DaTscan: Package Insert / Prescribing Info
Package insert / product label
Generic name: ioflupane i-123 and iodine
Dosage form: injection, solution
Drug class: Diagnostic radiopharmaceuticals
Medically reviewed by Drugs.com. Last updated on Jan 14, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
DATSCAN (ioflupane I 123 injection), for intravenous use
Initial U.S. Approval: 2011
Recent Major Changes
Indications and Usage for DaTscan
DATSCAN is a radioactive diagnostic agent indicated as an adjunct to other diagnostic evaluations for striatal dopamine transporter visualization using single photon emission computed tomography (SPECT) brain imaging in adult patients with:
- suspected Parkinsonian syndromes (PS) or
- suspected dementia with Lewy bodies (DLB). (1)
DaTscan Dosage and Administration
- Administer a thyroid-blocking agent at least one hour before the dose of DATSCAN. (2.2)
- The recommended dose of DATSCAN in adult patients is 111 MBq to 185 MBq (3 mCi to 5 mCi) administered intravenously over at least 20 seconds. (2.3)
- Measure patient dose using a dose calibrator immediately prior to administration. (2.4)
- Begin SPECT imaging between 3 hours and 6 hours post-injection. (2.6)
Dosage Forms and Strengths
Injection: 185 MBq (5 mCi) in 2.5 mL at a concentration of 74 MBq/mL (2 mCi/mL) of ioflupane I 123 at calibration date and time supplied in single-dose vials. (3)
Contraindications
Known serious hypersensitivity to ioflupane I 123 (4)
Warnings and Precautions
- Hypersensitivity Reactions: Hypersensitivity reactions including dyspnea, edema, rash, erythema, and pruritus have been reported. Have treatment measures available prior to DATSCAN administration. (5.1)
- Thyroid Accumulation of Iodine-123: Thyroid uptake of iodine-123 may result in an increased long-term risk for thyroid neoplasia. (2.2, 5.2)
- Radiation Risk: Ensure safe handling to minimize radiation exposure to the patient and healthcare providers. Advise patients to hydrate before and after administration and to void frequently after administration. (2.1, 5.3)
Adverse Reactions/Side Effects
The following adverse reactions were reported at ≤1%: headache, nausea, vertigo, dry mouth and dizziness. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact GE Healthcare at 1-800-654-0118 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Amoxapine, amphetamine, armodafinil, benztropine, bupropion, buspirone, cocaine, mazindol, methamphetamine, methylphenidate, modafinil norephedrine, phentermine, phenylpropanolamine, selegiline, sertraline, citalopram and paroxetine may interfere with DATSCAN imaging. The effects of dopamine agonists and antagonists on DATSCAN imaging have not been established. (7)
Use In Specific Populations
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 11/2022
Full Prescribing Information
1. Indications and Usage for DaTscan
DATSCAN is indicated as an adjunct to other diagnostic evaluations for striatal dopamine transporter visualization using single photon emission computed tomography (SPECT) brain imaging in adult patients with:
- suspected Parkinsonian syndromes (PS) or
- suspected dementia with Lewy bodies (DLB).
2. DaTscan Dosage and Administration
2.1 Radiation Safety
DATSCAN emits radiation and must be handled with safety measures to minimize radiation exposure to healthcare providers and patients [see Warnings and Precautions (5.3)]. Radiopharmaceuticals should be used by or under the control of healthcare providers who are qualified by specific training and experienced in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate government agency authorized to license the use of radionuclides.
To minimize radiation exposure, encourage hydration prior to and following DATSCAN administration in order to permit frequent voiding. Encourage the patient to void frequently for the first 48 hours following DATSCAN administration [see Dosage and Administration (2.5)].
2.2 Thyroid Blockade Before DATSCAN Administration
Administer Potassium Iodide Oral Solution or Strong Iodine Solution (e.g., Lugol's Solution) equivalent to 100 mg iodide to block uptake of iodine-123 by the patient's thyroid at least one hour before the dose of DATSCAN [see Warnings and Precautions (5.2)].
