HT-Tuss DM Elixir Prescribing Information
Package insert / product label
Generic name: dextromethorphan hydrobromide and guaifenesin
Dosage form: elixir
Drug class: Upper respiratory combinations
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
HT-Tuss DM Elixir Description
Each 5 mL (one teaspoonful) of HT-Tuss DM Elixir contains:
Dextromethorphan hydrobromide . . . 20 mg
Guaifenesin . . . . 200 mg
Alcohol . . . . . . . . . . . 5%
Also contains: Citric acid, FD&C blue #1, FD&C red #3, FD&C red #40, high fructose corn syrup, natural & artificial grape flavor, propylene glycol, purified water, saccharin sodium, sodium benzoate. Sodium citrate may be used to adjust pH.
Dextromethorphan hydrobromide, a synthetic monopioid antitussive, is a salt of the methyl ether of the dextrorotatory isomer of levorphanol, an opioid analgesic. The chemical name is 3- methoxy-17-methyl-9α, 13α, 14α-morphinan hydrobromide monohydrate. It has the following structural formula:
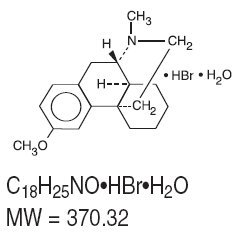
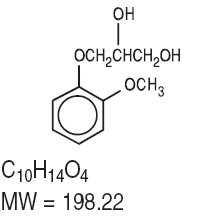
Guaifenesin is an expectorant. The chemical name is (±)-3-(o- Methoxyphenoxy)-1,2- propanediol. It has the following structural formula:
HT-Tuss DM Elixir - Clinical Pharmacology
HT-Tuss DM Elixir combines the cough suppressant dextromethorphan hydrobromide and the expectorant guaifenesin.
Dextromethorphan hydrobromide is a non-opioid antitussive agent. Dextromethorphan temporarily controls and suppresses the cough reflex by a direct action on the cough center. It has no significant analgesic or sedative properties, does not depress respiration or predispose to addiction with usual doses. In therapeutic dosage, dextromethorphan does not inhibit ciliary activity.
Dextromethorphan is rapidly absorbed from the gastrointestinal tract and exerts its effect 15-30 minutes after oral administration. The duration of action is approximately 3-6 hours. Dextromethorphan is rapidly and extensively metabolized by the liver. It is primarily renally excreted as unchanged dextromethorphan and demethylated metabolites including dextrorphan, an active metabolite.
Guaifenesin is an expectorant. Expectorants promote and facilitate the removal of respiratory tract secretions. By increasing respiratory tract fluid, guaifenesin reduces the viscosity of secretions, and facilitates expectoration of retained secretions. Guaifenesin is readily absorbed from the gastrointestinal tract and is rapidly metabolized and renally excreted. Guaifenesin has a plasma half-life of one hour. The major urinary metabolite is beta-(2-methoxyphenoxy) lactic acid.
Indications and Usage for HT-Tuss DM Elixir
HT-Tuss DM Elixir temporarily relieves coughs due to minor throat and bronchial irritation as may occur with upper respiratory tract infections or inhaled irritants, when complicated by viscous mucus. Dextromethorphan hydrobromide suppresses the cough control center and relieves coughing. Guaifenesin helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus, drain bronchial tubes, and make coughs more productive.
Contraindications
HT-Tuss DM Elixir is contraindicated in patients hypersensitive to any of the ingredients and in patients receiving monoamine oxidase inhibitor (MAOI) therapy, including 14 days after stopping MAOI therapy (see PRECAUTIONS: Drug Interactions).
Warnings
A persistent cough may be a sign of a serious condition. If cough persists for more than one week or tends to recur, or is accompanied by fever, rash, or persistent headache, patient reevaluation should be considered.
Precautions
General
Before prescribing any medication to suppress or modify cough, it is important that the underlying cause of cough is identified, that modification of cough does not increase the risk of clinical or physiological complications, and that appropriate therapy for the primary disease is instituted.
Information for patients
Patients should be warned not to use this product If they are now taking a prescription monoamine oxidase inhibitor [MAOI] (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), including 14 days after stopping the MAOI drug. If patients are uncertain whether a prescription drug contains an MAOI, they should be instructed to consult a health professional before taking this product.
Drug Interactions
Serious toxicity (serotonin syndrome) has been reported in patients receiving dextromethorphan hydrobromide and monoamine oxidase inhibitors [MAOIs] concomitantly. Dextromethorphan preparations should not be used in patients receiving MAOIs, including 14 days after stopping the MAOI drug. The use of dextromethorphan hydrobromide may result in additive CNS depressant effects when coadministered with alcohol, antihistamines, psychotropics or other drugs which produce CNS depression.
Laboratory Test Interactions
Guaifenesin interferes with the colorimetric determination of 5-HIAA (5-hydroxyindoleacetic acid) and VMA (vanillylmandelic acid).
