Gantrisin Prescribing Information
Package insert / product label
Generic name: acetyl sulfisoxazole
Dosage form: Pediatric Suspension
Drug class: Sulfonamides
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
Gantrisin Description
Gantrisin (sulfisoxazole) is an antibacterial sulfonamide available as a pediatric suspension for oral administration. Each teaspoonful (5 mL) of the pediatric suspension contains the equivalent of approximately 0.5 gm sulfisoxazole in the form of acetyl sulfisoxazole in a vehicle containing 0.3% alcohol, carboxymethylcellulose (sodium), citric acid, methylcellulose, parabens (methyl and propyl), partial invert sugar, sodium citrate, sorbitan monolaurate, sucrose, flavors and water.
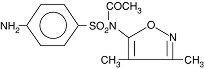
Acetyl sulfisoxazole, the tasteless form of sulfisoxazole, is N1-acetyl sulfisoxazole and must be distinguished from N4-acetyl sulfisoxazole, which is a metabolite of sulfisoxazole. Acetyl sulfisoxazole is a white or slightly yellow, crystalline powder that is slightly soluble in alcohol and practically insoluble in water. Acetyl sulfisoxazole has a molecular weight of 309.34 and the following structural formula:
Gantrisin - Clinical Pharmacology
Following oral administration, sulfisoxazole is rapidly and completely absorbed; the small intestine is the major site of absorption, but some of the drug is absorbed from the stomach. Sulfonamides are present in the blood as free, conjugated (acetylated and possibly other forms) and protein-bound forms. The amount present as "free" drug is considered to be the therapeutically active form. Approximately 85% of a dose of sulfisoxazole is bound to plasma proteins, primarily to albumin; 65% to 72% of the unbound portion is in the nonacetylated form.
Maximum plasma concentrations of intact sulfisoxazole following a single 2-gm oral dose of sulfisoxazole to healthy adult volunteers ranged from 127 to 211 mcg/mL (mean, 169 mcg/mL) and the time of peak plasma concentration ranged from 1 to 4 hours (mean, 2.5 hours). The elimination half-life of sulfisoxazole ranged from 4.6 to 7.8 hours after oral administration. The elimination of sulfisoxazole has been shown to be slower in elderly subjects (63 to 75 years) with diminished renal function (creatinine clearance, 37 to 68 mL/min).1 After multiple-dose oral administration of 500 mg qid to healthy volunteers, the average steady-state plasma concentrations of intact sulfisoxazole ranged from 49.9 to 88.8 mcg/mL (mean, 63.4 mcg/mL).2
Wide variation in blood levels may result following identical doses of a sulfonamide. Blood levels should be measured in patients receiving sulfonamides at the higher recommended doses or being treated for serious infections. Free sulfonamide blood levels of 50 to 150 mcg/mL may be considered therapeutically effective for most infections, with blood levels of 120 to 150 mcg/mL being optimal for serious infections. The maximum sulfonamide level should not exceed 200 mcg/mL, since adverse reactions occur more frequently above this concentration.
N1-acetyl sulfisoxazole is metabolized to sulfisoxazole by digestive enzymes in the gastrointestinal tract and is absorbed as sulfisoxazole. This enzymatic splitting is presumed to be responsible for slower absorption and lower peak blood concentrations than are attained following administration of an equal oral dose of sulfisoxazole. With continued administration of acetyl sulfisoxazole, blood concentrations approximate those of sulfisoxazole. Following a single 4-gm dose of acetyl sulfisoxazole to healthy volunteers, maximum plasma concentrations of sulfisoxazole ranged from 122 to 282 mcg/mL (mean, 181 mcg/mL) for the pediatric suspension and occurred between 2 and 6 hours postadministration. The half-life of elimination from plasma ranged from 5.4 to 7.4.
Sulfisoxazole and its acetylated metabolites are excreted primarily by the kidneys through glomerular filtration. Concentrations of sulfisoxazole are considerably higher in the urine than in the blood. The mean urinary excretion recovery following oral administration of sulfisoxazole is 97% within 48 hours, of which 52% is intact drug, with the remaining as the N4-acetylated metabolite. Following administration of acetyl sulfisoxazole pediatric suspension, approximately 58% is excreted in the urine as total drug within 72 hours.