2.3 Recommended Dosage
The recommended dose of DATSCAN in adult patients is 111 MBq to 185 MBq (3 mCi to 5 mCi) administered intravenously over at least 20 seconds.
2.4 Preparation and Administration
- Assess pregnancy status before administering DATSCAN to a female of reproductive potential [see Use in Specific Populations (8.1)].
- Use aseptic procedures and radiation shielding during preparation and administration.
- Inspect the DATSCAN vial for particulate matter and discoloration prior to administration and do not use it if the vial contains particulate matter or discoloration.
- Measure patient dose using a dose calibrator immediately prior to administration.
- Discard unused portion.
2.5 Radiation Dosimetry
The estimated radiation absorbed doses to an adult from intravenous injection of DATSCAN are shown in Table 1. The values are calculated assuming urinary bladder emptying at 4.8-hour intervals and appropriate thyroid blocking (iodine-123 is a known Auger electron emitter).
| ORGAN / TISSUE | ABSORBED DOSE PER UNIT ADMINISTERED ACTIVITY (µGy / MBq) |
|
|---|---|---|
|
||
| Adrenals | 12.9 | |
| Brain | 17.8 | |
| Striata | 230 | |
| Breasts | 7.8 | |
| Gallbladder Wall | 26.4 | |
| GI Tract | Esophagus | 10 |
| Stomach Wall | 11.2 | |
| Small Intestine Wall | 21.2 | |
| Colon Wall * | 39.8 | |
| Upper Large Intestine Wall | 38.1 | |
| Lower Large Intestine Wall | 42 | |
| Heart Wall | 12.9 | |
| Kidneys | 10.9 | |
| Liver | 27.9 | |
| Lungs | 41.2 | |
| Muscle | 9.4 | |
| Osteogenic Cells | 28.2 | |
| Ovaries | 16.8 | |
| Pancreas | 13 | |
| Red Marrow | 9.2 | |
| Skin | 6 | |
| Spleen | 10.4 | |
| Testes | 8.5 | |
| Thymus | 10 | |
| Thyroid | 9 | |
| Urinary Bladder Wall | 53.1 | |
| Uterus | 16.1 | |
| Total Body | 11.3 | |
| EFFECTIVE DOSE PER UNIT ADMINISTERED ACTIVITY (µSv/MBq) | 21.3 | |
The Effective Dose resulting from a DATSCAN administration with an administered activity of 185 MBq (5 mCi) is 3.94 mSv in an adult.
2.6 Imaging Instructions
Begin SPECT imaging 3 hours to 6 hours following DATSCAN administration.
Use a gamma camera fitted with high-resolution collimators and set to a photopeak of 159 keV with a ± 10% energy window. Angular sampling should be at least 120 views over 360 degrees.
Position the patient supine with the head on an off-the-table headrest, use a flexible head restraint such as a strip of tape across the chin or forehead if necessary to help avoid movement, and set a circular orbit for the detector heads with the radius as small as possible (typically 11 cm to 15 cm).
Obtain images with matrix size and zoom factors selected to give a pixel size of 3.5 mm to 4.5 mm. Collect a minimum of 1.5 million counts for optimal images.
2.7 Image Interpretation
Interpret DATSCAN images visually, based upon the appearance of the striata. Reconstructed pixel size should be between 3.5 mm and 4.5 mm with slices 1 pixel thick. Optimum presentation of the reconstructed images for visual interpretation is transaxial slices parallel to the anterior commissure-posterior commissure (AC-PC) line.
Determine whether an image is normal or abnormal by assessing the extent (as indicated by shape) and intensity of the striatal signal. Image interpretation does not involve integration of the striatal image appearance with clinical signs and/or symptoms.
