Altoprev Prescribing Information
Package insert / product label
Generic name: lovastatin
Dosage form: tablet, extended release
Drug class: Statins
Medically reviewed by Drugs.com. Last updated on Apr 15, 2024.
On This Page
- Indications and Usage
- Dosage and Administration
- Dosage Forms and Strengths
- Contraindications
- Warnings and Precautions
- Adverse Reactions/Side Effects
- Drug Interactions
- Use In Specific Populations
- Overdosage
- Description
- Clinical Pharmacology
- Nonclinical Toxicology
- Clinical Studies
- How Supplied/Storage and Handling
- Patient Counseling Information
Highlights of Prescribing Information
ALTOPREV® (lovastatin extended-release) Tablets for oral administration
Initial U.S. Approval: 2002
Recent Major Changes
Contraindications, Pregnancy and Lactation (4) Removed 03/2024
Indications and Usage for Altoprev
Altoprev is an HMG-CoA reductase inhibitor (statin) indicated:
- •
- To reduce the risk of myocardinal infarction (MI), unstable angina, and coronary revascularization procedures in adults at high risk for coronary heart disease (CHD). (1)
- •
- As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) and slow the progression of coronary atherosclerosis in adults with CHD. (1)
- •
- As an adjunct to diet to reduce LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH). (1)
Altoprev Dosage and Administration
- •
- Recommended dosage range: 20 to 60 mg orally once daily in the evening. (2.2)
- •
- Patients with severe renal impairment: consider the risks and benefits of increasing the dosage above 20 mg daily (2.3)
- •
- See full prescribing information for Altoprev dosage modifications due to drug interactions. (2.4)
Dosage Forms and Strengths
Extended-release tablets: 20 mg, 40 mg , and 60 mg of lovastatin (3)
Contraindications
Warnings and Precautions
- •
- Myopathy and Rhabdomyolysis: Cases of myopathy and rhabdomyolysis with acute renal failure secondary to myoglobinuria have been reported with HMG-CoA reductase inhibitors, including Altoprev. Predisposing factors include advanced age (≥65), female gender, uncontrolled hypothyroidism, and renal impairment. Advise patients to report promptly to their physician any unexplained and/or persistent muscle pain, tenderness, or weakness. Altoprev therapy should be discontinued immediately if myopathy is diagnosed or suspected. (5.1)
- •
- Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of IMNM, an autoimmune myopathy, have been reported. Discontinue Altoprev if IMNM is suspected. (5.2)
- •
- Hepatic Dysfunction: Increases in serum transaminases have occurred, some persistent. Rare reports of fatal and non-fatal hepatic failure have occurred. Consider testing liver enzyme before initiating therapy and as clinically indicated thereafter. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue Altoprev. (5.3)
Adverse Reactions/Side Effects
The most commonly reported adverse reactions (incidence ≥ 5%) in patients treated with Altoprev® in placebo-controlled trials were: infection, headache and accidental injury. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Covis Pharma at 1-877-411-2510 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
See full prescribing information for details regarding concomitant use of Altoprev with other drugs that increase the risk of myopathy and rhabdomyolysis. (7)
|
Drug Interactions Associated with Increased |
|
|
Interacting Agents |
Prescribing Recommendations |
|
Strong CYP3A inhibitors |
Contraindicated with Altoprev |
|
Gemfibrozil, cyclosporine |
Altoprev is not recommended |
|
Danazol, diltiazem, dronedarone, verapamil |
Do not exceed 20 mg Altoprev daily |
|
Amiodarone |
Do not exceed 40 mg Altoprev daily |
|
Grapefruit juice |
Grapefruit juice is not recommended |
Use In Specific Populations
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2024
Full Prescribing Information
1. Indications and Usage for Altoprev
Altoprev is indicated:
- •
- To reduce the risk of myocardial infarction, unstable angina, and coronary revascularization procedures in adults at high risk for coronary heart disease.
- •
- As an adjunct to diet to reduce low-density lipoprotein cholesterol (LDL-C) and slow the progression of coronary atherosclerosis in adults with coronary heart disease.
- •
- As an adjunct to diet to reduce LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH).
2. Altoprev Dosage and Administration
2.1 Important Dosage and Administration Information
- •
- Take Altoprev orally once daily in the evening.
- •
- The maximum recommended dosage of Altoprev is 60 mg orally once daily.
- •
- Swallow tablets whole. Do not split, crush, or chew the extended-release tablets [see Clinical Pharmacology (12.3)].
- •
- If as dose is missed, take the missed dose as soon as possible. Do not double the next dose.
- •
- The recommended dosage of Altoprev depends on a patient’s indication for usage, LDL-C level, and individual risk for cardiovascular events.
- •
- For patients that require a high-intensity statin or are unable to achieve their LDL-C goal receiving Altoprev 60 mg daily, prescribe alternative LDL-C-lowering treatment.
- •
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating Altoprev, and adjust the dosage if necessary.
2.2 Recommended Dosage in Adult Patients
The recommended dosage range of Altoprev is 20 to 60 mg once daily in the evening.
2.3 Recommended Dosage in Patients with Renal Impairment
- •
- In patients with severe renal impairment [creatinine clearance (CLcr) <30 mL/min], consider if the benefits of increasing the dosage above 20 mg daily outweighs the increased risk of myopathy and rhabdomyolysis [see Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
- •
- There are no dosage adjustment recommendations for patients with mild or moderate renal impairment.
2.4 Dosage Modifications Due to Drug Interactions
Concomitant use of Altoprev with the following drugs requires dosage modification of Altoprev [see Warnings and Precautions (5.1) and Drug Interactions (7.1)].
Patients Taking Danazol, Diltiazem, Dronedarone, or Verapamil
Do not exceed Altoprev 20 mg daily.
Patients Taking Amiodarone
Do not exceed Altoprev 40 mg daily.
3. Dosage Forms and Strengths
Altoprev extended-release tablets are available as follows:
|
Strength |
Tablet Description |
|
20 mg |
orange-colored round, convex shaped tablets imprinted with Andrx logo and 20 on one side |
|
40 mg |
peach-colored round, convex shaped tablets imprinted with Andrx logo and 40 on one side |
|
60 mg |
light peach-colored round, convex shaped tablets imprinted with Andrx logo and 60 on one side |
4. Contraindications
Altoprev is contraindicated in the following conditions:
- •
- Concomitant administration of strong CYP3A inhibitors and erythromycin [see Drug Interactions (7.1)].
- •
- Acute liver failure or decompensated cirrhosis [see Warnings and Precautions (5.3)].
- •
- Hypersensitivity to lovastatin or any excipients in Altoprev. Hypersensitivity reactions, including anaphylaxis, angioedema and Stevens-Johnson syndrome, have been reported [see Adverse Reactions (6.2)].
5. Warnings and Precautions
5.1 Myopathy and Rhabdomyolysis
Altoprev may cause myopathy and rhabdomyolysis. Acute kidney injury secondary to myoglobinuria and rare fatalities have occurred as a result of rhabdomyolysis with statins, including Altoprev.