Sulfisoxazole is distributed only in extracellular body fluid. It is excreted in human milk. It readily crosses the placental barrier and enters into fetal circulation and also crosses the blood-brain barrier. In healthy subjects, cerebrospinal fluid concentrations of sulfisoxazole vary; in patients with meningitis, however, concentrations of free drug in cerebrospinal fluid as high as 94 mcg/mL have been reported.
Microbiology
The sulfonamides are bacteriostatic agents and the spectrum of activity is similar for all. Sulfonamides inhibit bacterial synthesis of dihydrofolic acid by preventing the condensation of the pteridine with aminobenzoic acid through competitive inhibition of the enzyme dihydropteroate synthetase. Resistant strains have altered dihydropteroate synthetase with reduced affinity for sulfonamides or produce increased quantities of aminobenzoic acid.
Susceptibility Tests
Diffusion Techniques
Quantitative methods that require measurement of zone diameters give the most precise estimate of the susceptibility of bacteria to antimicrobial agents. One such standard procedure3 which has been recommended for use with disks to test susceptibility of organisms to sulfisoxazole uses the 250- or 300-mcg sulfisoxazole disk. Interpretation involves the correlation of the diameter obtained in the disk test with the minimum inhibitory concentration (MIC) for sulfisoxazole.
Reports from the laboratory giving results of the standard single-disk susceptibility test with a 250- or 300-mcg sulfisoxazole disk should be interpreted according to the following criteria:
| Zone Diameter (mm) | Interpretation |
|---|---|
| ≥ 17 | Susceptible |
| 13-16 | Moderately susceptible |
| ≤ 12 | Resistant |
A report of "susceptible" indicates that the pathogen is likely to be inhibited by generally achievable blood levels. A report of "moderately susceptible" suggests that the organism would be susceptible if high dosage is used or if the infection is confined to tissues and fluids in which high antimicrobial levels are attained. A report of "resistant" indicates that achievable concentrations are unlikely to be inhibitory, and other therapy should be selected.
Standardized procedures require the use of laboratory control organisms. The 250- or 300-mcg sulfisoxazole disk should give the following zone diameters:
| Organism | Zone Diameter (mm) | ||
|---|---|---|---|
| E. coli | 18-26 mm | ||
| ATCC 25922 | |||
| S. aureus | 24-34 mm | ||
| ATCC 25923 |
Dilution Techniques
Use a standardized dilution method4 (broth, agar, microdilution) or equivalent with sulfisoxazole powder. The MIC values obtained should be interpreted according to the following criteria:
| MIC (mcg/mL) | Interpretation |
|---|---|
| ≤256 | Susceptible |
| ≥512 | Resistant |
As with standard diffusion techniques, dilution methods require the use of laboratory control organisms. Dilutions of standard sulfisoxazole powder should provide the following MIC values:
| Organism | MIC (mcg/mL) | ||
|---|---|---|---|
| S. aureus | 32-128 | ||
| ATCC 29213 | |||
| E. faecalis | 32-128 | ||
| ATCC 29212 | |||
| E. coli | 8-32 | ||
| ATCC 25922 |
Related/similar drugs
amoxicillin, doxycycline, ciprofloxacin, cephalexin, azithromycin, metronidazole, clindamycin
Indications and Usage for Gantrisin
Acute, recurrent or chronic urinary tract infections (primarily pyelonephritis, pyelitis and cystitis) due to susceptible organisms (usually Escherichia coli, Klebsiella-Enterobacter, staphylococcus, Proteus mirabilis and, less frequently, Proteus vulgaris) in the absence of obstructive uropathy or foreign bodies.
Meningococcal meningitis where the organism has been demonstrated to be susceptible. Haemophilus influenzae meningitis as adjunctive therapy with parenteral streptomycin.
Meningococcal meningitis prophylaxis when sulfonamide-sensitive group A strains are known to prevail in family groups or larger closed populations. (The prophylactic usefulness of sulfonamides when group B or C infections are prevalent has not been proven and in closed population groups may be harmful.)
Acute otitis media due to Haemophilus influenzae when used concomitantly with adequate doses of penicillin or erythromycin (see appropriate labeling for prescribing information).
