, encourage adequate hydration. Encourage the patient to ingest at least 500 mL of fluid immediately prior and subsequent to the administration of Sodium Fluoride F 18 Injection.
- Encourage the patient to void one-half hour after administration of Sodium Fluoride F 18 Injection and as frequently thereafter as possible for the next 12 hours.
2.3 Drug Preparation and Administration
- Calculate the necessary volume to administer based on calibration time and dose.
- Inspect Sodium Fluoride F 18 Injection visually for particulate matter and discoloration before administration, whenever solution and container permit.
- Do not administer Sodium Fluoride F 18 Injection containing particulate matter or discoloration; dispose of these unacceptable or unused preparations in a safe manner, in compliance with applicable regulations.
- Aseptically withdraw Sodium Fluoride F 18 Injection from its container.
2.5 Recommended Dose for Pediatric Patients
In reported clinical experience in approximately 100 children, weight based doses (2.1 MBq/kg) ranging from 19 MBq– 148 MBq (0.5 mCi- 4 mCi) were used.
2.6 Radiation Dosimetry
The age/weight- based estimated absorbed radiation doses (mGy/MBq) from intravenous injection of Sodium Fluoride F 18 Injection are shown in Table 1. These estimates were calculated based on human data and using the data published by the Nuclear Regulatory Commission [1] and the International Commission on Radiological Protection for Sodium Fluoride Injection [2]. The bone, bone marrow and urinary bladder are considered target and critical organs.
| Estimated Radiation Dose mGy/MBq | ||||||
| Organ | Adult
70 kg * | 15 year
56.8 kg † | 10 year
33.2 kg † | 5 year
19.8 kg † | 1 year
9.7 kg † |
|
|---|---|---|---|---|---|---|
| Adrenals | 0.0062 | 0.012 | 0.018 | 0.028 | 0.052 | |
| Brain | 0.0056 | N/A | N/A | N/A | N/A | |
| Bone surfaces | 0.060 | 0.050 | 0.079 | 0.13 | 0.30 | |
| Breasts | 0.0028 | 0.0061 | 0.0097 | 0.015 | 0.030 | |
| GI | Gallbladder wall | 0.0044 | N/A | N/A | N/A | N/A |
| Stomach wall | 0.0038 | 0.008 | 0.013 | 0.019 | 0.036 | |
| Small intestine | 0.0066 | 0.012 | 0.018 | 0.028 | 0.052 | |
| Upper large intestine wall | 0.0058 | 0.010 | 0.016 | 0.026 | 0.046 | |
| Lower large intestine wall | 0.012 | 0.016 | 0.025 | 0.037 | 0.063 | |
| Heart wall | 0.0039 | N/A | N/A | N/A | N/A | |
| Kidneys | 0.019 | 0.025 | 0.036 | 0.053 | 0.097 | |
| Liver | 0.0040 | 0.0084 | 0.013 | 0.021 | 0.039 | |
| Lungs | 0.0041 | 0.0084 | 0.013 | 0.020 | 0.039 | |
| Muscle | 0.0060 | N/A | N/A | N/A | N/A | |
| Ovaries | 0.011 | 0.016 | 0.023 | 0.036 | 0.063 | |
| Pancreas | 0.0048 | 0.0096 | 0.015 | 0.023 | 0.044 | |
| Red marrow | 0.028 | 0.053 | 0.088 | 0.18 | 0.38 | |
| Skin | 0.0040 | N/A | N/A | N/A | N/A | |
| Spleen | 0.0042 | 0.0088 | 0.014 | 0.021 | 0.041 | |
| Testes | 0.0078 | 0.013 | 0.021 | 0.033 | 0.062 | |
| Thymus | 0.0035 | N/A | N/A | N/A | N/A | |
| Thyroid | 0.0044 | 0.0084 | 0.013 | 0.020 | 0.036 | |
| Urinary bladder wall | 0.25 | 0.27 | 0.4 | 0.61 | 1.1 | |
| Uterus | 0.019 | 0.023 | 0.037 | 0.057 | 0.099 | |
| Other tissue | N/A | 0.010 | 0.015 | 0.024 | 0.044 | |
| Effective Dose
Equivalent mSv/MBq | 0.027 | 0.034 | 0.052 | 0.086 | 0.17 | |
3. Dosage Forms and Strengths
Multiple-dose vial containing 370 - 7,400 MBq/mL (10 - 200 mCi/mL) at EOS reference time of no-carrier-added sodium fluoride F18 in aqueous 0.9% sodium chloride solution. Sodium Fluoride F 18 Injection is a clear, colorless, sterile, pyrogen-free and preservative-free solution for intravenous administration.