Carcinogenesis, mutagenesis, impairment of fertility
No data are available on the long term potential of dextromethorphan hydrobromide or of guaifenesin for carcinogenesis, mutagenesis, or impairment of fertility in animals or humans.
Pregnancy
Teratogenic effects
Pregnancy Category C: Animal reproduction studies have not been conducted with dextromethorphan hydrobromide or guaifenesin. It is also not known whether dextromethorphan hydrobromide or guaifenesin can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. HT-Tuss DM Elixir should be given to a pregnant woman only if clearly needed.
Nursing mothers
It is not known whether dextromethorphan hydrobromide or guaifenesin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when dextromethorphan hydrobromide or guaifenesin are administered to a nursing woman.
Pediatric use
The safety and effectiveness of dextromethorphan hydrobromide and guaifenesin in pediatric patients have been established in the age groups 2 years to over 12 years. Safety and effectiveness in pediatric patients below the age of 2 years have not been established. (see DOSAGE AND ADMINISTRATION).
Administration of dextromethorphan may be associated with histamine release and should be used with caution in atopic children.
Adverse Reactions/Side Effects
Adverse effects associated with dextromethorphan hydrobromide are rare, but nausea and/or other gastrointestinal disturbances, slight dizziness and drowsiness sometimes occur (see also Drug Interactions). Guaifenesin is well tolerated and has a wide margin of safety. Adverse effects associated with guaifenesin usually result from doses larger than those required for expectoration. Reported adverse reactions include nausea and vomiting, dizziness, headache and rash (including urticaria).
To report SUSPECTED ADVERSE REACTIONS, contact Hi-Tech Pharmacal Co., Inc. at 1-800-262- 9010.
Overdosage
Overdosage with dextromethorphan hydrobromide may produce central excitement and mental confusion. Very high doses may produce respiratory depression. Overdosage with guaifenesin is unlikely to produce toxic effects since its toxicity is low. When laboratory animals were administered guaifenesin in doses up to 5 g/kg by stomach tube, no toxicity resulted.
In severe cases of overdosage, the stomach should be emptied (emesis or gastric lavage) and further absorption prevented. Treatment should be directed toward supporting the patient and reversing the effects of the drug.
HT-Tuss DM Elixir Dosage and Administration
Adults and children over 12 years of age:
One teaspoonful (5 mL) every 4 hours, not to exceed 6 teaspoonfuls in 24 hours.
Children 6-12 years:
One half (1.2) teaspoonful (2.5 mL) every 4 hours, not to exceed 3 teaspoonfuls in 24 hours.
Children 2-6 years:
One-fourth (1.4) teaspoonful (1.25 mL) every 4 hours, not to exceed 11.2 teaspoonfuls in 24 hours. It is recommended that caregivers obtain and use a calibrated measuring device for administering this product to a child. Use extreme care in measuring the dosage. Do not exceed the recommended daily dosage except under the advice and supervision of the prescribing physician.
KEEP OUT OF REACH OF CHILDREN.
How is HT-Tuss DM Elixir supplied
HT-Tuss DM Elixir is a purple-colored, fruit-flavored liquid containing 20 mg dextromethorphan hydrobromide and 200 mg guaifenesin per 5 mL with 5% alcohol. It is supplied in containers of 1 pint (473 mL), NDC 50383-135-16.
Store at controlled room temperature, 15°-30°C (59°-86°F).
Rx Only
Manufactured by:
Hi-Tech Pharmacal Co. Inc.
Amityville, NY 11701
MG #13883
Rev. 135:01 3/09
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL

NDC 50383-135-16
HT-TUSS DM ELIXIR
20 mg DEXTROMETHORPHANHYDROBROMIDE
AND
200 mg GUAIFENESIN
per 5 mL
Rx only
Each teaspoonful (5 mL) contains:
Dextromethorphan hydrobromide.....20 mg
Guaifenesin.....200 mg
Alcohol.....5 %
USUAL DOSAGE: Adults and children over 12 years of age: one teaspoonful (5 mL) every 4 hours, not to exceed 6 teaspoonfuls in 24 hours. Children 6-12 years: one-half (½) teaspoonful (2.5 mL) every 4 hours, not to exceed 3 teaspoonfuls in 24 hours. Children 2-6 years: one-fourth (¼) teaspoonful (1.25 mL) every 4 hours, not to exceed 1½ teaspoonfuls in 24 hours.
See package insert for full prescribing information.
PHARMACIST: Dispense in a tight, light-resistant container with a child-resistant closure.
Store at controlled room temperature 15°-30°C (59°-86°F).
WARNING: Keep this and all medication out of reach of children.
| HT-TUSS DM
dextromethorphan hydrobromide and guaifenesin elixir |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Hi-Tech Pharmacal Co., Inc. (101196749) |
More about HT Tuss DM (dextromethorphan / guaifenesin)
- Check interactions
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: upper respiratory combinations
Professional resources
Other brands
Mucinex DM, Tussin DM, Mucinex DM Maximum Strength, Mucinex Children's Cough, Biocotron