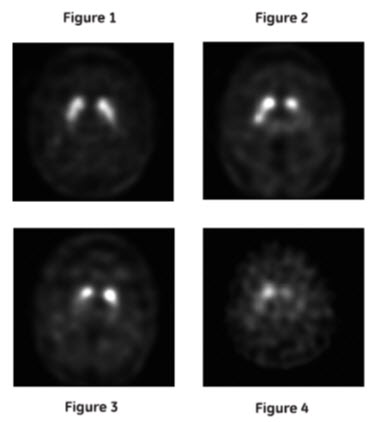
Normal: In transaxial images, normal images are characterized by two symmetric comma- or crescent-shaped focal regions of activity mirrored about the median plane. Striatal activity is distinct, relative to surrounding brain tissue (Figure 1).
Abnormal: Abnormal DATSCAN images fall into at least one of the following three categories (all are considered abnormal).
- Activity is asymmetric, e.g., activity in the region of the putamen of one hemisphere is absent or greatly reduced with respect to the other. Activity is still visible in the caudate nuclei of both hemispheres resulting in a comma or crescent shape in one and a circular or oval focus in the other. There may be reduced activity between at least one striatum and surrounding tissues (Figure 2).
- Activity is absent in the putamen of both hemispheres and confined to the caudate nuclei. Activity is relatively symmetric and forms two roughly circular or oval foci. Activity of one or both is generally reduced (Figure 3).
- Activity is absent in the putamen of both hemispheres and greatly reduced in one or both caudate nuclei. Activity of the striata with respect to the background is reduced (Figure 4).
3. Dosage Forms and Strengths
Injection: clear, colorless solution containing 185 MBq (5 mCi) in 2.5 mL at a concentration of 74 MBq/mL (2 mCi/mL) of ioflupane I 123 at calibration date and time supplied in single-dose vials.
4. Contraindications
DATSCAN is contraindicated in patients with known serious hypersensitivity to ioflupane I 123 [see Warnings and Precautions (5.1)].
5. Warnings and Precautions
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including dyspnea, edema, rash, erythema, and pruritus, have been reported following DATSCAN administration [see Adverse Reactions (6.2)]. The reactions have generally occurred within minutes of DATSCAN administration and have either resolved spontaneously or following the administration of corticosteroids and antihistamines.
DATSCAN is contraindicated in patients with known serious hypersensitivity to ioflupane I 123 [see Contraindications (4)]. Have treatment measures available and monitor patients for symptoms or signs of a hypersensitivity reaction.
5.2 Thyroid Accumulation of Iodine-123
DATSCAN may contain up to 6% of free iodide (iodine-123). Thyroid uptake of iodine-123 may result in an increased long-term risk for thyroid neoplasia. To decrease thyroid accumulation of iodine-123, block the thyroid gland before administration of DATSCAN [see Dosage and Administration (2.2)].
5.3 Radiation Risk
DATSCAN contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Ensure safe handling to minimize radiation exposure to patients and healthcare providers. Advise patients to hydrate before and after administration and to void frequently after administration [see Dosage and Administration (2.1)].
6. Adverse Reactions/Side Effects
The following clinically significant adverse reaction is described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of DATSCAN cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data from clinical trials reflect exposure to DATSCAN in 942 subjects with a mean age of 66 years (range 25 years to 90 years). Among these subjects, 42% were female and 99% White. Subjects received 88 MBq to 287 MBq (2 mCi to 8 mCi) [median 173 MBq (4.7 mCi)] intravenously as a single dose. The recommended dose of DATSCAN is 111 MBq to 185 MBq (3 mCi to 5 mCi) [see Dosage and Administration (2.3)].