5. Warnings and Precautions
5.1 Allergic Reactions
As with any injectable drug product, allergic reactions and anaphylaxis may occur. Emergency resuscitation equipment and personnel should be immediately available.
5.2 Radiation Risks
Sodium Fluoride F 18 Injection may increase the risk of cancer. Carcinogenic and mutagenic studies with Sodium Fluoride F18 injection have not been performed. Use the smallest dose necessary for imaging and ensure safe handling to protect the patient and health care worker [ see Dosage and Administration (2.1)].
6. Adverse Reactions/Side Effects
No adverse reactions have been reported for Sodium Fluoride F 18 Injection based on a review of the published literature, publicly available reference sources, and adverse drug reaction reporting systems. However, the completeness of these sources is not known.
Related/similar drugs
7. Drug Interactions
The possibility of interactions of Sodium Fluoride F 18 Injection with other drugs taken by patients undergoing PET imaging has not been studied.
8. Use In Specific Populations
8.1 Pregnancy
Pregnancy Category C
Any radiopharmaceutical including Sodium Fluoride F18 Injection has a potential to cause fetal harm. The likelihood of fetal harm depends on the stage of fetal development, and the radionuclide dose. Animal reproduction and developmental toxicity studies have not been conducted with Sodium Fluoride F 18 Injection. Prior to the administration of Sodium Fluoride F 18 Injection to women of childbearing potential, assess for presence of pregnancy. Sodium Fluoride F 18 Injection should be given to a pregnant woman only if clearly needed.
8.3 Nursing Mothers
It is not known whether Sodium Fluoride F 18 Injection is excreted into human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to interrupt nursing after administration of Sodium Fluoride F 18 Injection or not to administer Sodium Fluoride F 18 Injection, taking into account the importance of the drug to the mother. The body of scientific information related to radioactivity decay, drug tissue distribution and drug elimination shows that less than 0.01% of the radioactivity administered remains in the body after 24 hours (10 half-lives). To minimize the risks to a nursing infant, interrupt nursing for at least 24 hours.
8.4 Pediatric Use
In reported clinical experience in approximately 100 children, weight based doses (2.1 MBq/kg) ranging from 19 MBq – 148 MBq (0.5 mCi - 4 mCi) were used. Sodium Fluoride F18 was shown to localize to areas of bone turnover including rapidly growing epiphyses in developing long bones. Children are more sensitive to radiation and may be at higher risk of cancer from Sodium Fluoride F18 injection.
11. Sodium Fluoride F 18 Injection Description
11.1 Chemical Characteristics
Sodium Fluoride F 18 Injection is a positron emitting radiopharmaceutical, containing no-carrier-added, radioactive fluoride F18 that is used for diagnostic purposes in conjunction with PET imaging. It is administered by intravenous injection. The active ingredient, sodium fluoride F18, has the molecular formula Na[ 18F] with a molecular weight of 40.99, and has the following chemical structure:
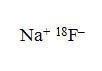
Sodium Fluoride F 18 Injection is provided as a ready-to-use, isotonic, sterile, pyrogen-free, preservative-free, clear and colorless solution. Each mL of the solution contains between 370 MBq and 7,400 MBq (10 mCi to 200 mCi) sodium fluoride F18, at the EOS reference time, in 0.9% aqueous sodium chloride. The pH of the solution is between 4.5 and 8. The solution is presented in XX mL multiple- dose glass vials with variable total volume and total radioactivity in each vial.