Trachoma. Inclusion conjunctivitis. Nocardiosis. Chancroid. Toxoplasmosis as adjunctive therapy with pyrimethamine. Malaria due to chloroquine-resistant strains of Plasmodium falciparum, when used as adjunctive therapy.
Currently, the increasing frequency of resistant organisms is a limitation of the usefulness of antibacterial agents including the sulfonamides, especially in the treatment of chronic and recurrent urinary tract infections.
Important Note: In vitro sulfonamide susceptibility tests are not always reliable. The test must be carefully coordinated with bacteriologic and clinical response. When the patient is already taking sulfonamides, follow-up cultures should have aminobenzoic acid added to the culture media.
Contraindications
Gantrisin is contraindicated in the following patient populations: patients with a known hypersensitivity to sulfonamides; infants less than 2 months of age (except in the treatment of congenital toxoplasmosis as adjunctive therapy with pyrimethamine); pregnant women at term; and mothers nursing infants less than 2 months of age.
Use in pregnant women at term, in infants less than 2 months of age and in mothers nursing infants less than 2 months of age is contraindicated because sulfonamides may promote kernicterus in the newborn by displacing bilirubin from plasma proteins.
Warnings
FATALITIES ASSOCIATED WITH THE ADMINISTRATION OF SULFONAMIDES, ALTHOUGH RARE, HAVE OCCURRED DUE TO SEVERE REACTIONS, INCLUDING STEVENS-JOHNSON SYNDROME, TOXIC EPIDERMAL NECROLYSIS, FULMINANT HEPATIC NECROSIS, AGRANULOCYTOSIS, APLASTIC ANEMIA AND OTHER BLOOD DYSCRASIAS.
SULFONAMIDES, INCLUDING SULFISOXAZOLE, SHOULD BE DISCONTINUED AT THE FIRST APPEARANCE OF SKIN RASH OR ANY SIGN OF AN ADVERSE REACTION. In rare instances, a skin rash may be followed by more severe reactions such as Stevens-Johnson syndrome, toxic epidermal necrolysis, hepatic necrosis and serious blood disorders (see PRECAUTIONS).
Clinical signs such as rash, sore throat, fever, arthralgia, pallor, purpura or jaundice may be early indications of serious reactions.
Cough, shortness of breath and pulmonary infiltrates are hypersensitivity reactions of the respiratory tract that have been reported in association with sulfonamide treatment.
The sulfonamides should not be used for the treatment of group A beta-hemolytic streptococcal infections. In an established infection, they will not eradicate the streptococcus and, therefore, will not prevent sequelae such as rheumatic fever.
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including sulfisoxazole, and may range in severity from mild to life-threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that toxin produced by Clostridium difficile is one primary cause of "antibiotic-associated colitis."
After the diagnosis of pseudomembranous colitis has been established, therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation, and treatment with an antibacterial drug clinically effective against C. difficile colitis.
Precautions
General
Sulfonamides should be given with caution to patients with impaired renal or hepatic function and to those with severe allergy or bronchial asthma. In glucose-6-phosphate dehydrogenase-deficient individuals, hemolysis may occur; this reaction is frequently dose-related.
The frequency of resistant organisms limits the usefulness of antibacterial agents, including the sulfonamides, as sole therapy in the treatment of urinary tract infections. Since sulfonamides are bacteriostatic and not bactericidal, a complete course of therapy is needed to prevent immediate regrowth and the development of resistant uropathogens.
Information for Patients
Patients should maintain an adequate fluid intake to prevent crystalluria and stone formation.
Laboratory Tests
Complete blood counts should be done frequently in patients receiving sulfonamides. If a significant reduction in the count of any formed blood element is noted, sulfonamide therapy should be discontinued. Urinalyses with careful microscopic examination and renal function tests should be performed during therapy, particularly for those patients with impaired renal function. Blood levels should be measured in patients receiving a sulfonamide for serious infections (see INDICATIONS AND USAGE).
Drug Interactions
It has been reported that sulfisoxazole may prolong the prothrombin time in patients who are receiving anticoagulants, including warfarin. This interaction should be kept in mind when Gantrisin is given to patients already on anticoagulant therapy, and prothrombin time or other suitable coagulation test should be monitored.