Risk Factors for Myopathy
Risk factors for myopathy include age 65 years or greater, uncontrolled hypothyroidism, renal impairment, concomitant use with certain other drugs (including other lipid-lowering therapies), and higher Altoprev dosage [see Drug Interactions (7.1)].
Steps to Prevent or Reduce the Risk of Myopathy and Rhabdomyolysis
The concomitant use of strong CYP3A4 inhibitors with Altoprev is contraindicated. If short-term treatment with strong CYP3A4 inhibitors is required, temporarily suspend Altoprev during the duration of strong CYP3A4 inhibitor treatment. The concomitant use of Altoprev with gemfibrozil or cyclosporine is not recommended [see Contraindications (4) and Drug Interactions (7.1)].
Altoprev dosage modifications are recommended for patients taking amiodarone, danazol, diltiazem, dronedarone, and verapamil [see Dosage and Administration (2.4) and Drug Interactions (7.1)].
Lipid modifying doses (>1 gram/day) of niacin, fibrates, colchicine, and ranolazine may also increase the risk of myopathy and rhabdomyolysis [see Drug Interactions (7.1)].
Use of grapefruit juice is not recommended when taking Altoprev [see Drug Interactions (7.1)].
Discontinue Altoprev if markedly elevated CK levels occur or if myopathy is either diagnosed or suspected. Muscle symptoms and CK elevations may resolve if Altoprev is discontinued. Temporarily discontinue Altoprev in patients experiencing an acute or serious condition at high risk of developing renal failure secondary to rhabdomyolysis (e.g., sepsis; shock; severe hypovolemia; major surgery; trauma; severe metabolic, endocrine, or electrolyte disorders; or uncontrolled epilepsy).
Inform patients of the risk of myopathy and rhabdomyolysis when starting or increasing the Altoprev dosage. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness, particularly if accompanied by malaise or fever.
5.2 Immune-Mediated Necrotizing Myopathy
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use, including reports of recurrence when the same or a different statin was administered. IMNM is characterized by proximal muscle weakness and elevated serum creatine kinase that persist despite discontinuation of statin treatment; positive anti-HMGCoA reductase antibody; muscle biopsy showing necrotizing myopathy; and improvement with immunosuppressive agents. Additional neuromuscular and serologic testing may be necessary. Treatment with immunosuppressive agents may be required. Discontinue Altoprev if IMNM is suspected.
5.3 Hepatic Dysfunction
Increases in serum transaminases have occurred with Altoprev [see Adverse Reactions (6.1)]. In most cases, these changes appeared soon after initiation, were transient, were not accompanied by symptoms, and resolved or improved on continued therapy or after a brief interruption in therapy. Persistent increases to more than three times the upper limit of normal (ULN) in serum transaminases have occurred in approximately 1.9% of patients receiving Altoprev in clinical studies. Marked persistent increases of hepatic transaminases have also occurred with Altoprev. There have been rare post marketing reports of fatal and non-fatal hepatic failure in patients taking statins, including Altoprev.
Patients who consume substantial quantities of alcohol and/or have a history of liver disease may be at increased risk for hepatic injury.
Consider liver enzyme testing before Altoprev initiation and when clinically indicated thereafter. Altoprev is contraindicated in patients with acute liver failure or decompensated cirrhosis [see Contraindications (4)]. If serious hepatic injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, promptly discontinue Altoprev.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- •
- Myopathy and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- •
- Immune-Mediated Necrotizing Myopathy [see Warnings and Precautions (5.2)]
- •
- Hepatic Dysfunction [see Warnings and Precautions (5.3)]
- •
- Increases in HbA1c and Fasting Serum Glucose Levels [see Warnings and Precautions (5.4)]
6.1 Clinical Trial Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials 467 patients were treated with Altoprev with mean exposure of approximately 11.6 weeks. The mean age of the population was 56 years, 43% of the population were female, 86% were White, 6% were Black or African American, 1% were Asian, and 7% were other races [see Clinical Studies (14)]. Adverse reactions occurring in ≥5% of patients in any treatment group are shown in Table 1 below.
|
Adverse Reaction |
Treatment |
||
|
Placebo |
Altoprev |
Lovastatin |
|
|
Infection |
9 |
11 |
16 |
|
Headache |
6 |
7 |
8 |
|
Accidental Injury |
9 |
6 |
4 |
|
Arthralgia |
6 |
5 |
6 |
|
Flu Syndrome |
3 |
5 |
5 |
|
Back Pain |
3 |
5 |
5 |
|
Sinusitis |
3 |
4 |
6 |
|
Diarrhea |
6 |
3 |
2 |
|
Myalgia |
15 |
3 |
3 |
|
Pain |
0 |
3 |
2 |
|
Asthenia |
6 |
3 |
2 |
|
Dizziness |
6 |
2 |
2 |
|
Urinary Tract Infection |
6 |
2 |
3 |
Elevations in Liver Enzyme Tests
In the AFCAPS/TexCAPS study, 6,605 patients were treated with lovastatin immediate-release (n=3,304) or placebo (n=3,301). Patients with consecutive elevations of either alanine aminotransferase (ALT) or aspartate aminotransferase (AST) (>3 times ULN), over a median of 5.1 years of follow-up, was not significantly different between the lovastatin immediate-release and placebo groups. Elevated transaminases resulted in discontinuation of 6 (0.2%) patients from therapy in the lovastatin immediate-release group and 4 (0.1%) in the placebo group.
In the EXCEL study, the incidence of persistent increases in serum transaminases over 48 weeks was 0.1% for placebo, 0.1% at 20 mg daily, 0.9% at 40 mg daily, and 1.5% at 80 mg daily in patients treated with lovastatin immediate-release (not an approved dose of Altoprev) [see Dosage and Administration (2.2)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Altoprev. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skeletal: muscle cramps, myopathy, rhabdomyolysis. There have been rare reports of immune-mediated necrotizing myopathy associated with statin use.
Neurological: dysfunction of certain cranial nerves (including alteration of taste, impairment of extra-ocular movement, facial paresis), tremor, vertigo, paresthesia, peripheral neuropathy, peripheral nerve palsy, psychic disturbances, anxiety, insomnia, depression.
There have been rare post marketing reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. These cognitive issues have been reported for all statins. The reports are generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks). There have been rare reports of new-onset or exacerbation of myasthenia gravis, including ocular myasthenia, and reports of recurrence when the same or a different statin was administered.
Hypersensitivity Reactions: An apparent hypersensitivity syndrome has been reported rarely which has included one or more of the following features: anaphylaxis, angioedema, lupus erythematous-like syndrome, polymyalgia rheumatica, dermatomyositis, vasculitis, purpura, thrombocytopenia, leukopenia, hemolytic anemia, positive ANA, ESR increase, eosinophilia, arthritis, urticaria, photosensitivity, fever, chills, flushing, malaise, dyspnea, toxic epidermal necrolysis, erythema multiforme, including Stevens-Johnson syndrome.
