Lumizyme: Package Insert / Prescribing Info
Package insert / product label
Generic name: alglucosidase alfa
Dosage form: injection, powder, for solution
Drug class: Lysosomal enzymes
J Code (medical billing code): J0220 (10 mg, injection)
Medically reviewed by Drugs.com. Last updated on Jan 23, 2025.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Use In Specific Populations
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
LUMIZYME® (alglucosidase alfa), for injection, for intravenous use
Initial U.S. Approval: 2010
WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS, IMMUNE-MEDIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE
See full prescribing information for complete boxed warning.
Hypersensitivity Reactions Including Anaphylaxis
Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate LUMIZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LUMIZYME and immediately initiate appropriate medical treatment, including use of epinephrine. (5.1)
Immune-Mediated Reactions
Immune-mediated reactions presenting as proteinuria, nephrotic syndrome, and necrotizing skin lesions have occurred in some patients following LUMIZYME treatment. Monitor patients for the development of systemic immune-mediated reactions involving skin and other organs while receiving LUMIZYME. (5.3)
Risk of Acute Cardiorespiratory Failure
Infantile-onset Pompe disease (IOPD) patients with compromised cardiac or respiratory function may be at risk of serious acute exacerbation of their cardiac or respiratory compromise due to fluid overload and require additional monitoring. (5.4)
Recent Major Changes
Indications and Usage for Lumizyme
LUMIZYME® is a hydrolytic lysosomal glycogen-specific enzyme indicated for patients with Pompe disease (GAA deficiency). (1)
Lumizyme Dosage and Administration
- Administration of LUMIZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. (2.1)
- Recommended dosage is 20 mg/per kg body weight administered every 2 weeks as an intravenous infusion. The initial infusion rate should be no more than 1 mg/kg/hour (2.2)
- Reconstitute and dilute LUMIZYME prior to use. (2.3)
- See full prescribing information for storage of the reconstituted and diluted product and administration instructions (2.4, 2.5)
Dosage Forms and Strengths
For injection: 50 mg of LUMIZYME as lyophilized powder in a single-dose vial for reconstitution. (3)
Contraindications
None. (4)
Warnings and Precautions
- Infusion-Associated Reactions (IARs): If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administration of antihistamines and/or antipyretics may ameliorate the symptoms. (5.2)
- Risk of Cardiac Arrhythmia and Sudden Cardiac Death during General Anesthesia for Central Venous Catheter Placement: Caution should be used when administering general anesthesia for the placement of a central venous catheter intended for LUMIZYME infusion. (5.5)
- Risk of Developing Anti-alglucosidase Alfa Antibodies: Patients with IOPD should have a cross-reactive immunologic material (CRIM) assessment early in their disease course and be managed by a clinical specialist knowledgeable in immune tolerance induction in Pompe disease. (5.6)
Adverse Reactions/Side Effects
The most frequently reported adverse reactions (≥5%) in clinical trials were hypersensitivity reactions and included: anaphylaxis, rash, pyrexia, flushing/feeling hot, urticaria, headache, hyperhidrosis, nausea, cough, decreased oxygen saturation, tachycardia, tachypnea, chest discomfort, dizziness, muscle twitching, agitation, cyanosis, erythema, hypertension/increased blood pressure, pallor, rigors, tremor, vomiting, fatigue, and myalgia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme at 1-800-633-1610, option 1 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 12/2024
Full Prescribing Information
WARNING: HYPERSENSITIVITY REACTIONS INCLUDING ANAPHYLAXIS, IMMUNE-MEDIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE
Hypersensitivity Reactions Including Anaphylaxis
Patients treated with enzyme replacement therapies have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate LUMIZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LUMIZYME and immediately initiate appropriate medical treatment, including use of epinephrine. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and to seek immediate medical care should symptoms occur [see Warnings and Precautions (5.1)].
Immune-Mediated Reactions
Immune-mediated reactions presenting as proteinuria, nephrotic syndrome, and necrotizing skin lesions have occurred in some patients following LUMIZYME treatment. Monitor patients for the development of systemic immune-mediated reactions involving skin and other organs while receiving LUMIZYME [see Warnings and Precautions (5.3)].
Risk of Acute Cardiorespiratory Failure
Infantile-onset Pompe disease (IOPD) patients with compromised cardiac or respiratory function may be at risk of serious acute exacerbation of their cardiac or respiratory compromise due to fluid overload and require additional monitoring [see Warnings and Precautions (5.4)].
1. Indications and Usage for Lumizyme
LUMIZYME® is a hydrolytic lysosomal glycogen-specific enzyme indicated for patients with Pompe disease (acid α-glucosidase [GAA] deficiency).
2. Lumizyme Dosage and Administration
2.1 Recommendations prior to LUMIZYME Treatment
- Administration of LUMIZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis [see Warnings and Precautions (5.1)].
- Initiate LUMIZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment [see Warnings and Precautions (5.1)].
- Prior to LUMIZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids [see Warnings and Precautions (5.1, 5.2)].
- LUMIZYME must be reconstituted and diluted prior to use [see Dosage and Administration (2.3)].
- Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during LUMIZYME administration [see Warnings and Precautions (5.1)].
2.2 Recommended Dosage and Administration
- The recommended dosage of LUMIZYME is 20 mg/kg body weight administered every 2 weeks as an intravenous infusion. The initial infusion rate should be no more than 1 mg/kg/hour [see Dosage and Administration (2.5)].
2.3 Reconstitution and Dilution Instructions
Reconstitute and dilute LUMIZYME in the following manner.
Use aseptic technique during preparation. Do not use filter needles during preparation.
Reconstitute the Lyophilized Powder
- Determine the number of LUMIZYME vials to be reconstituted based on the actual body weight in kg and the recommended dose of 20 mg/kg. Round the number of vials up to the next whole number.
- Remove the required number of LUMIZYME vials from the refrigerator and allow the vials to sit for approximately 30 minutes at room temperature 20°C to 25°C (68°F to 77°F) prior to reconstitution.
- Reconstitute each vial by slowly injecting 10.3 mL of Sterile Water for Injection, down the inside wall of each vial. Avoid adding the Sterile Water for Injection to the vial forcefully or directly onto the lyophilized powder to minimize foaming.
- Gently tilt and roll each vial. Do not invert, swirl, or shake the vial. Each vial will yield a concentration of 5 mg/mL of LUMIZYME. The total extractable dose per vial is 50 mg per 10 mL.