It has been proposed that sulfisoxazole competes with thiopental for plasma protein binding. In one study involving 48 patients, intravenous sulfisoxazole resulted in a decrease in the amount of thiopental required for anesthesia and in a shortening of the awakening time. It is not known whether chronic oral doses of sulfisoxazole would have a similar effect. Until more is known about this interaction, physicians should be aware that patients receiving sulfisoxazole might require less thiopental for anesthesia.
Sulfonamides can displace methotrexate from plasma protein-binding sites, thusincreasing free methotrexate concentrations. Studies in man have shown sulfisoxazole infusions to decrease plasma protein-bound methotrexate by one-fourth.
Sulfisoxazole can also potentiate the blood sugar lowering activity of sulfonylureas, as well as cause hypoglycemia by itself.
Carcinogenesis, Mutagenesis and Impairment of Fertility
Carcinogenesis
Sulfisoxazole was not carcinogenic to mice in either sex when administered by gavage for 103 weeks at dosages up to approximately 18 times the highest recommended human daily dose or to rats at 4 times the highest recommended human daily dose. Rats appear to be especially susceptible to the goitrogenic effects of sulfonamides and long-term administration of sulfonamides has resulted in thyroid malignancies in this species.
Mutagenesis
There are no studies available that adequately evaluate the mutagenic potential of Gantrisin. Ames mutagenic assays have not been performed with sulfisoxazole. However, sulfisoxazole was not observed to be mutagenic in E. coli Sd-4-73 when tested in the absence of a metabolic activating system.
Impairment of Fertility
Gantrisin has not undergone adequate trials relating to impairment of fertility. In a reproduction study in rats given 7 times the highest recommended human dose per day of sulfisoxazole, no effects were observed regarding mating behavior, conception rate or fertility index (percent pregnant).
Pregnancy
Teratogenic Effects
Pregnancy Category C. At dosages 7 times the highest recommended human daily dose, sulfisoxazole was not teratogenic in either rats or rabbits. However, in two other teratogenicity studies, cleft palates developed in both rats and mice, and skeletal defects were also observed in rats after administration of 9 times the highest recommended human daily dose of sulfisoxazole.
There are no adequate and well-controlled studies of Gantrisin in pregnant women. It is not known whether Gantrisin can cause fetal harm when administered to a pregnant woman prior to term or can affect reproduction capacity. Gantrisin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
Kernicterus may occur in the newborn as a result of treatment of a pregnant woman at term with sulfonamides (see CONTRAINDICATIONS).
Nursing Mothers
Gantrisin is excreted in human milk. Because of the potential for the development of kernicterus in neonates due to the displacement of bilirubin from plasma proteins by sulfisoxazole, a decision should be made whether to discontinue nursing or discontinue the drug taking into account the importance of the drug to the mother (see CONTRAINDICATIONS).
Pediatric Use
Gantrisin is not recommended for use in infants less than 2 months of age except in the treatment of congenital toxoplasmosis as adjunctive therapy with pyrimethamine (see CONTRAINDICATIONS).
Adverse Reactions/Side Effects
The listing that follows includes adverse reactions both that have been reported with Gantrisin and some which have not been reported with this specific drug; however, the pharmacologic similarities among the sulfonamides require that each of the reactions be considered with the administration of Gantrisin.
Allergic/Dermatologic: Anaphylaxis, erythema multiforme (Stevens-Johnson syndrome), toxic epidermal necrolysis, exfoliative dermatitis, angioedema, arteritis and vasculitis, allergic myocarditis, serum sickness, rash, urticaria, pruritus, photosensitivity, and conjunctival and scleral injection, generalized allergic reactions and generalized skin eruptions. In addition, periarteritis nodosa and systemic lupus erythematosus have been reported (see WARNINGS).
Cardiovascular: Tachycardia, palpitations, syncope, cyanosis.
Endocrine: The sulfonamides bear certain chemical similarities to some goitrogens, diuretics (acetazolamide and thiazides) and oral hypoglycemia agents. Cross-sensitivity may exist with these agents. Development of goiter, diuresis and hypoglycemia have occurred rarely in patients receiving sulfonamides.