Gastrointestinal: pancreatitis, hepatitis, including chronic active hepatitis, cholestatic jaundice, fatty change in liver; and rarely, cirrhosis, fulminant hepatic necrosis, and hepatoma; anorexia, vomiting, fatal and non-fatal hepatic failure.
Skin: alopecia, pruritus, lichen planus. A variety of skin changes (e.g., nodules, discoloration, dryness of skin/mucous membranes, changes to hair/nails) have been reported.
Reproductive: gynecomastia, loss of libido, erectile dysfunction.
Eye: progression of cataracts (lens opacities), ophthalmoplegia.
Laboratory Abnormalities: alkaline phosphatase, g-glutamyl transpeptidase, and bilirubin; thyroid function abnormalities.
Respiratory: interstitial lung disease.
7. Drug Interactions
7.1 Drug Interactions that Increase the Risk of Myopathy and Rhabdomyolysis with Altoprev
Table 2 includes a list of drugs that increase the risk of myopathy and rhabdomyolysis when used concomitantly with Altoprev and instructions for preventing or managing them [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
|
Strong CYP3A4 inhibitors |
|
|
Clinical Impact: |
Lovastatin is a substrate of CYP3A4. Concomitant use of strong CYP3A4 inhibitors with Altoprev increases lovastatin plasma levels and increases the risk of myopathy and rhabdomyolysis, particularly with higher Altoprev dosages. |
|
Intervention: |
Concomitant use of strong CYP3A4 inhibitors with Altoprev is contraindicated [see Contraindications (4)]. If treatment with a CYP3A4 inhibitor is unavoidable, suspend Altoprev during the course of strong CYP3A4 inhibitor treatment. |
|
Examples: |
Select azole anti-fungals (itraconazole, ketoconazole, posaconazole, and voriconazole), select macrolide antibiotics (erythromycin and clarithromycin), select anti-viral medications (dasabuvir/ombitasvir/paritprevir/ritonavir) including cobicistat-containing products (elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate), and nefazodone. |
|
Cyclosporine or Gemfibrozil |
|
|
Clinical Impact: |
The risk of myopathy and rhabdomyolysis is increased with concomitant use of cyclosporine or gemfibrozil with Altoprev. Gemfibrozil may cause myopathy when given alone. |
|
Intervention: |
The concomitant use of Altoprev with cyclosporine or gemfibrozil is not recommended. |
|
Amiodarone, Danazol, Diltiazem, Dronedarone, Verapamil |
|
|
Clinical Impact: |
The risk of myopathy and rhabdomyolysis is increased by concomitant use of amiodarone, danazol, diltiazem, dronedarone, or verapamil with Altoprev. |
|
Intervention: |
For patients taking danazol, diltiazem, dronedarone, or verapamil, do not exceed Altoprev 20 mg daily. For patients taking amiodarone do not exceed Altoprev 40 mg daily [see Dosage and Administration (2.4)]. |
|
Niacin |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been observed with concomitant use of lipid modifying dosages of niacin-containing products (≥1 gram/day niacin) with Altoprev. |
|
Intervention: |
Consider if the benefit of using lipid-modifying doses of niacin concomitantly with Altoprev outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
|
Fibrates (other than Gemfibrozil) |
|
|
Clinical Impact: |
Fibrates may cause myopathy when given alone. The risk of myopathy and rhabdomyolysis is increased with concomitant use of fibrates with Altoprev. |
|
Intervention: |
Consider if the benefit of using fibrates concomitantly with Altoprev outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
|
Ranolazine |
|
|
Clinical Impact: |
The risk of myopathy, including rhabdomyolysis, may be increased by concomitant administration of ranolazine. |
|
Intervention: |
Consider if the benefit of using ranolazine concomitantly with Altoprev outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
|
Colchicine |
|
|
Clinical Impact: |
Cases of myopathy and rhabdomyolysis have been reported with concomitant use of colchicine with Altoprev |
|
Intervention: |
Consider if the benefit of using colchicine concomitantly with Altoprev outweighs the increased risk of myopathy and rhabdomyolysis. If concomitant use is decided, monitor patients for signs and symptoms of myopathy, particularly during initiation of therapy and during upward dose titration of either drug. |
|
Grapefruit Juice |
|
|
Clinical Impact: |
Grapefruit juice can raise the plasma levels of lovastatin and may increase the risk of myopathy and rhabdomyolysis. |
|
Intervention: |
Grapefruit juice is not recommended when taking Altoprev. |
7.2 Altoprev Effects on Other Drugs
Table 3 presents Altoprev’s effect on other drugs and instructions for preventing or managing them.
|
Warfarin |
|
|
Clinical Impact: |
There are postmarketing reports of clinically evident bleeding and/or increased INR in patients taking concomitant statins, including lovastatin, and warfarin. |
|
Intervention: |
In patients taking warfarin, obtain an INR before starting Altoprev and frequently enough after initiation, dose titration or discontinuation to ensure that no significant alteration in INR occurs. Once the INR is stable, monitor INR at regularly recommended intervals. |
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
Discontinue Altoprev when pregnancy is recognized. Alternatively, consider the ongoing therapeutic needs of the individual patient. Altoprev decreases synthesis of cholesterol and possibly other biologically active substances derived from cholesterol; therefore, Altoprev may cause fetal harm when administered to pregnant patients based on the mechanism of action [see Clinical Pharmacology (12.1)]. In addition, treatment of hyperlipidemia is not generally necessary during pregnancy. Atherosclerosis is a chronic process and the discontinuation of lipid-lowering drugs during pregnancy should have little impact on the outcome of long-term therapy of primary hyperlipidemia for most patients.
Available data from case series and prospective and retrospective observational cohort studies over decades of use with statins in pregnant women have not identified a drug-associated risk of major congenital malformations. Published data from prospective and retrospective observational cohort studies with lovastatin use in pregnant women are insufficient to determine if there is a drug-associated risk of miscarriage (see Data). In animal reproduction studies, no teratogenic effects were observed in pregnant rats, mice or rabbits orally administered lovastatin at doses that resulted in up to 25, 5, and 4 times, respectively, the human exposure at the maximum human dose of 80 mg lovastatin immediate-release based on body surface area (mg/m2) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
A Medicaid cohort linkage study of 1,152 statin-exposed pregnant women compared to 886,996 controls did not find a significant teratogenic effect from maternal use of statins in the first trimester of pregnancy, after adjusting for potential confounders – including maternal age, diabetes mellitus, hypertension, obesity, and alcohol and tobacco use – using propensity scorebased methods. The relative risk of congenital malformations between the group with statin use and the group with no statin use in the first trimester was 1.07 (95% confidence interval 0.85 to 1.37) after controlling for confounders, particularly pre-existing diabetes mellitus. There were also no statistically significant increases in any of the organ-specific malformations assessed after accounting for confounders. In the majority of pregnancies, statin treatment was initiated prior to pregnancy and was discontinued at some point in the first trimester when pregnancy was identified. Study limitations include reliance on physician coding to define the presence of a malformation, lack of control for certain confounders such as body mass index, use of prescription dispensing as verification for the use of a statin, and lack of information on non-live births.