The following adverse reactions were reported at a rate of 1% or less: headache, nausea, vertigo, dry mouth, and dizziness.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of DATSCAN. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: Hypersensitivity, including dyspnea, edema, rash, erythema, and pruritus
General disorders and administration site conditions: Injection site pain
Related/similar drugs
7. Drug Interactions
Ioflupane I 123 binds to the dopamine transporter [see Clinical Pharmacology (12.1)]. Drugs that bind to the dopamine transporter with high affinity may interfere with the image obtained following DATSCAN administration. These potentially interfering drugs consist of: amoxapine, amphetamine, armodafinil, benztropine, bupropion, buspirone, cocaine, mazindol, methamphetamine, methylphenidate, modafinil, norephedrine, phentermine, phenylpropanolamine, selegiline, and sertraline. Selective serotonin reuptake inhibitors (paroxetine and citalopram) may increase or decrease ioflupane binding to the dopamine transporter. Whether discontinuation of these drugs prior to DATSCAN administration may minimize the interference with a DATSCAN image is unknown.
The impact of dopamine agonists and antagonists upon DATSCAN imaging results has not been established.
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Radioactive iodine products cross the placenta and can permanently impair fetal thyroid function. Administration of an appropriate thyroid blocking agent is recommended before use of DATSCAN in a pregnant woman to protect the woman and fetus from accumulation of iodine-123 [see Dosage and Administration (2.2)].
There are no available data on DATSCAN use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Animal reproduction studies have not been conducted with ioflupane I 123. All radiopharmaceuticals have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose. Administration of DATSCAN at a dose of 185 MBq (5 mCi) results in an absorbed radiation dose to the uterus of 0.3 rad (3.0 mGy). Radiation doses greater than 15 rad (150 mGy) have been associated with congenital anomalies but doses under 5 rad (50 mGy) generally have not. Advise pregnant women of the potential risks of fetal exposure to radiation doses with administration of DATSCAN.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
Iodine-123, the radionuclide in DATSCAN, is present in human milk. There is no information on the effects on the breastfed infant or on milk production. Advise a lactating woman to interrupt breastfeeding and pump and discard breastmilk for at least 6 days (>10 physical half-lives) after DATSCAN administration in order to minimize radiation exposure to a breastfed infant.
8.4 Pediatric Use
The safety and efficacy of DATSCAN have not been established in pediatric patients.
8.5 Geriatric Use
In the two principal clinical studies of DATSCAN for suspected Parkinsonian syndromes, 45% of the subjects were aged 65 and over, while 10% were 75 and over [see Clinical Studies (14.1)]. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
10. Overdosage
The risks of overdose relate predominantly to increased radiation exposure, with the long-term risks for neoplasia. In case of overdosage of radioactivity, frequent urination and defecation should be encouraged to minimize radiation exposure to the patient.
11. DaTscan Description
11.1 Chemical Characteristics
DATSCAN (ioflupane I 123 injection) is a sterile, pyrogen-free radioactive diagnostic agent for intravenous use. DATSCAN is a clear and colorless solution supplied in single-dose vials. Each milliliter contains 0.07 to 0.13 mcg ioflupane, 74 MBq (2 mCi) of iodine-123 (as ioflupane I 123) at calibration time, 5.8 mg acetic acid, 8.2 mg sodium acetate and 0.05 mL (5%) ethanol. The pH of the solution is between 4.2 and 5.2.
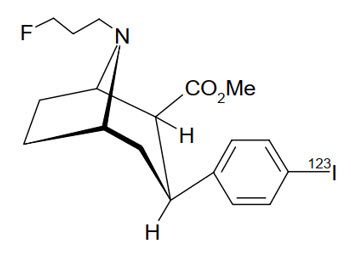
The chemical name for ioflupane I 123 is N-ω-fluoropropyl-2β-carbomethoxy-3β-(4-[123I]iodophenyl)nortropane and it has the following structural formula:
11.2 Physical Characteristics
Iodine-123 is a cyclotron-produced radionuclide that decays to tellurium-123 by electron capture and has a physical half-life of 13.2 hours. The photon that is useful for detection and imaging studies is listed in Table 2.