- Visually inspect the reconstituted solution in the vials for particulate matter and discoloration. Discard if particles are present or the solution is discolored. The reconstituted solution may occasionally contain some LUMIZYME particles (typically less than 10 in a vial) in the form of thin white strands or translucent fibers subsequent to the initial inspection. This may also happen following dilution for infusion. These particles have been shown to contain LUMIZYME and may appear after the initial reconstitution step and increase over time. Studies have shown that these particles are removed via in-line filtration without having a detectable effect on the purity or strength.
Dilute the Reconstituted Solution
- Select and prepare an appropriate size 0.9% Sodium Chloride for Injection infusion bag with quantity sufficient of 0.9% Sodium Chloride for Injection to obtain the recommended total infusion volume per table 1 based on patient weight and dilute.
- Slowly withdraw the required volume of reconstituted solution from the LUMIZYME vial(s). Avoid foaming in the syringe. Discard any unused reconstituted solution remaining in the vial.
- Remove airspace from the prepared 0.9% Sodium Chloride for Injection infusion bag to minimize particle formation due to the sensitivity of LUMIZYME to air-liquid interfaces.
- Inject the LUMIZYME reconstituted solution slowly and directly into the port of the prepared 0.9% Sodium Chloride for Injection infusion bag. Avoid foaming and introducing air in the infusion bag.
- Gently invert or massage the infusion bag to mix the solution. Do not shake. After dilution, the solution will have a final concentration of 0.5 to 4 mg/mL of LUMIZYME.
2.4 Storage Instructions for the Reconstituted and Diluted Product
- The reconstituted and diluted solution should be administered without delay. Storage of the reconstituted solution at room temperature is not recommended.
- If immediate use is not possible, the reconstituted and diluted solution is stable for up to 24 hours refrigerated at 2°C to 8°C (36°F to 46°F).
- The reconstituted and diluted LUMIZYME solution should be protected from light.
- Do not freeze or shake.
2.5 Administration Instructions
- The total volume of infusion is determined by the patient's body weight and should be administered over approximately 4 hours.
- Administer LUMIZYME using an in-line low protein binding 0.2-micron filter.
- Infusions should be administered in a step-wise manner using an infusion pump. The initial infusion rate should be no more than 1 mg/kg/hr. The infusion rate may be increased by 2 mg/kg/hr every 30 minutes, after patient tolerance to the infusion rate is established, until a maximum rate of 7 mg/kg/hr is reached.
- Vital signs should be obtained at the end of each step. If the patient is stable, LUMIZYME may be administered at the maximum rate of 7 mg/kg/hr until the infusion is completed.
- The infusion rate may be slowed or temporarily stopped in the event of mild to moderate hypersensitivity reactions. In the event of anaphylaxis or severe hypersensitivity reaction, immediately discontinue administration of LUMIZYME and initiate appropriate medical treatment. See Table 1 below for the rate of infusion at each step, expressed as mL/hr based on the recommended infusion volume by patient weight.
- Do not infuse LUMIZYME in the same intravenous line with other products. Discard any unused product.
| Patient Weight Range | Total infusion volume | Step 1 1 mg/kg/hr | Step 2 3 mg/kg/hr | Step 3 5 mg/kg/hr | Step 4 7 mg/kg/hr |
|---|---|---|---|---|---|
| Infusion Rate in mL/hr | |||||
| 1.25 to 2.5 kg | 25 mL | 1.25 | 3.75 | 6.25 | 6.6 |
| 2.6 to 10 kg | 50 mL | 3 | 8 | 13 | 18 |
| 10.1 to 20 kg | 100 mL | 5 | 15 | 25 | 35 |
| 20.1 to 30 kg | 150 mL | 8 | 23 | 38 | 53 |
| 30.1 to 35 kg | 200 mL | 10 | 30 | 50 | 70 |
| 35.1 to 50 kg | 250 mL | 13 | 38 | 63 | 88 |
| 50.1 to 60 kg | 300 mL | 15 | 45 | 75 | 105 |
| 60.1 to 100 kg | 500 mL | 25 | 75 | 125 | 175 |
| 100.1 to 120 kg | 600 mL | 30 | 90 | 150 | 210 |
| 120.1 to 140 kg | 700 mL | 35 | 105 | 175 | 245 |
| 140.1 to 160 kg | 800 mL | 40 | 120 | 200 | 280 |
| 160.1 to 180 kg | 900 mL | 45 | 135 | 225 | 315 |
| 180.1 to 200 kg | 1,000 mL | 50 | 150 | 250 | 350 |
3. Dosage Forms and Strengths
For injection: 50 mg of LUMIZYME is supplied as a sterile, nonpyrogenic, white to off-white, lyophilized cake or powder in a single-dose vial for reconstitution. After reconstitution, the resultant solution concentration is 5 mg/mL.
5. Warnings and Precautions
5.1 Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity reactions including anaphylaxis have been observed in patients during and up to 3 hours after a LUMIZYME infusion. Some of the hypersensitivity reactions were life-threatening and included anaphylactic shock, cardiac arrest, respiratory arrest, respiratory distress, hypoxia, apnea, dyspnea, bradycardia, tachycardia, bronchospasm, throat tightness, hypotension, angioedema (including tongue or lip swelling, periorbital edema, and face edema), and urticaria. Other accompanying reactions included chest discomfort/pain, wheezing, tachypnea, cyanosis, decreased oxygen saturation, convulsions, pruritus, rash, hyperhidrosis, nausea, dizziness, hypertension/increased blood pressure, flushing/feeling hot, erythema, pyrexia, pallor, peripheral coldness, restlessness, nervousness, headache, back pain, and paresthesia. Some of these reactions were IgE-mediated.
In clinical trials, hypersensitivity reactions including anaphylaxis were managed with infusion interruption; decreased infusion rate; and administration of antihistamines, corticosteroids, intravenous fluids, and/or oxygen. In some cases of anaphylaxis, epinephrine was administered. Some LUMIZYME-treated patients who experienced a hypersensitivity reaction and who tested positive for alglucosidase alfa-specific IgE antibodies were successfully rechallenged with a slower infusion rate at a lower dosage of LUMIZYME and continued to receive LUMIZYME under close clinical supervision. Because LUMIZYME-treated patients who develop anti-IgE alglucosidase alfa antibodies appear to be at a higher risk for developing hypersensitivity reactions including anaphylaxis, these patients should be monitored more closely during LUMIZYME administration.