Animal Data
In pregnant rats given oral gavage doses of 100, 200, 400, 800 mg/kg/day lovastatin from gestation days 6 through 20 during the period of organogenesis, reduced fetal body weights were observed at all doses and skeletal malformations were observed at ≥400 mg/kg/day, corresponding to 50 times the 80 mg/day -release maximum human dose, based on body surface area (mg/m2).
In pregnant mice given oral gavage doses of 8, 80, 800 mg/kg/day lovastatin from gestation days 6 through 15 during the period of organogenesis, reduced fetal body weights were observed at ≥80 mg/kg/day and skeletal malformations were observed at 800 mg/kg/day, corresponding to 5 and 50 times the 80 mg/day lovastatin immediate-release maximum human dose, based on body surface area (mg/m2).
In pregnant rabbits given an oral gavage dose of 15 mg/kg/day lovastatin from gestation days 6 through 18 during the period of organogenesis, no teratogenic effects were observed. Doses were 4 times the 80 mg/day lovastatin immediate-release maximum human dose, based on body surface area (mg/m2).
In pregnant rats given oral gavage doses of 2, 20, 200 mg/kg/day lovastatin by oral gavage from gestation day 15 through lactation day 21 (weaning), increased mortality of offspring was observed at ≥20 mg/kg/day and development delays were observed at 200 mg/kg/day, corresponding to 2.4 and 24 times, respectively, the 80 mg/day lovastatin immediate-release maximum human dose, based on body surface area (mg/m2).
8.2 Lactation
Risk Summary
There is no information about the presence of lovastatin in human or animal milk, the effects of the drug on the breastfed infant or the effects of the drug on milk production. However, it has been shown that another drug in this class passes into human milk. Statins, including Altoprev, decrease cholesterol synthesis and possibly the synthesis of other biologically active substances derived from cholesterol and may cause harm to the breastfed infant.
Because of the potential for serious adverse reactions in a breastfed infant, based on the mechanism of action, advise patients that breastfeeding is not recommended during treatment with Altoprev [see Use in Specific Populations (8.1) and Clinical Pharmacology (12.1)].
8.4 Pediatric Use
The safety and effectiveness of Altoprev have not been established in pediatric patients.
8.5 Geriatric Use
Of the 467 patients who received Altoprev in clinical studies, 18% were 65 years and older. In the clinical studies conducted with lovastatin immediate-release (EXCEL and AFCAPS/TexCAPS), 21% (3094/14850) of patients were ≥65 years of age. In pharmacokinetic studies with lovastatin immediate-release, the mean plasma level of HMG-CoA reductase inhibitory activity was shown to be approximately 45% higher in elderly patients between 70-78 years of age compared with patients between 18-30 years of age [see Clinical Pharmacology (12.3)].
Advanced age (≥65 years) is a risk factor for Altoprev-associated myopathy and rhabdomyolysis. Dose selection for an elderly patient should be cautious, recognizing the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of myopathy. Monitor geriatric patients receiving Altoprev for the increased risk of myopathy [see Warnings and Precautions (5.1)].
8.6 Renal Impairment
In a study of patients with severe renal impairment (creatinine clearance 10–30 mL/min), the plasma concentrations of total inhibitors after a single dose of lovastatin were approximately two-fold higher than those in healthy volunteers.
Renal impairment is a risk factor for myopathy and rhabdomyolysis. Monitor all patients with renal impairment for development of myopathy. In patients with severe renal impairment, consider if the benefits of increasing the dosage above 20 mg daily outweighs the increased risk of myopathy and rhabdomyolysis [see Dosage and Administration (2.3), Warnings and Precautions (5.1), and Clinical Pharmacology (12.3)].
10. Overdosage
No specific antidotes for Altoprev are known. In the event of an overdose of Altoprev, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11. Altoprev Description
Altoprev (lovastatin extended-release) tablets are an inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase (statin) isolated from a strain of Aspergillus terreus.
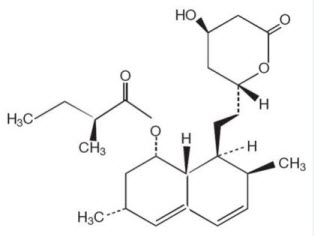
Lovastatin is [1 S –[1α(R*),3α,7β,8β(2 S*,4 S*),8aβ]]-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl]-1-naphthalenyl 2-methylbutanoate. The empirical formula of lovastatin is C24H36O5 and its molecular weight is 404.55. Its structural formula is:
Lovastatin is a white, nonhygroscopic crystalline powder that is insoluble in water and sparingly soluble in ethanol, methanol, and acetonitrile.
Altoprev extended-release tablets for oral use contain 20 mg, 40 mg, or 60 mg of lovastatin. In addition, each tablet contains the following inactive ingredients: acetyltributyl citrate; butylated hydroxy anisole; candelilla wax; cellulose acetate; confectioner’s sugar (contains corn starch); F D & C yellow # 6; glyceryl monostearate; hypromellose; hypromellose phthalate; lactose; methacrylic acid copolymer, type B; polyethylene glycols (PEG 400, PEG 8000); polyethylene oxides; polysorbate 80; propylene glycol; silicon dioxide; sodium chloride; sodium lauryl sulfate; synthetic black iron oxide; red iron oxide; talc; titanium dioxide and triacetin.
12. Altoprev - Clinical Pharmacology
12.1 Mechanism of Action
Lovastatin is a lactone prodrug that is hydrolyzed in vivo to its active β-hydroxyacid form, an inhibitor of HMG-CoA reductase, the rate-limiting enzyme that converts HMG-CoA to mevalonate, a precursor of cholesterol.
12.2 Pharmacodynamics
Inhibition of HMG-CoA reductase by lovastatin accelerates the expression of LDL-receptors, followed by the uptake of LDL-C from blood to the liver, leading to a decrease in plasma LDL-C and total cholesterol. Sustained inhibition of cholesterol synthesis in the liver also decreases levels of very-low-density lipoproteins. The maximum LDL-C reduction of lovastatin is usually achieved by 4 weeks and is maintained after that.