| Radiation | Energy Level (keV) | Abundance (%) |
|---|---|---|
| Gamma | 159 | 83 |
11.3 External Radiation
The specific gamma-ray constant for iodine-123 is 1.6 R/mCi-hr at 1 cm. The first half-value thickness of lead (Pb) for iodine-123 is 0.04 cm. The relative transmission of radiation emitted by the radionuclide that results from interposition of various thicknesses of Pb is shown in Table 3 (e.g., the use of 2.16 cm Pb will decrease the external radiation exposure by a factor of about 1,000).
| Shield Thickness cm of lead (Pb) | Reduction in In-air Collision Kerma |
|---|---|
|
|
| 0.04 | 0.5 |
| 0.13 | 10-1 |
| 0.77 | 10-2 |
| 2.16 | 10-3 |
| 3.67 | 10-4 |
12. DaTscan - Clinical Pharmacology
12.1 Mechanism of Action
The active drug substance in DATSCAN is N-ω-fluoropropyl-2β-carbomethoxy-3β-(4-[123I]iodophenyl)nortropane or ioflupane I 123.
In vitro, ioflupane binds reversibly to the human dopamine transporter (DaT) (Ki = 0.62 nM; IC50 = 0.71 nM). Autoradiography of post-mortem human brain slices exposed to radiolabeled ioflupane shows concentration of the radiolabel in striatum (caudate nucleus and putamen). The specificity of the binding of ioflupane I 125 to dopamine transporter was demonstrated by competition studies with the DaT inhibitor GBR 12909 (a dopamine reuptake inhibitor), the serotonin reuptake inhibitor citalopram, and the norepinephrine reuptake inhibitor desipramine in post-mortem human brain slices exposed to radiolabeled ioflupane. Citalopram reduced binding in the neocortex and thalamus with only minor effects in the striatum. This indicated that the binding in the cortex and thalamus is mainly to the serotonin reuptake sites. Desipramine showed no effect on the level of striatal binding of ioflupane I 125, but reduced extrastriatal binding by 60 to 85%. The binding of ioflupane I 125 to the striatum was abolished in the presence of high concentrations of GBR 12909, indicating selectivity of ioflupane binding for the pre-synaptic DaT.
Following administration of DATSCAN to humans, radioactive decay of the iodine-123 emits gamma radiation which can be detected externally using gamma detectors, allowing visualization of the brain striata through SPECT imaging [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
As DATSCAN contains a very small quantity of ioflupane, no ioflupane pharmacologic effects are expected [see Description (11)].
12.3 Pharmacokinetics
The pharmacokinetics of ioflupane I 123 were studied by monitoring radioactivity following intravenous injection; only 5% of the administered radioactivity remained in whole blood at 5 minutes post-injection. Uptake in the brain reached approximately 7% of injected radioactivity at 10 minutes post-injection and decreased to 3% after 5 hours; striata to background ratios were relatively constant between 3 hours and 6 hours post-injection. About 30% of the whole brain radioactivity was attributed to striatal uptake. By 48 hours post-injection, approximately 60% of the injected radioactivity has been excreted in the urine, with fecal excretion estimated to be approximately 14%.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies on reproductive toxicity have not been conducted. Ioflupane showed no evidence of mutagenic potential in in vitro or in vivo mutagenicity studies. Studies to assess the carcinogenic potential of ioflupane have not been performed.
13.2 Animal Toxicology and/or Pharmacology
Single- and repeated-dose intravenous toxicity studies have been performed using ioflupane in rats, rabbits, and dogs. Additionally, single-dose acute toxicity studies have been performed in cynomolgus monkeys. No mortality or other toxicity was observed at doses up to 5,500 times the maximum clinical dose of DATSCAN; at doses greater than 1,500 times the maximum clinical dose, pharmacological responses such as mydriasis and hyperactivity were seen in some species.