Recommendations to Prevent, Mitigate, and Monitor for Hypersensitivity Reactions
Prior to LUMIZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. Administration of LUMIZYME should be supervised by a healthcare provider knowledgeable in the management of hypersensitivity reactions including anaphylaxis. Anaphylaxis has occurred during the early course of enzyme replacement therapy and after extended duration of therapy. Initiate LUMIZYME in a healthcare setting with appropriate medical monitoring and support measures, including access to cardiopulmonary resuscitation equipment.
- If a severehypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue LUMIZYME and immediately initiate appropriate medical treatment, including use of epinephrine.
- If a mild or moderate hypersensitivity reaction occurs, consider temporarily holding the LUMIZYME infusion or slowing the infusion rate.
Consider the risks and benefits of readministering LUMIZYME following a hypersensitivity reaction including anaphylaxis. Patients may be rechallenged using slower infusion rates at a lower dosage than the recommended dosage [see Adverse Reactions (6.2)]. Inform patients of the symptoms of life-threatening hypersensitivity reactions, including anaphylaxis and recommend they seek immediate medical care should these symptoms occur.
5.2 Infusion-Associated Reactions
Infusion-associated reactions (IARs) such as pyrexia, chills, flu-like illness, myalgia, arthralgia, pain, fatigue, urticaria, rash, pruritus, erythema, dyspnea, tachycardia, flushing, nausea, headache and syncope occurred in LUMIZYME-treated patients [see Adverse Reactions (6.2)].
If an IAR occurs, decreasing the infusion rate, temporarily stopping the infusion, and/or administering antihistamines and/or antipyretics may ameliorate the symptoms. Closely monitor patients who have experienced IARs when re-administering LUMIZYME.
5.3 Immune-Mediated Reactions
Immune-mediated cutaneous reactions have been reported with LUMIZYME including necrotizing skin lesions [see Adverse Reactions (6.2)]. Systemic immune-mediated reactions, including possible type III immune-mediated reactions have been observed with LUMIZYME. These reactions occurred several weeks to 3 years after initiation of LUMIZYME infusions. Skin biopsy in one patient demonstrated deposition of anti-rhGAA antibodies in the lesion. Another patient developed severe inflammatory arthropathy in association with pyrexia and elevated erythrocyte sedimentation rate. Nephrotic syndrome secondary to membranous glomerulonephritis was observed in some LUMIZYME-treated patients with Pompe disease who had persistently positive anti-rhGAA IgG antibody titers. In these patients, renal biopsy was consistent with immune complex deposition. Patients improved following LUMIZYME treatment interruption [see Adverse Reactions (6.2)].
LUMIZYME treated patients should be monitored for the development of systemic immune-mediated reactions involving skin and other organs including periodic urinalysis. If immune-mediated reactions occur, consider discontinuing the LUMIZYME administration, and initiating appropriate medical treatment. The risks and benefits of readministering LUMIZYME following an immune-mediated reaction should be considered. Some patients have been able to be rechallenged and have continued to receive LUMIZYME under close clinical supervision. Immune tolerance induction administered in conjunction with LUMIZYME may also aide tolerability of LUMIZYME under the management of a clinical specialist knowledgeable in immune tolerance induction in pediatric patients with IOPD or LOPD.
5.4 Risk of Acute Cardiorespiratory Failure
Patients with acute underlying respiratory illness or compromised cardiac and/or respiratory function may be at risk of serious exacerbation of their cardiac or respiratory compromise during infusions. Appropriate medical support and monitoring measures should be readily available during LUMIZYME infusion, and some patients may require prolonged observation times that should be individualized based on the needs of the patient. Acute cardiorespiratory failure has been observed in infantile-onset Pompe disease patients with underlying cardiac hypertrophy, possibly associated with fluid overload with intravenous administration of LUMIZYME [see Dosage and Administration (2.2)].
5.5 Risk of Cardiac Arrhythmia and Sudden Cardiac Death during General Anesthesia for Central Venous Catheter Placement
Administration of general anesthesia can be complicated by the presence of severe cardiac and skeletal (including respiratory) muscle weakness. Therefore, caution should be used when administering general anesthesia. Ventricular arrhythmias and bradycardia, resulting in cardiac arrest or death, or requiring cardiac resuscitation or defibrillation have been observed in patients with IOPD with cardiac hypertrophy during general anesthesia for central venous catheter placement.
5.6 Risk of Developing Anti-alglucosidase Alfa Antibodies
As shown from clinical trials and published literature, individualized immune tolerance induction regimen administered prior to and with initiation of LUMIZYME has been reported to aid tolerability of LUMIZYME and reduce the development of high ADA titers in CRIM-negative IOPD patients. Furthermore, CRIM status has been shown to be associated with immunogenicity and patients' responses to LUMIZYME. LUMIZYME-treated infants with IOPD who are CRIM-negative (indicating no endogenous enzyme is detected) have shown poorer clinical response (loss of motor function, ventilator dependence, or death) in the presence of high sustained IgG ADA titers and positive inhibitory antibodies compared to CRIM-positive infants [see Adverse Reactions (6.2)]. However, high and sustained ADA titers has also occurred in a limited number of CRIM-positive patients, generally with very low endogenous enzyme [see Clinical Pharmacology (12.6)]. Therefore, these patients must be managed by a clinical specialist knowledgeable in immune tolerance induction in Pompe disease.
Some alglucosidase alfa-treated patients who developed high sustained IgG ADA titers had reduced efficacy. Some alglucosidase alfa-treated patients with high IgG ADA titers had a higher incidence of IARs.
Patients with IOPD should have a cross-reactive immunologic material (CRIM) assessment early in their disease course.
Anti-alglucosidase alfa antibody (referred to as ADA) titers should be obtained during LUMIZYME treatment. Contact Genzyme Corporation at 1-800-745-4447 for information on ADA testing.
Recommend the following ADA testing:
- Baseline serum ADA sample collection prior to the first LUMIZYME infusion is strongly encouraged.
-
For patients with:
- IOPD, suggest regular ADA monitoring during first year of treatment (example: every 3 months).
- LOPD, suggest ADA be monitored within six months of LUMIZYME initiation with subsequent monitoring as clinically warranted based on safety and efficacy considerations.