12.3 Pharmacokinetics
Absorption
A pharmacokinetic study carried out with Altoprev involved measurement of the systemic concentrations of lovastatin (pro-drug), lovastatin acid (active-drug), and total and active inhibitors of HMG-CoA reductase. The pharmacokinetic parameters in 12 subjects with hypercholesterolemia at steady state, after 28 days of treatment of Altoprev 40 mg, is summarized in Table 4.
| L = lovastatin, LA = lovastatin acid, TI = total inhibitors of HMG-CoA reductase, AI = active inhibitors of HMG-CoA reductase, Cmax = highest observed plasma concentration, Cmin = trough concentration at t = 24 hours after dosing, Tmax = time at which the Cmax occurred, AUC0-24hr = area under the plasma concentration-time curve from time 0 to 24 hr after dosing, calculated by the linear trapezoidal rule. * Administered at bedtime. |
||||||||||||||
|
Drug |
Cmax (ng/mL) |
Cmin (ng/mL) |
Tmax (h) |
AUC0-24hr
|
||||||||||
|
|
L |
LA |
TI |
AI |
L |
LA |
TI |
AI |
L |
LA |
L |
LA |
TI |
AI |
|
Altoprev |
5.5 |
5.8 |
17.3 |
13.4 |
2.6 |
3.1 |
9.1 |
4.3 |
14.2 |
11.8 |
77 |
87 |
263 |
171 |
|
Lovastatin IR |
7.8 |
11.9 |
36.2 |
26.6 |
0.4 |
0.7 |
2.4 |
2.1 |
3.3 |
5.3 |
45 |
83 |
252 |
186 |
The extended-release properties of Altoprev are characterized by a prolonged absorptive phase, which results in a longer Tmax and lower Cmax for lovastatin (pro-drug) and its major metabolite, lovastatin acid, compared to lovastatin immediate-release.
At the 40 mg dose, the bioavailability of lovastatin (pro-drug) as measured by the AUC0-24hr was greater for Altoprev compared to lovastatin immediate-release (as measured by a chemical assay), while the bioavailability of total and active inhibitors of HMG-CoA reductase were equivalent to lovastatin immediate-release (as measured by an enzymatic assay).
Altoprev appears to have dose linearity for doses from 10 mg up to 60 mg per day.
Effect of Food
When Altoprev was given after a meal, plasma concentrations of lovastatin and lovastatin acid were about 0.5 - 0.6 times those found when Altoprev was administered in the fasting state, indicating that food decreases the bioavailability of Altoprev. There was an association between the bioavailability of Altoprev and dosing after mealtimes. Bioavailability was lowered under the following conditions, (from higher bioavailability to lower bioavailability) in the following order: under overnight fasting conditions, before bedtime, with dinner, and with a high fat breakfast. In a multicenter, randomized, parallel group study, patients were administered 40 mg of Altoprev at three different times; before breakfast, after dinner and at bedtime. Although there was no statistical difference in the extent of lipid change between the three groups, there was a numerically greater reduction in LDL-C and TG and an increase in HDL-C when Altoprev was administered at bedtime. Results of this study are displayed in Table 5.
| N = 22 for the Before Breakfast group, N = 23 for the After Dinner group, and N = 23 for the Before Bedtime group. * All changes from baseline are statistically significant. |
|||||||||||||||||||
|
|
LDL-C |
HDL-C |
TOTAL-C |
TG |
|||||||||||||||
|
Before Breakfast |
-32.0% |
8.4% |
-22.2% |
-10.2% |
|||||||||||||||
|
After Dinner |
-34.1% |
7.4% |
-23.6% |
-11.2% |
|||||||||||||||
|
Before Bedtime |
-36.9% |
11.1% |
-25.5% |
-19.7% |
|||||||||||||||
Distribution: Both lovastatin and its β-hydroxyacid metabolite are highly bound (>95%) to human plasma proteins. Animal studies demonstrated that lovastatin crosses the blood-brain and placental barriers.
Elimination
Metabolism
Lovastatin is a lactone that is readily hydrolyzed in vivo to the corresponding β-hydroxyacid, an inhibitor of HMG-CoA reductase. Inhibition of HMG-CoA reductase is the basis for an assay in pharmacokinetic studies of the β-hydroxyacid metabolites (active inhibitors) and, following base hydrolysis, active plus latent inhibitors (total inhibitors) in plasma following administration of lovastatin.
The major active metabolites present in human plasma are the β-hydroxyacid of lovastatin, its 6’-hydroxy derivative, and two additional metabolites.
Excretion
In a single-dose study with Altoprev, the amounts of lovastatin and lovastatin acid excreted in the urine were below the lower limit of quantitation of the assay (1.0 ng/mL), indicating that negligible excretion of Altoprev occurs through the kidney.
Lovastatin undergoes extensive first-pass extraction in the liver, its primary site of action, with subsequent excretion of drug equivalents in the bile.
Specific Populations
Geriatric Patients:
In a study with lovastatin immediate-release which included 16 elderly patients between 70-78 years of age who received lovastatin immediate-release 80 mg/day (not an approved dose of Altoprev) [see Dosage and Administration (2.2)], the mean plasma level of HMG-CoA reductase inhibitory activity was increased approximately 45% compared with 18 patients between 18-30 years of age [see Use in Specific Populations (8.5)].
Male and Female Patients
In a single dose pharmacokinetic study with Altoprev, there were no statistically significant differences in pharmacokinetic parameters between males (n=12) and females (n=10), although exposure tended to be higher in males than females.
Renal Impairment
In a study of patients with severe renal impairment (creatinine clearance 10-30 mL/min), the plasma concentrations of total inhibitors after a single dose of lovastatin were approximately two-fold higher than those in healthy volunteers.
Drug Interaction Studies
The risk of myopathy is increased by high levels of HMG-CoA reductase inhibitory activity in plasma. Strong inhibitors of CYP3A can raise the plasma levels of HMG-CoA reductase inhibitory activity and increase the risk of myopathy [see Warnings and Precautions (5.1) and Drug Interactions (7)].
Lovastatin is a substrate for CYP3A4 [see Drug Interactions (7)]. Grapefruit juice contains one or more components that inhibit CYP3A and can increase the plasma concentrations of drugs metabolized by CYP3A4. In one study, 10 subjects consumed 200 mL of double-strength grapefruit juice (one can of frozen concentrate diluted with one rather than 3 cans of water) three times daily for 2 days and an additional 200 mL double-strength grapefruit juice together with and 30 and 90 minutes following a single dose of 80 mg lovastatin on the third day. This regimen of grapefruit juice resulted in mean increases in the concentration of lovastatin and its beta-hydroxyacid metabolite (as measured by the area under the concentration-time curve) of 15-fold and 5-fold respectively (as measured using a chemical assay – liquid chromatography/tandem mass spectrometry). In a second study, 15 subjects consumed one 8 oz glass of single-strength grapefruit juice (one can of frozen concentrate diluted with 3 cans of water) with breakfast for 3 consecutive days and a single dose of 40 mg lovastatin in the evening of the third day. This regimen of grapefruit juice resulted in a mean increase in the plasma concentration (as measured by the area under the concentration-time curve) of active and total HMG-CoA reductase inhibitory activity [using a validated enzyme inhibition assay different from that used in the first study, both before (for active inhibitors) and after (for total inhibitors) base hydrolysis] of 1.34-fold and 1.36-fold, respectively, and of lovastatin and its β-hydroxyacid metabolite (measured using a chemical assay – liquid chromatography/tandem mass spectrometry) of 1.94-fold and 1.57-fold, respectively. The effect of amounts of grapefruit juice between those used in these two studies on lovastatin pharmacokinetics has not been studied.