11.2 Physical Characteristics
Fluoride F18 decays by positron (β+) emission and has a half-life of 109.7 minutes. Ninety-seven percent of the decay results in emission of a positron with a maximum energy of 633 keV and 3% of the decay results in electron capture with subsequent emission of characteristic X-rays of oxygen. The principal photons useful for diagnostic imaging are the 511 keV gamma photons, resulting from the interaction of the emitted positron with an electron (Table 2). Fluorine F18 atom decays to stable 18O-oxygen.
| Radiation/Emission | % per Disintegration | Mean Energy |
|---|---|---|
|
||
| Positron (β+) | 96.73 | 249.8 keV |
| Gamma (±) * | 193.46 | 511.0 keV |
The specific gamma ray constant for fluoride F18 is 5.7 R/hr/mCi (1.35 x 10 -6Gy/hr/kBq) at 1 cm. The half-value layer (HVL) for the 511 keV photons is 4.1 mm lead (Pb). A range of values for the attenuation of radiation results from the interposition of various thickness of Pb. The range of attenuation coefficients for this radionuclide is shown in Table 3. For example, the interposition of an 8.3 mm thickness of Pb with a coefficient of attenuation of 0.25 will decrease the external radiation by 75%.
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 0 | 0.00 |
| 4 | 0.50 |
| 8 | 0.25 |
| 13 | 0.10 |
| 26 | 0.01 |
| 39 | 0.001 |
| 52 | 0.0001 |
Table 4 lists the fraction of radioactivity remaining at selected time intervals from the calibration time. This information may be used to correct for physical decay of the radionuclide.
12. Sodium Fluoride F 18 Injection - Clinical Pharmacology
12.1 Mechanism of Action
Fluoride F18 ion normally accumulates in the skeleton in an even fashion, with greater deposition in the axial skeleton (e.g. vertebrae and pelvis) than in the appendicular skeleton and greater deposition in the bones around joints than in the shafts of long bones.
12.2 Pharmacodynamics
Increased fluoride F18 ion deposition in bone can occur in areas of increased osteogenic activity during growth, infection, malignancy (primary or metastatic) following trauma, or inflammation of bone.
12.3 Pharmacokinetics
After intravenous administration, fluoride F18 ion is rapidly cleared from the plasma in a biexponential manner. The first phase has a half-life of 0.4 h, and the second phase has a half-life of 2.6 h. Essentially all the fluoride F18 that is delivered to bone by the blood is retained in the bone. One hour after administration of fluoride F18 only about 10% of the injected dose remains in the blood. Fluoride F18 diffuses through capillaries into bone extracellular fluid space, where it becomes bound by chemisorption at the surface of bone crystals, preferentially at sites of newly mineralizing bone.
Deposition of fluoride F18 in bone appears to be primarily a function of blood flow to the bone and the efficiency of the bone in extracting the fluoride F18. Fluoride F18 does not appear to be bound to serum proteins.
In patients with normal renal function, 20% or more of the fluorine ion is cleared from the body in the urine within the first 2 hours after intravenous administration.
14. Clinical Studies
14.1 Metastatic Bone Disease
The doses used in reported studies ranged from 2.7 mCi to 20 mCi (100 MBq to 740 MBq), with an average median dose of 10 mCi (370 MBq) and an average mean dose of 9.2 mCi (340 MBq). In PET imaging of bone metastases with Sodium Fluoride F 18 Injection, focally increased tracer uptake is seen in both osteolytic and osteoblastic bone lesions. Negative PET imaging results with Sodium Fluoride F 18 Injection do not preclude the diagnosis of bone metastases. Also, as benign bone lesions are also detected by Sodium Fluoride F 18 Injection, positive PET imaging results cannot replace biopsy to confirm a diagnosis of cancer.
15. References
- Stabin, M.G., Stubbs, J.B. and Toohey R.E., Radiation Dose Estimates for Radiopharmaceuticals,U.S. Nuclear Regulatory Commission report NUREG/CR-6345, page 10, 1996.
- Radiation Dose to Patients from Radiopharmaceuticals,ICRP publication 53, Ann ICRP, 18 pages 15 and 74, 1987.
- Kocher, D.C., "Radioactive Decay Data Tables: A Handbook of decay data for application to radiation dosimetry and radiological assessments" DOE/TIC-11026, page 69, 1981.