-
If patients [see Adverse Reactions (6.2) and Clinical Pharmacology (12.6)]:
- Develop hypersensitivity reactions, consider testing for IgG ADA, IgE ADA and other mediators.
- Develop immune-mediated reactions that are not hypersensitivity reactions, consider testing for IgG ADA.
- Lose or have a reduced clinical response, consider testing for IgG ADA and for inhibitory antibody activity.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are described below and elsewhere in the labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In clinical trials, the most common adverse reactions (≥5%) following intravenous alglucosidase alfa treatment were hypersensitivity reactions, and included anaphylaxis, rash, pyrexia, flushing/feeling hot, urticaria, headache, hyperhidrosis, nausea, cough, decreased oxygen saturation, tachycardia, tachypnea, chest discomfort, dizziness, muscle twitching, agitation, cyanosis, erythema, hypertension/increased blood pressure, pallor, rigors, tremor, vomiting, fatigue, and myalgia.
Adverse Reactions in Clinical Trials in Infantile-Onset and Juvenile-Onset Pompe Disease
Two multicenter, open-label clinical trials (Trials 1 and 2) [see Clinical Studies (14.1)] were conducted in 39 patients with infantile-onset Pompe disease (IOPD), aged 1 month to 3.5 years old. Approximately half of the patients (54%) were male. Patients were treated with intravenous alglucosidase alfa 20 or 40 mg/kg every other week for periods ranging from 1 to 106 weeks (mean: 61 weeks).
The most serious adverse reactions reported with alglucosidase alfa treatment included anaphylaxis and acute cardiorespiratory failure.
The most common adverse reactions requiring intervention in these clinical trials were hypersensitivity reactions, that occurred in 20 of 39 (51%) patients treated with alglucosidase alfa, and included rash, pyrexia, urticaria, flushing, decreased oxygen saturation, cough, tachypnea, tachycardia, hypertension/increased blood pressure, pallor, rigors, vomiting, cyanosis, agitation, and tremor. These reactions were more likely to occur with higher infusion rates or doses. Some patients who were pretreated with antihistamines, antipyretics and/or corticosteroids still experienced hypersensitivity reactions.
Table 2 summarizes all adverse reactions that occurred in ≥5% of patients (2 or more patients) treated with alglucosidase alfa in clinical trials described above.
| Number of Patients (N=39) n (%) |
|
|---|---|
| Adverse Reaction | 20 (51) |
| Rash (including rash erythematous, rash macular and maculopapular) | 7 (18) |
| Pyrexia | 6 (15) |
| Urticaria | 5 (13) |
| Flushing | 5 (13) |
| Hypertension/Increased Blood Pressure | 4 (10) |
| Decreased Oxygen Saturation | 3 (8) |
| Cough | 3 (8) |
| Tachypnea | 3 (8) |
| Tachycardia | 3 (8) |
| Erythema | 2 (5) |
| Vomiting | 2 (5) |
| Rigors | 2 (5) |
| Pallor | 2 (5) |
| Cyanosis | 2 (5) |
| Agitation | 2 (5) |
| Tremor | 2 (5) |
An open-label, single-center trial (Trial 3) was conducted in 18 treatment-naive patients with IOPD who were treated with alglucosidase alfa [see Clinical Studies (14.1)]. Adverse reactions observed in these patients were similar to patients with IOPD who received alglucosidase alfa in other clinical trials.
Additional hypersensitivity reactions observed in patients with IOPD treated with alglucosidase alfa in other clinical trials and expanded access programs included livedo reticularis, irritability, retching, increased lacrimation, ventricular extrasystoles, nodal rhythm, rales, respiratory tract irritation, and cold sweat.
Safety was also evaluated in 99 patients (51 male, 48 females) with Pompe disease in an ongoing, open-label, prospective study in patients 12 months of age and older who were previously treated with another alglucosidase alfa product and switched to LUMIZYME. Patients were aged 1 to 18 years with a median duration of treatment of 437 days (range 13 to 466 days). No new safety findings were observed following the switch to 4000 L scale of alglucosidase alfa.
Adverse Reactions in Clinical Trials in Late-Onset Pompe Disease
Assessment of adverse reactions in patients with late-onset Pompe disease (LOPD) is based on the exposure of 90 patients (45 male, 45 female), aged 10 to 70 years, to intravenous infusions of 20 mg/kg alglucosidase alfa or placebo in a randomized, double-blind, placebo-controlled trial (Trial 4). All patients were naive to enzyme replacement therapy. Patients were randomized in a 2:1 ratio and received intravenous alglucosidase alfa or placebo every other week for 78 weeks (18 months). Two patients who received alglucosidase alfa discontinued the trial due to anaphylactic reactions.
Serious adverse reactions reported with alglucosidase alfa included anaphylaxis, which presented as angioedema, throat tightness and chest pain/discomfort. One patient with a history of Wolff-Parkinson-White syndrome experienced a serious adverse reaction of supraventricular tachycardia.
The most common adverse reactions (≥3%; 2 or more patients) observed in alglucosidase alfa-treated patients were hypersensitivity reactions and included anaphylaxis, headache, nausea, urticaria, dizziness, chest discomfort, vomiting, hyperhidrosis, flushing/feeling hot, increased blood pressure, paresthesia, pyrexia, local swelling, diarrhea, pruritus, rash, and throat tightness.
Delayed-onset reactions, defined as adverse reactions that occurred 2 to 48 hours after completion of alglucosidase alfa infusion, that were observed in ≥3% more patients in the alglucosidase alfa-treated group compared to patients in the placebo-treated group in the controlled trial, included hyperhidrosis. Additional delayed-onset reactions that occurred in alglucosidase alfa-treated patients included fatigue, myalgia, and nausea.