| * Results based on a chemical assay. † Lovastatin acid refers to the β-hydroxyacid of lovastatin. ‡ Results could be representative of strong CYP3A inhibitors such as ketoconazole, posaconazole, clarithromycin, telithromycin, HIV protease inhibitors, and nefazodone. § The mean total AUC of lovastatin without itraconazole phase could not be determined due to assay’s detection limit. ¶ The effect of amounts of grapefruit juice between those used in these two studies on lovastatin pharmacokinetics has not been studied. # Double-strength: one can of frozen concentrate diluted with one can of water. Grapefruit juice was administered TID for 2 days, and 200 mL together with single dose lovastatin and 30 and 90 minutes following single dose lovastatin on Day 3. Þ Single-strength: one can of frozen concentrate diluted with 3 cans of water. Grapefruit juice was administered with breakfast for 3 days, and lovastatin was administered in the evening on Day 3. ß Cyclosporine-treated post kidney transplant patients with stable graft function, transplanted at least 9 months prior to study. à NR = Analyte not reported. è Lactone converted to acid by hydrolysis prior to analysis. Figure represents total unmetabolized acid and lactone. Π Analyte not determined |
||||||||||||||||||||||||||
|
Coadministered Drug or
|
Dosing of
|
Dosing of
|
AUC Ratio*
(with / without
No Effect = 1.00 |
Cmax Ratio*
(with / without
No Effect = 1.00 |
||||||||||||||||||||||
|
Lovastatin |
Lovastatin acid† |
Lovastatin |
Lovastatin acid† |
|||||||||||||||||||||||
|
Contraindicated with lovastatin, [see Contraindications (4) and Warnings and Precautions (5.1)] |
||||||||||||||||||||||||||
|
Itraconazole‡ |
200 mg QD for 4 days |
40 mg on Day 4 |
§ |
20 |
> 25§ |
13.0 |
||||||||||||||||||||
|
|
100 mg QD for 4 days |
40 mg on Day 4 |
> 14.8§ |
15.4 |
14.5 |
11.5 |
||||||||||||||||||||
|
Not recommended with lovastatin, [see Warnings and Precautions (5.1)] |
||||||||||||||||||||||||||
|
Cyclosporine |
Not describedß |
10 mg QD for 10 days on Day 10 |
3- to 5-fold |
NRà |
NRà |
NRà |
||||||||||||||||||||
|
Gemfibrozil |
600 mg BID for |
40 mg on Day 3 |
0.96 |
2.80 |
0.88 |
2.81 |
||||||||||||||||||||
|
Grapefruit Juice¶ |
200 mL of double-strength TID for 2 days |
80 mg on Day 3 |
15.3 |
5.0 |
11.8 |
4.0 |
||||||||||||||||||||
|
Grapefruit Juice¶ |
8 oz (about 250 mL) of single-strengthÞ for 4 days |
40 mg on Day 3 |
1.94 |
1.57 |
2.26 |
1.65 |
||||||||||||||||||||
|
Not recommended with > 20 mg lovastatin, [see Warnings and Precautions (5.1)] |
||||||||||||||||||||||||||
|
Diltiazem |
120 mg BID for 14 days |
20 mg on Day 14 |
3.57è |
NDΠ |
4.33è |
NDΠ |
||||||||||||||||||||
|
No dosing adjustments required for the following: |
||||||||||||||||||||||||||
|
Propanolol |
40 mg BID for 2.5 days |
20 mg on Day 2 |
NDΠ |
0.87 |
NDΠ |
0.81 |
||||||||||||||||||||
Digoxin
In patients with hypercholesterolemia, concomitant administration of lovastatin and digoxin resulted in no effect on digoxin plasma concentrations.
Oral Hypoglycemic Agents
In pharmacokinetic studies of lovastatin immediate-release in hypercholesterolemic non-insulin dependent diabetic patients, there was no drug interaction with glipizide or with chlorpropamide.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 21-month carcinogenic study in mice with lovastatin immediate-release, there was a statistically significant increase in the incidence of hepatocellular carcinomas and adenomas in both males and females at 500 mg/kg/day. This dose produced a total plasma drug exposure 3 to 4 times that of humans given the highest recommended dose of lovastatin (drug exposure was measured as total HMG-CoA reductase inhibitory activity in extracted plasma). Tumor increases were not seen at 20 and 100 mg/kg/day, doses that produced drug exposures of 0.3 to 2 times that of humans at the 80 mg/day lovastatin immediate-release dose. A statistically significant increase in pulmonary adenomas was seen in female mice at approximately 4 times the human drug exposure. [Although mice were given 300 times the human dose (HD) on a mg/kg body weight basis, plasma levels of total inhibitory activity were only 4 times higher in mice than in humans given 80 mg of lovastatin immediate-release].
There was an increase in incidence of papilloma in the non-glandular mucosa of the stomach of mice beginning at exposures of 1 to 2 times that of humans given lovastatin immediate-release. The glandular mucosa was not affected. The human stomach contains only glandular mucosa.
In a 24-month carcinogenicity study in rats, there was a positive dose response relationship for hepatocellular carcinogenicity in males at drug exposures between 2-7 times that of human exposure at 80 mg/day lovastatin immediate-release (doses in rats were 5, 30 and 180 mg/kg/day).
An increased incidence of thyroid neoplasms in rats appears to be a response that has been seen with other HMG-CoA reductase inhibitors.
A chemically similar drug in this class was administered to mice for 72 weeks at 25, 100, and 400 mg/kg body weight, which resulted in mean serum drug levels approximately 3, 15, and 33 times higher than the mean human serum drug concentration (as total inhibitory activity) after a 40 mg oral dose of lovastatin immediate-release. Liver carcinomas were significantly increased in high-dose females and mid- and high-dose males, with a maximum incidence of 90 percent in males. The incidence of adenomas of the liver was significantly increased in mid- and high-dose females. Drug treatment also significantly increased the incidence of lung adenomas in mid- and high-dose males and females. Adenomas of the Harderian gland (a gland of the eye of rodents) were significantly higher in high dose mice than in controls.
No evidence of mutagenicity was observed with lovastatin immediate-release in a microbial mutagen test using mutant strains of Salmonella typhimurium with or without rat or mouse liver metabolic activation. In addition, no evidence of damage to genetic material was noted in an in vitro alkaline elution assay using rat or mouse hepatocytes, a V-79 mammalian cell forward mutation study, an in vitro chromosome aberration study in CHO cells, or an in vivo chromosomal aberration assay in mouse bone marrow.