14. Clinical Studies
14.1 Suspected Parkinsonian Syndromes
The safety and efficacy of DATSCAN were evaluated in two multicenter, single-arm, open-label studies (Study 1 and Study 2) that enrolled 284 adult patients with suspected PS.
In these studies, DATSCAN image interpretation was compared to a reference clinical diagnostic standard of "PS" or "non-PS". PS consisted of Parkinson's disease (PD), multiple system atrophy (MSA), and progressive supranuclear palsy (PSP). These conditions have been associated with dopaminergic neurodegeneration and DATSCAN imaging was not designed to distinguish among the conditions. Among the non-PS population, subjects with a history of the following conditions were included: vascular parkinsonism, psychogenic parkinsonism, essential tremor (ET), various forms of tremor, and other non-PS diagnoses; DATSCAN imaging was not designed to distinguish among these conditions.
Patients underwent SPECT imaging with a variety of multi-headed cameras or multi-detector single-slice systems 3 to 6 hours after DATSCAN administration. The DATSCAN activity in these two clinical studies ranged from 111 MBq to 201 MBq (3 mCi to 5.4 mCi) [median 167.8 MBq (4.5 mCi)]. Readers blinded to clinical information interpreted DATSCAN imaging as normal or abnormal [see Dosage and Administration (2.7)]. A normal image is inconsistent with clinical conditions associated with PS and an abnormal image is consistent with clinical conditions associated with PS.
Study 1 consisted of 99 patients with early features of Parkinsonism; patients with features suggestive of MSA or PSP were excluded. The baseline clinical diagnoses consisted of: probable PD (44%), possible PD (31%), "benign" PD (6%), possible ET (11%), and other diagnoses (7%). The patients were 44% female, 42% of age 65 years or over, and all were White. Study 1 readers had no other role in patient assessment. The reference clinical diagnostic standard consisted of the clinical diagnoses established by a consensus panel of movement disorder specialists that evaluated data inclusive through 36 months of follow-up.
Study 2 consisted of 185 patients with clinically established diagnosis of PS (PD, MSA, PSP) or ET. The baseline clinical diagnoses consisted of: PD (70%), MSA (10%), PSP (5%), and ET (15%). The patients were 35% female, 48% of age 65 years or over, and 99% were White. Study 2 readers included site investigators. The reference clinical diagnostic standard consisted of the investigator-determined baseline clinical diagnosis.
Table 4 shows the positive percent agreement and negative percent agreement of the DATSCAN image results with the reference clinical diagnostic standard. Positive percent agreement represents the percent of patients with abnormal DATSCAN images among all the patients with a clinical diagnostic reference standard of PS. Negative percent agreement represents the percent of patients with normal DATSCAN images among the patients with a non-PS clinical diagnostic reference standard.
| Positive percent agreement (95 % CI) | Negative percent agreement (95 % CI) |
|
|---|---|---|
| Study 1 (patients with early signs and/or symptoms of PS) | ||
| Reader A, n = 99 | 77 (66, 87) | 96 (82, 100) |
| Reader B, n = 96 | 78 (66, 87) | 96 (82, 100) |
| Reader C, n = 98 | 79 (67, 87) | 96 (82, 100) |
| Study 2 (patients with established diagnoses of PS or ET) | ||
| Reader A, n = 185 | 93 (88, 97) | 96 (81, 100) |
| Reader B, n = 185 | 97 (93, 99) | 74 (54, 89) |
| Reader C, n = 185 | 96 (92, 99) | 85 (66, 96) |
| Reader D, n = 185 | 92 (87, 96) | 93 (76, 99) |
| Reader E, n = 185 | 94 (90, 97) | 93 (76, 99) |
14.2 Suspected Dementia with Lewy Bodies
The evidence for the efficacy of DATSCAN in visualizing striatal dopamine transporters in adult patients with DLB is derived from the scientific literature. In 55 patients clinically diagnosed with DLB or with Alzheimer's disease, the results of consensus blinded visual assessment of the DATSCAN images were compared to neuropathological diagnosis made using standard international criteria at autopsy. The patients were 38% female and of ages 63 to 95 years (mean 77 years) at the time of DATSCAN imaging. The median administered DATSCAN activity was 150 MBq to 185 MBq (4.1 mCi to 4.9 mCi). DATSCAN images were rated as normal or abnormal [see Dosage and Administration (2.7)]. Based on autopsy diagnosis, the sensitivity and specificity of DATSCAN for DLB was 80% (95% CI 61% to 92%) and 92% (95% CI 74% to 99%), respectively.