Table 3 summarizes the most common adverse reactions that occurred in at least 3% of alglucosidase alfa-treated patients and with a higher incidence than the placebo-treated patients in Trial 4 (patients with LOPD).
| Adverse Reaction | Alglucosidase Alfa n=60 N (%) | Placebo n=30 N (%) |
|---|---|---|
|
||
| Hyperhidrosis | 5 (8.3) | 0 (0) |
| Urticaria | 5 (8.3) | 0 (0) |
| Anaphylaxis | 4 (6.7) | 0 (0) |
| Chest Discomfort | 4 (6.7) | 1 (3.3) |
| Muscle Twitching | 4 (6.7) | 1 (3.3) |
| Myalgia | 3 (5.0) | 1 (3.3) |
| Flushing/Feeling Hot | 3 (5.0) | 0 (0) |
| Increased Blood Pressure | 3 (5.0) | 0 (0) |
| Vomiting | 3 (5.0) | 0 (0) |
| Edema, Peripheral | 2 (3.3) | 0 (0) |
| Pruritus | 2 (3.3) | 0 (0) |
| Rash Papular | 2 (3.3) | 0 (0) |
| Throat Tightness | 2 (3.3) | 0 (0) |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of alglucosidase alfa. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In the postmarketing experience with LUMIZYME, serious adverse reactions have been reported, including anaphylaxis [see Boxed Warning and Warnings and Precautions (5.1)]. Acute cardiorespiratory failure, possibly associated with fluid overload, has been reported in infantile-onset Pompe disease patients with pre-existing hypertrophic cardiomyopathy [see Boxed Warning and Warning and Precautions (5.3)].
Infusion associated reactions, including pyrexia, chills, fatigue, urticaria, rash, pruritus, erythema, dyspnea, hypotension, bradycardia, tachycardia, flushing, nausea, headache, and syncope have been reported with alglucosidase alfa.
In addition to the hypersensitivity reactions reported in clinical trials [see Adverse Reactions (6.1)], the following hypersensitivity reactions have been reported in at least 2 patients and included: anaphylactic shock, respiratory failure, respiratory arrest, cardiac arrest, hypoxia, dyspnea, wheezing, convulsions, peripheral coldness, restlessness, nervousness, back pain, stridor, pharyngeal edema, abdominal pain, apnea, muscle spasm, and conjunctivitis. In addition, one case of hyperparathyroidism has been reported.
Systemic and cutaneous immune-mediated reactions, including proteinuria and nephrotic syndrome secondary to membranous glomerulonephritis, and necrotizing skin lesions have been reported in postmarketing safety experience with alglucosidase alfa [see Warnings and Precautions (5.2)].
Related/similar drugs
8. Use In Specific Populations
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to LUMIZYME during pregnancy. Pregnant women and women of reproductive potential should be encouraged to enroll in the Pompe patient registry. The registry will monitor the effect of LUMIZYME on pregnant women and their offspring. For more information, visit www.registrynxt.com or call 1-800-745-4447, extension 15500.
Risk Summary
Data from postmarketing reports and published case reports with alglucosidase alfa use in pregnant women have not identified a LUMIZYME-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. The continuation of treatment for Pompe disease during pregnancy should be individualized to the pregnant woman. Untreated Pompe disease may result in worsening disease symptoms in pregnant women (see Clinical Considerations).
Reproduction studies performed in mice and rabbits at doses resulting in exposures up to 0.4 or 0.5 times the human steady-state AUC (area under the plasma concentration-time curve), respectively, during the period of organogenesis revealed no evidence of effects on embryo-fetal development. In mice there was an increase in pup mortality during lactation at maternal exposures 0.4 times the human steady-state AUC (see Data).
The background risk of major birth defects and miscarriage in the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
All reproductive studies included pretreatment with diphenhydramine to prevent or minimize hypersensitivity reactions. The effects of alglucosidase alfa were evaluated based on comparison to a control group treated with diphenhydramine alone. Daily intravenous administration of alglucosidase alfa up to 40 mg/kg in mice and rabbits (0.4 and 0.5 times the human steady-state AUC, respectively, at the recommended biweekly dose) during the period of organogenesis had no effects on embryo-fetal development. Administration of 40 mg/kg intravenously every other day in mice (0.4 times the human steady-state AUC at the recommended biweekly dose) during the period of organogenesis through lactation produced an increase in mortality of offspring during the lactation period.
8.2 Lactation
Risk Summary
Available published literature suggests the presence of alglucosidase alfa in human milk. There are no reports of adverse effects of alglucosidase alfa on the breastfed infant. There is no information on the effects of alglucosidase alfa on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for LUMIZYME and any potential adverse effects on the breastfed child from LUMIZYME or from the underlying maternal condition.
Lactating women with Pompe disease treated with LUMIZYME should be encouraged to enroll in the Pompe disease registry [see Use in Specific Populations (8.1)].
8.4 Pediatric Use
The safety and effectiveness of LUMIZYME has been established in pediatric patients with Pompe disease [see Adverse Reactions (6.2)].The use of LUMIZYME for this pediatric indication is supported by evidence from an adequate and well-controlled trial in 57 treatment-naive pediatric patients with IOPD treated with alglucosidase alfa, aged 0.2 month to 3.5 years at first infusion, (Trials 1, 2, and 3) [see Clinical Studies (14.1)] and 90 adult and pediatric patients with LOPD in a randomized, double-blind, placebo-controlled trial including 2 patients 16 years of age or less [see Clinical Studies (14.2)].
Anaphylaxis, hypersensitivity reactions, and acute cardiorespiratory failure have occurred in pediatric patients [see Boxed Warning, Warnings and Precautions (5.1, 5.3)]. Additionally, cardiac arrhythmia and sudden cardiac death have occurred in pediatric patients during general anesthesia for central venous catheter placement [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
The randomized, double-blind, placebo-controlled study of alglucosidase alfa did not include sufficient numbers (n=4) of patients aged 65 years and over to determine whether they respond differently from younger adult patients [see Clinical Studies (14.1)].
11. Lumizyme Description
Alglucosidase alfa is a hydrolytic lysosomal glycogen-specific enzyme encoded by the predominant of nine observed haplotypes of the human acid α-glucosidase (GAA) gene. Alglucosidase alfa is produced by recombinant DNA technology in a Chinese hamster ovary cell line. Alglucosidase alfa degrades glycogen by catalyzing the hydrolysis of α-1,4- and α-1,6- glycosidic linkages of lysosomal glycogen.