16. How is Sodium Fluoride F 18 Injection supplied
Sodium Fluoride F 18 Injection is supplied in a multiple-dose Type I glass vial with elastomeric stopper and aluminum crimp seal containing between 370 and 7,400 MBq/mL (10 - 200 mCi/mL) of no-carrier-added sodium fluoride F18, at the EOS reference time, in aqueous 0.9% sodium chloride solution. The total volume and total radioactivity per vial are variable. Each vial is enclosed in a shielding container of appropriate thickness.
The product is available in a 30 mL vial configuration with a variable fill volume. The NDC number is:
49609-102-01 (30 mL)
17. Patient Counseling Information
17.1 Pre-study Hydration
Encourage patients to drink at least 500 mL of water prior to drug administration.
17.2 Post-study Voiding
To help protect themselves and others in their environment, patients should take the following precautions for 12 hours after injection: whenever possible, use a toilet and flush several times after each use; wash hands thoroughly after each voiding or fecal elimination. If blood, urine or feces soil clothing, wash the clothing separately.
| Manufactured by:
| SOFIE Co.
21000 Atlantic Boulevard, Suite 730 Dulles, Virginia, 20166, USA |
| Distributed by: | SOFIE Co.
21000 Atlantic Boulevard, Suite 730 Dulles, Virginia, 20166, USA |
PRINCIPAL DISPLAY PANEL - 30 mL Vial Label
Sodium Fluoride F 18 Injection, USP
10-200mCi/mL at End of Synthesis (EOS)
Diagnostic – For Intravenous Use Only
Sterile, Non-Pyrogenic
Batch #: __________________________
EOS date: ________ EOS time: _______
Activity @ EOS: _______________ mCi
Concentration: ____________ mCi/mL
Volume: _________________ mL
Exp. date: _______ Exp time: ________
NDC# 49609-102-01
Each mL contains 0.37 to 7.4 GBq (10
to 200 mCi) of no carrier added
Sodium [
18F] Fluoride in aqueous
0.9% sodium chloride solution at
EOS.
Use within 12 hours of EOS.
30 mL Multiple-Dose Vial
Store at 25°C (77°F); excursions
permitted to 15 – 30°C (59-86°F)
[see USP controlled room
temperature]. Store upright in
shielded container. Aseptically
withdraw and handle doses.
Do not use if cloudy or contains particulate matter.
Calculate correct dosage from date and time of calibration
CAUTION: RADIOACTIVE MATERIAL
RX ONLY
Manufactured By: SOFIE Co. dba SOFIE, 21000 Atlantic Blvd.,
Suite 730, Dulles, VA 20166
18F half life = 109.7 min

Sodium Fluoride F 18 Injection, USP
10-200mCi/mL at End of Synthesis (EOS)
Diagnostic – For Intravenous Use Only
Sterile, Non-Pyrogenic
Batch #: __________________________
EOS date: ________ EOS time: _______
Activity @ EOS: _______________ mCi
Concentration: ____________ mCi/mL
Volume: _________________ mL
Exp. date: _______ Exp time: ________
NDC# 49609-102-01
Each mL contains 0.37 to 7.4 GBq (10
to 200 mCi) of no carrier added
Sodium [ 18F] Fluoride in aqueous
0.9% sodium chloride solution at
EOS.
Use within 12 hours of EOS.
30 mL Multiple-Dose Vial
Store at 25°C (77°F); excursions
permitted to 15 – 30°C (59-86°F)
[see USP controlled room
temperature]. Store upright in
shielded container. Aseptically
withdraw and handle doses.
Do not use if cloudy or contains particulate matter.
Calculate correct dosage from date and time of calibration
CAUTION: RADIOACTIVE MATERIAL
RX ONLY
Manufactured By: N-Molecular, Inc. dba SOFIE, 21000
Atlantic Blvd., Suite 730, Dulles, VA, 20166 for SOFIE Co.
18F half life = 109.7 min

| SODIUM FLUORIDE F 18
sodium fluoride f-18 injection |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - SOFIE Co. (957704737) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 006320902 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 008017970 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 025600556 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 032324142 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 078623800 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 079854636 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| N-Molecular, Inc. dba SOFIE | 079932600 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| N-Molecular, Inc. dba SOFIE | 079932640 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 829109441 | positron emission tomography drug production(49609-102) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| SOFIE Co. dba SOFIE | 832599976 | positron emission tomography drug production(49609-102) | |