The effectiveness of DATSCAN as a screening or confirmatory test and for monitoring disease progression or response to therapy has not been established.
16. How is DaTscan supplied
How Supplied
DATSCAN (ioflupane I 123 injection) is a clear, colorless solution supplied in single-dose glass vials containing 185 MBq (5 mCi) in 2.5 mL at a concentration of 74 MBq/mL (2 mCi/mL) of ioflupane I 123 at calibration date and time. Each vial is enclosed in a lead container of appropriate thickness.
NDC 17156-210-01
Storage and Handling
Store DATSCAN at 20° to 25°C (68° to 77°F). This product does not contain a preservative. Store DATSCAN within the original lead container or equivalent radiation shielding.
Do not use DATSCAN preparations after the expiration date and time stated on the label.
This preparation is approved for use by persons licensed by the Illinois Emergency Management Agency pursuant to 32 IL. Adm. Code Section 330.260(a) and 335.4010 or equivalent licenses of the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
17. Patient Counseling Information
Hypersensitivity Reactions
Inform the patient that hypersensitivity reactions, including dyspnea, edema, rash, erythema and pruritus, have been reported following DATSCAN administration [see Warnings and Precautions (5.1)].
Adequate Hydration
Instruct patients to drink a sufficient amount of water prior to and after receiving DATSCAN and to void frequently for the first 48 hours following DATSCAN administration [see Dosage and Administration (2.1)].
Lactation
Advise a lactating woman to interrupt breastfeeding and pump and discard breastmilk for at least 6 days (>10 physical half-lives) after DATSCAN administration in order to minimize radiation exposure to a breastfed infant [see Use in Specific Populations (8.2)].
Pregnancy
Advise a pregnant woman of the potential risks of fetal exposure to radiation doses with DATSCAN [see Use in Specific Populations (8.1)].
Manufactured and Distributed by
GE Healthcare, Medi-Physics, Inc.
Arlington Heights, IL 60004 U.S.A.
GE Healthcare
DATSCAN is a trademark of GE Healthcare or one of its subsidiaries.
GE and the GE Monogram are trademarks of General Electric Company.
© 2022 General Electric Company – All rights reserved.
43-2010F
PRINCIPAL DISPLAY PANEL - 2.5 mL Vial Label
GE Healthcare
NDC 17156-210-01
DaTscan™
Ioflupane I 123 Injection
Ioflupane I 123 185 MBq (5 mCi) in 2.5 mL
at calibration
Radiopharmaceutical for Intravenous Injection
Single-Dose Vial. Discard unused portion.
Rx ONLY
VOLUME: 2.5 mL
RADIOACTIVE
CONCENTRATION:
74 MBq/mL
(2 mCi/mL)
CALIB.
DATE:
DDMMMYY
CALIB.
TIME:
1200 XXX

| DATSCAN
ioflupane i-123 injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Medi-Physics Inc. dba GE Healthcare. (095263729) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Medi-Physics Inc. dba GE Healthcare. | 095263729 | MANUFACTURE(17156-210) | |
More about DaTscan (ioflupane I 123)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Side effects
- Dosage information
- During pregnancy
- Generic availability
- FDA approval history
- Drug class: diagnostic radiopharmaceuticals
- Breastfeeding
- En español