Alglucosidase alfa is a glycoprotein with a calculated mass of 99,377 Daltons for the polypeptide chain, and a total mass of approximately 109,000 Daltons, including carbohydrates. Alglucosidase alfa has a specific activity of 3.6 to 5.4 units/mg (one unit is defined as that amount of activity that results in the hydrolysis of 1 micromole of synthetic substrate per minute under specified assay conditions). Alglucosidase alfa is intended for intravenous infusion. It is supplied as a sterile, nonpyrogenic, white to off-white, lyophilized cake or powder for reconstitution with 10.3 mL Sterile Water for Injection, USP. Each 50 mg vial contains 52.5 mg alglucosidase alfa, 210 mg mannitol, 0.5 mg polysorbate 80, 9.9 mg sodium phosphate dibasic heptahydrate, and 31.2 mg sodium phosphate monobasic monohydrate. Following reconstitution as directed, each vial contains 10.5 mL reconstituted solution and a total extractable volume of 10 mL at 5 mg/mL alglucosidase alfa. Alglucosidase alfa does not contain preservatives; each vial is for single dose only.
12. Lumizyme - Clinical Pharmacology
12.1 Mechanism of Action
Pompe disease (acid maltase deficiency, glycogen storage disease type II, GSD II, glycogenosis type II) is an inherited disorder of glycogen metabolism caused by the absence or marked deficiency of the lysosomal enzyme GAA.
Alglucosidase alfa provides an exogenous source of GAA. Binding to mannose-6-phosphate receptors on the cell surface has been shown to occur via carbohydrate groups on the GAA molecule, after which it is internalized and transported into lysosomes, where it undergoes proteolytic cleavage that results in increased enzymatic activity. It then exerts enzymatic activity in cleaving glycogen.
12.2 Pharmacodynamics
Clinical pharmacodynamic studies have not been conducted for alglucosidase alfa.
12.3 Pharmacokinetics
The pharmacokinetics of alglucosidase alfa was evaluated in 13 patients with infantile-onset Pompe disease, aged 1 month to 7 months, who received 20 mg/kg (approximately as a 4-hour infusion) or 40 mg/kg (approximately as a 6.5-hour infusion) of alglucosidase alfa every 2 weeks. The measurement of alglucosidase alfa plasma concentration was based on an activity assay using an artificial substrate. Systemic exposure was approximately dose proportional between the 20 and 40 mg/kg doses. Based on the pharmacokinetic blood samples collected for 12 hours after a 4-hour intravenous infusion of 20 mg/kg (n=5), the estimated mean AUC was 811 mcg∙hr/mL with 17% coefficient of variation [CV], Cmax was 162 mcg/mL with 19% CV, clearance was 25 mL/hr/kg with 16% CV, and half-life was 2.3 hours with 17% CV.
The pharmacokinetics of alglucosidase alfa was also evaluated in a separate trial of 14 patients with infantile-onset Pompe disease, aged 6 months to 3.5 years, who received 20 mg/kg of alglucosidase alfa as a 4-hour infusion every 2 weeks. The pharmacokinetic parameters were similar to those observed for the infantile-onset Pompe disease patients aged 1 month to 7 months who received the 20 mg/kg dose.
The pharmacokinetics of alglucosidase alfa was evaluated in another trial of 10 adult and 10 pediatric patients with Pompe disease who received a single dose of 20 mg/kg of alglucosidase alfa as a 4-hour infusion. In pediatric patients, aged 7 months to 13.7 years, the estimated mean AUC was 1,110 mcg∙hr/mL with 68% CV and Cmax was 204 mcg/mL with 46% CV. In adult patients, aged 19 to 57 years, the estimated mean AUC was 1,890 mcg∙hr/mL with 51% CV and Cmax was 307 mcg/mL with 47% CV.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADAs in the studies described below with the incidence of ADAs in other studies, including those of LUMIZYME or of other alglucosidase alfa products.
Immunogenicity in Patients with Infantile-onset Pompe Disease
Thirty-nine patients with infantile-onset Pompe disease (IOPD), aged 1 month to 3.5 years, were treated with 20 or 40 mg/kg of intravenous alglucosidase alfa every other week for periods that ranged from 1 to 106 weeks (mean: 61 weeks) in two open-label clinical trials (Trials 1 and 2) [see Clinical Studies (14.1, 14.2)]. In Trials 1 and 2, 34/38 (89%) of alglucosidase alfa-treated patients tested positive for anti-alglucosidase alfa antibodies (referred to as ADA).
Immunogenicity in Patients with Late-onset Pompe Disease
Ninety patients with late-onset Pompe disease (LOPD), aged 10 to 70 years, were administered 20 mg/kg of intravenous alglucosidase alfa or intravenous placebo every other week (2:1 ratio) for 78 weeks in a randomized, double-blinded, placebo-controlled trial (Trial 4) [see Clinical Studies (14.2)]. In Trial 4, 59/59 (100%) of alglucosidase alfa-treated patients with available samples developed ADA. These patients were all cross-reactive immunologic material (CRIM) positive. Most patients who developed ADA did so within the first three months of exposure (median time to seroconversion was four weeks).
Antibody titers for cellular uptake inhibition were present in 18 of 59 (31%) alglucosidase alfa-treated patients by Week 78. Patients who tested positive for cellular uptake inhibition tended to have higher IgG titers than patients who tested negative for cellular uptake inhibition. None of the 59 evaluable patients tested positive for inhibition of enzyme activity.
Immunogenicity in Patients with Infantile-onset Pompe Disease or Late-onset Pompe Disease
Some patients with IOPD or LOPD who were ADA positive in Trials 1, 2 and 4 tested positive for inhibition of enzyme activity and/or uptake in in vitro assays. However, the clinical relevance of this in vitro inhibition is unclear.
Negative CRIM status (indicating no endogenous enzyme is detected) is a risk factor for LUMIZYME-treated patients to develop high and sustained ADA titers. However, high and sustained ADA titers have also occurred in a limited number of CRIM-positive patients, generally with very low endogenous enzyme.
Anti-Drug Antibody Effects on Pharmacokinetics
In Trials 1 and 2, 19 of 21 (90%) alglucosidase alfa-treated patients with IOPD - who had pharmacokinetics (PK) and antibody titer data available at Week 12 - developed ADA. Of these 19 patients, 5 patients with antibody titers ≥12,800 at Week 12 had a 50% mean increase in clearance (range 5% to 90%) from Week 1 to Week 12, while the 14 patients with antibody titers <12,800 at Week 12 had no significant change in the mean clearance values at Week 1 and Week 12.
In Trial 4, 5/32 (16%) of alglucosidase alfa-treated patients with LOPD that had evaluable PK samples tested positive for cellular uptake inhibition. An approximately 1.2-fold to 1.8- fold greater clearance was observed at Week 52 in 4 of 5 patients that tested positive for antibodies that inhibit the cellular uptake of enzyme in an in vitro assay compared to Week 0. PK in 4 of these 5 patients over time indicated an increase in clearance with increase in IgG titer.