Gastrointestinal: Hepatitis, hepatocellular necrosis, jaundice, pseudomembranous colitis, nausea, emesis, anorexia, abdominal pain, diarrhea, gastrointestinal hemorrhage, melena, flatulence, glossitis, stomatitis, salivary gland enlargement, pancreatitis.
Onset of pseudomembranous colitis symptoms may occur during or after treatment with sulfisoxazole (see WARNINGS).
Sulfisoxazole has been reported to cause increased elevations of liver-associated enzymes in patients with hepatitis.
Genitourinary: Crystalluria, hematuria, BUN and creatinine elevations, nephritis and toxic nephrosis with oliguria and anuria. Acute renal failure and urinary retention have also been reported. The frequency of renal complications, commonly associated with some sulfonamides, is lower in patients receiving the more soluble sulfonamides such as sulfisoxazole.
Hematologic: Leukopenia, agranulocytosis, aplastic anemia, thrombocytopenia, purpura, hemolyticanemia, anemia, eosinophilia, clotting disorders including hypoprothrombinemia, and hypofibrinogenemia, sulfhemoglobinemia, methemoglobinemia.
Musculoskeletal: Arthralgia, myalgia.
Neurologic: Headache, dizziness, peripheral neuritis, paresthesia, convulsions, tinnitus, vertigo, ataxia, intracranial hypertension.
Psychiatric: Psychosis, hallucination, disorientation, depression, anxiety, apathy.
Respiratory: Cough, shortness of breath, pulmonary infiltrates (see WARNINGS).
Vascular: Angioedema, arteritis, vasculitis.
Miscellaneous: Edema (including periorbital), pyrexia, drowsiness, weakness, fatigue, lassitude, rigors, flushing, hearing loss, insomnia, pneumonitis, chills.
Overdosage
The amount of a single dose of sulfisoxazole that is associated with symptoms of overdosage or is likely to be life-threatening has not been reported. Signs and symptoms of overdosage reported with sulfonamides include anorexia, colic, nausea, vomiting, dizziness, headache, drowsiness and unconsciousness. Pyrexia, hematuria and crystalluria may be noted. Blood dyscrasias and jaundice are potential late manifestations of overdosage.
General principles of treatment include the immediate discontinuation of the drug; institution of gastric lavage or emesis; forcing oral fluids; and the administration of intravenous fluids if urine output is low and renal function is normal. The patient should be monitored with blood counts and appropriate blood chemistries, including electrolytes. If the patient becomes cyanotic, the possibility of methemoglobinemia should be considered and, if present, the condition should be treated appropriately with intravenous 1% methylene blue. If a significant blood dyscrasia or jaundice occurs, specific therapy should be instituted for these complications.
Peritoneal dialysis is not effective and hemodialysis is only moderately effective in eliminating sulfonamides.
Gantrisin Dosage and Administration
Systemic sulfonamides are contraindicated in infants less than 2 months of age, except in the treatment of congenital toxoplasmosis as adjunctive therapy with pyrimethamine.
How is Gantrisin supplied
Pediatric Suspension (raspberry flavored), containing acetyl sulfisoxazole equivalent to approximately 0.5 gm sulfisoxazole per teaspoonful (5 mL) — bottles of 16 oz (1 pint) (NDC 0004-1003-28).
References
- Boisvert A, Barbeau G, Belanger PM. Pharmacokinetics of sulfisoxazole in young and elderly subjects. Gerontology. 1984; 30:125-131.
- Oie S, Gambertoglio JG, Fleckenstein L. Comparison of the disposition of total and unbound sulfisoxazole after single and multiple dosing. J Pharmacokinet Biopharm. 1982; 10:157-172.
- National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Disk Susceptibility Tests. 4th ed. Villanova, PA: April 1990. Approved Standard NCCLS Document M2-A4, Vol. 10, No. 7 NCCLS.
- National Committee for Clinical Laboratory Standards. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically. 2nd ed. Villanova, PA: April 1990. Approved Standard NCCLS Document M7-A2, Vol. 10, No. 8 NCCLS.
| GANTRISIN
acetyl sulfisoxazole suspension |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Roche Laboratories Inc. |
More about Gantrisin (sulfisoxazole)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: sulfonamides
- Breastfeeding
Related treatment guides
Copyright © 1997 by Roche Laboratories Inc. All rights reserved.