Drug-related testicular atrophy, decreased spermatogenesis, spermatocytic degeneration and giant cell formation were seen in dogs starting at 20 mg/kg/day with lovastatin immediate-release. Similar findings were seen with another drug in this class. No drug-related effects on fertility were found in studies with lovastatin in rats. However, in studies with a similar drug in this class, there was decreased fertility in male rats treated for 34 weeks at 25 mg/kg body weight, although this effect was not observed in a subsequent fertility study when this same dose was administered for 11 weeks (the entire cycle of spermatogenesis, including epididymal maturation). In rats treated with this same reductase inhibitor at 180 mg/kg/day, seminiferous tubule degeneration (necrosis and loss of spermatogenic epithelium) was observed. No microscopic changes were observed in the testes from rats of either study. The clinical significance of these findings is unclear.
14. Clinical Studies
Primary Hyperlipidemia in Adults
Altoprev has been shown to reduce Total-C, LDL-C, and TG and increase HDL-C in patients with hyperlipidemia. Near maximal response was observed after four weeks of treatment and the response was maintained with continuation of therapy for up to 6 months.
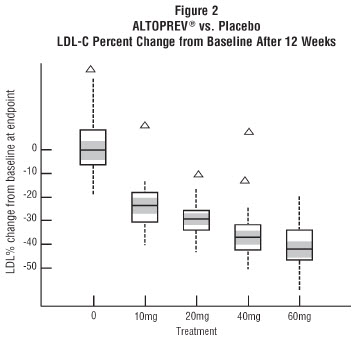
In a 12-week, multicenter, placebo-controlled, double-blind, trial in adult males and females 21 to 70 years of age with primary hyperlipidemia, once daily administration of Altoprev 20 to 60 mg in the evening was compared to placebo. Altoprev produced dose related reductions in LDL-C and Total-C. The lipid changes with Altoprev treatment in this trial, from baseline to endpoint, are displayed in Table 7.
| N= the number of patients with values at both baseline and endpoint. *All lipid changes with Altoprev were statistically significant compared to placebo. **For LDL-C, 33 patients had values at baseline and endpoint. |
|||||
|
Treatment |
N |
LDL-C |
HDL-C |
TOTAL-C |
TG |
|
Placebo |
34 |
1.3 |
5.6 |
3.4 |
8.7 |
|
Altoprev® 20 mg |
34** |
-29.6 |
12.0 |
-20.9 |
-13.0 |
|
Altoprev® 40 mg |
33 |
-35.8 |
13.1 |
-25.4 |
-9.9 |
|
Altoprev® 60 mg |
35 |
-40.8 |
11.6 |
-29.2 |
-25.1 |
The range of LDL-C responses is represented graphically in the following figure (Figure 1):
The distribution of LDL-C responses is represented graphically by the boxplots in Figure 1. The bottom line of the box represents the 25th percentile and the top line, the 75th percentile. The horizontal line in the box represents the median and the gray area is the 95% confidence interval for the median. The range of responses is depicted by the tails and outliers.
Expanded Clinical Evaluation of Lovastatin Immediate-Release (EXCEL) Study
Lovastatin immediate-release was compared to placebo in 8,245 patients with hyperlipidemia [Total-C 240 to 300mg/dL, LDL-C >160 mg/dL] in the randomized, double-blind, parallel, 48-week EXCEL trial. All changes in the lipid measurements (see Table 8) observed in patients administered lovastatin immediate-release were dose-related and significantly different from placebo (p≤0.001). These results were sustained throughout the trial.
| b.i.d. = twice daily, q.p.m. = every evening **Patients enrolled ***Not an approved dose of Altoprev |
|||||||||||
|
DOSAGE |
N** |
TOTAL-C
|
LDL-C
|
HDL-C
|
TG
|
||||||
|
Placebo |
1663 |
+0.7 |
+0.4 |
+2.0 |
+4 |
||||||
|
Lovastatin IR 20 mg q.p.m. |
1642 |
-17 |
-24 |
+6.6 |
-10 |
||||||
|
40 mg q.p.m. |
1645 |
-22 |
-30 |
+7.2 |
-14 |
||||||
|
20 mg b.i.d. |
1646 |
-24 |
-34 |
+8.6 |
-16 |
||||||
|
40 mg b.i.d.*** |
1649 |
-29 |
-40 |
+9.5 |
-19 |
||||||
Heterozygous Familial Hypercholesterolemia in Adults
Lovastatin immediate-release has been shown to be effective in reducing Total-C and LDL-C in heterozygous familial and non-familial forms of primary hypercholesterolemia. A response was seen within 2 weeks, and the maximum response occurred within 4-6 weeks. The response was maintained during continuation of therapy.
Prevention of Coronary Heart Disease
The Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS), a double-blind, randomized, placebo-controlled, primary prevention trial, demonstrated that treatment with lovastatin immediate-release decreased the rate of acute major coronary events (composite endpoint of myocardial infarction, unstable angina, and sudden cardiac death) compared with placebo during a median of 5.1 years of follow-up. Participants were males (ages 45 to 73) and females (ages 55 to 73) without symptomatic cardiovascular disease with average to moderately elevated Total-C and LDL-C, below average HDL-C, and who were at high risk based on elevated Total-C/HDL-C. In addition to age, 63% of the participants had at least one other risk factor (baseline HDL-C <35 mg/dL, hypertension, family history, smoking and diabetes).
AFCAPS/TexCAPS enrolled 6,605 participants (5,608 males, 997 females) based on the following lipid entry criteria: Total-C range of 180 to 264 mg/dL, LDL-C range of 130 to 190 mg/dL, HDL-C of ≤45 mg/dL for males and ≤47 mg/dL for females, and TG of ≤400 mg/dL. Participants were treated with standard care, including diet, and either lovastatin immediate-release 20 mg to 40 mg daily (n= 3,304) or placebo (n= 3,301). Approximately 50% of the participants treated with lovastatin immediate-release were titrated to 40 mg daily when their LDL-C remained >110 mg/dL at the 20-mg starting dose.
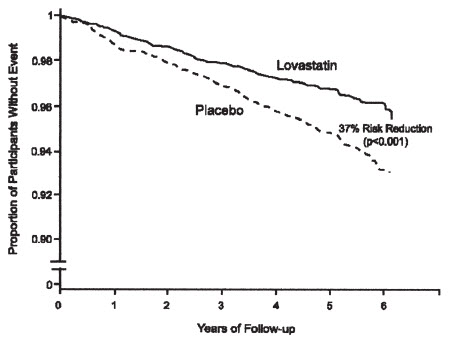
Lovastatin immediate-release reduced the risk of a first acute major coronary event, the primary efficacy endpoint, by 37% (lovastatin immediate-release 3.5%, placebo 5.5%; p<0.001; Figure 2). A first acute major coronary event was defined as myocardial infarction (54 participants on lovastatin immediate-release, 94 on placebo) or unstable angina (54 vs. 80) or sudden cardiac death (8 vs. 9). Furthermore, among the secondary endpoints, lovastatin immediate-release reduced the risk of unstable angina by 32% (1.8% vs. 2.6%; p=0.023), of myocardial infarction by 40% (1.7% vs. 2.9%; p=0.002), and of undergoing coronary revascularization procedures (e.g., coronary artery bypass grafting or percutaneous transluminal coronary angioplasty) by 33% (3.2% vs. 4.8%; p=0.001). Trends in risk reduction associated with treatment with lovastatin immediate-release were consistent across males and females, smokers and non-smokers, hypertensives and non-hypertensives, and older and younger participants. Participants with ≥2 risk factors had risk reductions (RR) in both acute major coronary events (RR 43%) and coronary revascularization procedures (RR 37%). Because there were too few events among those participants with age as their only risk factor in this study, the effect of lovastatin immediate-release on outcomes could not be adequately assessed in this subgroup.