Anti-Drug Antibody Effects on Safety and Efficacy
A small number of alglucosidase alfa-treated patients with LOPD who tested positive for alglucosidase alfa-specific IgE antibodies experienced anaphylactic reactions [see Warnings and Precautions (5.1)].
Some alglucosidase alfa-treated patients who developed high sustained ADA titers had reduced efficacy. Furthermore, CRIM-negative infants have shown reduced clinical effect in the presence of high sustained ADA titers with inhibitory activity [see Warnings and Precautions (5.6)].
There was no identified clinically significant effect of high ADA titers on the development of IARs in LUMIZYME-treated patients.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential or studies to evaluate mutagenic potential have not been performed with alglucosidase alfa.
Intravenous administration of alglucosidase alfa every other day in mice at doses up to 40 mg/kg (0.4 times the human AUC at the recommended biweekly dose) had no effect on fertility and reproductive performance.
14. Clinical Studies
14.1 Clinical Trials in Infantile-Onset Pompe Disease
The safety and effectiveness of alglucosidase alfa in the treatment patients with infantile-onset Pompe disease (IOPD) were assessed in 57 treatment-naive infantile-onset Pompe disease patients, aged 0.2 month to 3.5 years at first alglucosidase alfa infusion, in three separate open-label, single-arm clinical trials (Trials 1, 2, and 3).
Trial 1 (Patients with IOPD)
Trial 1 was an international, multicenter, open-label, clinical trial of 18 patients with IOPD. This trial was conducted between 2003 and 2005. Patients were randomized 1:1 to receive either 20 mg/kg or 40 mg/kg of intravenous alglucosidase alfa every two weeks, with length of treatment ranging from 52 to 106 weeks. Enrollment was restricted to patients 7 months of age or younger at first infusion with clinical signs of Pompe disease and cardiac hypertrophy, and who did not require ventilatory support at trial entry. Fourteen patients were cross reactive immunologic material (CRIM) positive, and 4 patients were CRIM negative.
Efficacy was assessed by comparing the proportions of alglucosidase alfa-treated patients who died or needed invasive ventilator support at 18 months of age with the mortality experience of a historical cohort of untreated patients with IOPD with similar age and disease severity. In the historical cohort, 61 untreated patients with IOPD diagnosed by age 6 months, born between 1982 and 2002, were identified by a retrospective review of medical charts. By 18 months of age, 15 of 18 (83%) alglucosidase alfa-treated patients were alive without invasive ventilatory support and 3 (17%) required invasive ventilator support, whereas only one of the 61 (2%) historical control patients was alive. No differences in outcome were observed between patients who received 20 mg/kg versus 40 mg/kg.
Other outcome measures in this trial included unblinded assessments of motor function by the Alberta Infant Motor Scale (AIMS), a measure of infant motor performance that assesses motor maturation of the infant through age 18 months. Although gains in motor function were noted in 13 patients, the motor function was substantially delayed compared to normal infants of comparable age in the majority of patients. Two of 9 patients who had initially demonstrated gains in motor function after 12 months of alglucosidase alfa treatment regressed despite continued treatment.
Changes from baseline to Month 12 in left ventricular mass index (LVMI), a measure of pharmacodynamic effect, were evaluated by echocardiography. Fifteen patients who underwent both baseline and Month 12 echocardiograms demonstrated decreases from baseline in LVMI (mean decrease 118 g/m2, range 45 to 193 g/m2). However, the magnitude of the decrease in LVMI did not correlate with the clinical outcome measure of ventilator-free survival.
Trial 2 (Patients with IOPD)
Trial 2 was an international, multicenter, non-randomized, open-label clinical trial that enrolled 21 patients with IOPD aged 3 months to 3.5 years at first infusion. Eighteen patients were CRIM positive and 3 patients were CRIM negative. All patients received intravenous 20 mg/kg alglucosidase alfa every other week for up to 104 weeks. Five of 21 patients were receiving invasive ventilatory support at the time of first infusion.
The primary outcome measure was the proportion of patients alive at the conclusion of treatment. At the 52-week interim analysis, 16 of 21 patients were alive. Sixteen patients were free of invasive ventilatory support at the time of first infusion; of these, 4 died, 2 required invasive ventilatory support, and 10 were free of invasive ventilatory support after 52 weeks of treatment. For the 5 patients who were receiving invasive ventilatory support at baseline, 1 died, and 4 remained on invasive ventilatory support at Week 52.
Trial 3 (Patients with IOPD)
Trial 3 was an open-label, single-center trial in 18 patients with IOPD who had a confirmed diagnosis of Pompe disease as identified through a newborn screening program. All patients were CRIM positive. Patients were treated with intravenous alglucosidase alfa prior to 6 months of age (0.2 to 5.8 months at first infusion). Sixteen patients reached 18 months of age at the time of analysis, and all (100%) were alive without invasive ventilator support.
14.2 Clinical Trials in Late-Onset Pompe Disease
The safety and efficacy of alglucosidase alfa were assessed in 90 patients with late-onset Pompe disease (LOPD), aged 10 to 70 years, in a randomized, double-blind, placebo-controlled trial (Trial 4). All patients were naive to enzyme replacement therapy. Patients were allocated in a 2:1 ratio and received 20 mg/kg of intravenous alglucosidase alfa (n=60) or intravenous placebo (n=30) every other week for 78 weeks (18 months).
The youngest alglucosidase alfa-treated patient was 16 years of age, and the youngest placebo-treated patient was 10 years of age. The trial population included 34 males and 26 females (n=60) in the alglucosidase alfa group and 11 males and 19 females (n=30) in the placebo group. At baseline, all patients were ambulatory (some required assistive walking devices), did not require invasive ventilator support or non-invasive ventilation while awake and sitting upright, and had a forced vital capacity (FVC) between 30 and 79% of predicted in the sitting position. Patients who could not walk 40 meters in 6 minutes or were unable to perform appropriate pulmonary and muscle function testing were excluded from the study.
A total of 81 of 90 patients completed the trial. Of the 9 patients who discontinued, 5 were in the alglucosidase alfa group and 4 were in the placebo group. Three patients discontinued the study due to an adverse event; two patients were in the alglucosidase alfa treatment group and one patient was in placebo group.