Atherosclerosis
In the Canadian Coronary Atherosclerosis Intervention Trial (CCAIT), the effect of therapy with lovastatin immediate-release on coronary atherosclerosis was assessed by coronary angiography in hyperlipidemic patients. In this randomized, double-blind, controlled clinical trial, patients were treated with conventional measures (usually diet and 325 mg of aspirin every other day) and either lovastatin immediate-release 20 mg to 80 mg daily or placebo (the 80 mg dose is not approved for Altoprev [see Dosage and Administration (2.2)]). Angiograms were evaluated at baseline and at two years by computerized quantitative coronary angiography (QCA). Lovastatin immediate-release significantly slowed the progression of lesions as measured by the mean change per-patient in minimum lumen diameter (the primary endpoint) and percent diameter stenosis, and decreased the proportions of patients categorized with disease progression (33% vs. 50%) and with new lesions (16% vs. 32%).
16. How is Altoprev supplied
Altoprev extended-release tablets are supplied as follows:
|
Strength |
How Supplied |
NDC |
Tablet Description |
|
20 mg |
bottles of 30 |
70515-628-30 |
orange-colored round, convex shaped tablets imprinted with Andrx logo and 20 on one side |
|
40 mg |
bottles of 30 |
70515-629-30 |
peach-colored round, convex shaped tablets imprinted with Andrx logo and 40 on one side |
|
60 mg |
bottles of 30 |
70515-630-30 |
light peach-colored round, convex shaped tablets imprinted with Andrx logo and 60 on one side |
Storage
Store at 20-25°C (68-77°F) - Excursions Permitted to 15°C -30°C (59°F -86°F) [See USP Controlled Room Temperature]. Avoid excessive heat and humidity.
17. Patient Counseling Information
Myopathy and Rhabdomyolysis
Advise patients that Altoprev may cause myopathy and rhabdomyolysis. Inform patients that the risk is also increased when taking certain types of medication or consuming grapefruit juice and they should discuss all medication, both prescription and over the counter, with their healthcare provider. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever [see Warnings and Precautions (5.1), Drug Interactions (7.1)].
Hepatic Dysfunction
Inform patients that Altoprev may cause liver enzyme elevations and possibly liver failure. Advise patients to promptly report fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice [see Warnings and Precautions (5.3)].
Increases in HbA1c and Fasting Serum Glucose Levels
Inform patients that increases in HbA1c and fasting serum glucose levels may occur with Altoprev. Encourage patients to optimize lifestyle measures, including regular exercise, maintaining a healthy body weight, and making healthy food choices [see Warnings and Precautions (5.4)].
Pregnancy
Advise pregnant patients and patients who can become pregnant of the potential risk to a fetus. Advise patients to inform their healthcare provider of a known or suspected pregnancy to discuss if Altoprev should be discontinued [see Use in Specific Populations (8.1)].
Lactation
Advise patients that breastfeeding is not recommended during treatment with Altoprev [see Use in Specific Populations (8.2)].
Missed Dose
Instruct patients to take Altoprev only as prescribed. If a dose is missed, it should be taken as soon as possible. Advise patients not to double their next dose.
PRINCIPAL DISPLAY PANEL - 20 mg BOTTLE
NDC 70515-628-30 30 Tablets
Altoprev®
(lovastatin)
extended-release tablets
20 mg
Rx only
COVIS
Each tablet contains lovastatin 20 mg.
Read package insert for prescribing
information.
KEEP OUT OF THE REACH OF CHILDREN.
Dispense in tight, light-resistant container
as defined in the USP.
Store at 20°-25°C (68°-77°F). Excursions
permitted to 15°-30°C (59°-86°F) [See
USP Controlled Room Temperature].
Avoid excessive heat and humidity.
Manufactured for:
Covis Pharma
Zug, 6300 Switzerland
Made in Hungary Rev. 3/16 100242
PRINCIPAL DISPLAY PANEL - 40 mg BOTTLE
NDC 70515-629-30 30 Tablets
Altoprev®
(lovastatin)
extended-release tablets
40 mg
Rx only
COVIS
Each tablet contains lovastatin 40 mg.
Read package insert for prescribing
information.
KEEP OUT OF THE REACH OF CHILDREN.
Dispense in tight, light-resistant container
as defined in the USP.
Store at 20°-25°C (68°-77°F). Excursions
permitted to 15°-30°C (59°-86°F) [See
USP Controlled Room Temperature].
Avoid excessive heat and humidity.
Manufactured for:
Covis Pharma
Zug, 6300 Switzerland
Made in Hungary Rev. 3/16 100243
PRINCIPAL DISPLAY PANEL - 60 mg BOTTLE
NDC 70515-630-30 30 Tablets
Altoprev®
(lovastatin)
extended-release tablets
60 mg
Rx only
COVIS
Each tablet contains lovastatin 60 mg.
Read package insert for prescribing
information.
KEEP OUT OF THE REACH OF CHILDREN.
Dispense in tight, light-resistant container
as defined in the USP.
Store at 20°-25°C (68°-77°F). Excursions
permitted to 15°-30°C (59°-86°F) [See
USP Controlled Room Temperature].
Avoid excessive heat and humidity.
Manufactured for:
Covis Pharma
Zug, 6300 Switzerland
Made in Hungary Rev. 3/16 100244
| ALTOPREV
lovastatin tablet, extended release |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALTOPREV
lovastatin tablet, extended release |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALTOPREV
lovastatin tablet, extended release |
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Covis Pharma US, Inc (118094829) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Actavis Laboratories FL, Inc. | 020778751 | MANUFACTURE(70515-628, 70515-629, 70515-630) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Actavis Laboratories FL, Inc. | 014759176 | ANALYSIS(70515-628, 70515-629, 70515-630) , LABEL(70515-628, 70515-629, 70515-630) , MANUFACTURE(70515-628, 70515-629, 70515-630) , PACK(70515-628, 70515-629, 70515-630) | |
Frequently asked questions
More about Altoprev (lovastatin)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (2)
- Drug images
- Latest FDA alerts (3)
- Side effects
- Dosage information
- During pregnancy
- Drug class: statins
- Breastfeeding
- En español