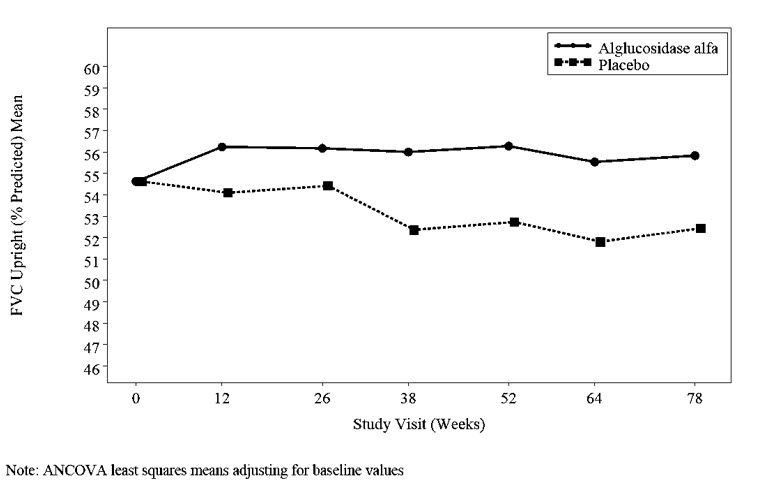
At trial entry, the mean % predicted FVC in the sitting position among all patients was about 55%. After 78 weeks, the mean % predicted FVC increased to 56.2% for alglucosidase alfa-treated patients and decreased to 52.8% for placebo-treated patients indicating an alglucosidase alfa treatment effect of 3.4% (95% confidence interval: [1.3% to 5.5%]; p=0.004). Stabilization of % predicted FVC in the alglucosidase alfa-treated patients was observed (see Figure 1).
Figure 1: Mean FVC Upright (% Predicted) Over Time in Patients with LOPD (Trial 4)
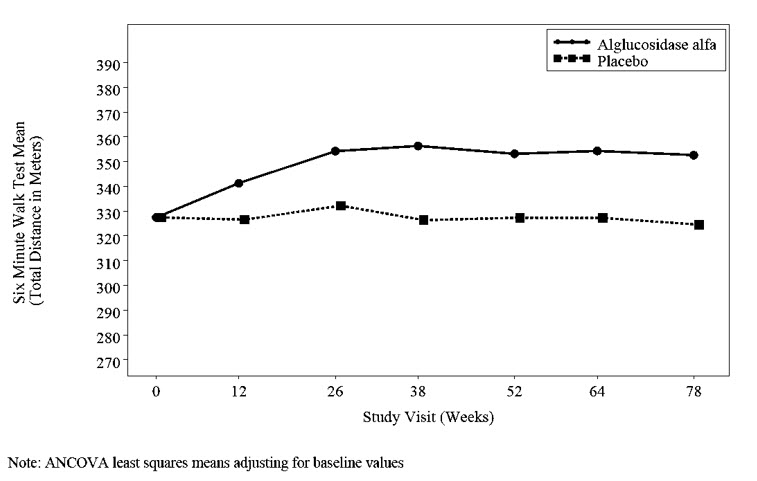
At trial entry, the mean 6 minute walk test (6MWT) among all patients was about 330 meters. After 78 weeks, the mean 6MWT increased by 25 meters for alglucosidase alfa-treated patients and decreased by 3 meters for placebo-treated patients indicating an alglucosidase alfa treatment effect of 28 meters (95% confidence interval: [-1 to 52 meters]; p=0.06) (see Figure 2).
Figure 2: Mean Six Minute Walk Test Total Distance Walked Over Time in Patients with LOPD (Trial 4)
16. How is Lumizyme supplied
LUMIZYME 50 mg vials are supplied as a sterile, nonpyrogenic, preservative-free, white to off-white lyophilized cake or powder in single-dose vials.
NDC 58468-0160-1 (Carton of one single-dose vial)
NDC 58468-0160-2 (Carton of ten single-dose vials)
17. Patient Counseling Information
Hypersensitivity Reactions Including Anaphylaxis, Infusion-Associated Reactions (IARs) and Immune-Mediated Reactions
Advise the patient and caregiver that life-threatening hypersensitivity reactions, including anaphylaxis, IARs, and immune-mediated reactions may occur with LUMIZYME treatment.
Advise the patient and caregiver that anaphylaxis and immune-mediated reactions may occur during the early course of enzyme replacement therapy and after extended duration of therapy.
Inform the patient and caregiver of the symptoms of life-threatening hypersensitivity reactions including anaphylaxis, IARs, and immune-mediated reactions and have them seek immediate medical care should these symptoms occur [see Warning and Precautions (5.1, 5.2, 5.3)].
Risk of Acute Cardiorespiratory Failure
Advise the patient and caregiver that patients with underlying respiratory illness or compromised cardiac or respiratory function may be at risk of acute cardiorespiratory failure. Patients with compromised cardiac or respiratory function may require close observation during and after alglucosidase alfa administration.
Pompe Registry
Inform the patient and their caregiver(s) that the Pompe Registry has been established to better understand the variability and progression of Pompe disease, and to continue to monitor and evaluate long-term effects of LUMIZYME on pregnant women and their offspring [see Use in Specific Populations (8.1)]. Inform the patient and their caregiver(s) that their participation is voluntary and may involve long-term follow-up and to visit www.registrynxt.com or call 1-800-745-4447, extension 15500.
| LUMIZYME
alglucosidase alfa injection, powder, for solution |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Genzyme Corporation (025322157) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genzyme Flanders | 372153895 | ANALYSIS(58468-0160) , MANUFACTURE(58468-0160) , API MANUFACTURE(58468-0160) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genzyme Ireland Limited | 985127419 | ANALYSIS(58468-0160) , MANUFACTURE(58468-0160) , PACK(58468-0160) , LABEL(58468-0160) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Genzyme Corporation | 050424395 | LABEL(58468-0160) , PACK(58468-0160) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Eurofins Biopharma Product Testing Ireland Limited | 238239933 | ANALYSIS(58468-0160) | |
Biological Products Related to Lumizyme
Find detailed information on biosimilars for this medication.
More about Lumizyme (alglucosidase alfa)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- FDA approval history
- Drug class: lysosomal enzymes
- Breastfeeding