The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Thyroxine L Tablets
This page contains information on Thyroxine L Tablets for veterinary use.The information provided typically includes the following:
- Thyroxine L Tablets Indications
- Warnings and cautions for Thyroxine L Tablets
- Direction and dosage information for Thyroxine L Tablets
Thyroxine L Tablets
This treatment applies to the following species: Manufacturer: Butler Schein™ Animal Health
Manufacturer: Butler Schein™ Animal Health
Levothyroxine Sodium Tablets, USP
Thyroid Replacement Therapy
Thyroxine L Tablets Caution
Federal law restricts this drug to use by or on the order of a veterinarian.
Description:
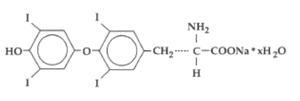
Each tablet provides synthetic crystalline levothyroxine sodium (L-thyroxine). The structural formula for levothyroxine sodium is:
Levothyroxine Sodium Action:
Levothyroxine sodium acts, as does endogenous thyroxine, to stimulate metabolism, growth, development and differentiation of tissues. It increases the rate of energy exchange and increases the maturation rate of the epiphyses. Levothyroxine sodium is absorbed rapidly from the gastrointestinal tract after oral administration. Following absorption, the compound becomes bound to the serum alpha globulin fraction. For purposes of comparison, 0.1mg of levothyroxine sodium elicits a clinical response approximately equal to that produced by one grain (65mg) of desiccated thyroid.
Thyroxine L Tablets Indications
Provides thyroid replacement therapy in all conditions of inadequate production of thyroid hormones (hypothyroidism) in the dog. Hypothyroidism is the generalized metabolic disease resulting from deficiency of the thyroid hormones levothyroxine (T4) and liothyronine (T3).
Levothyroxine Sodium tablets will provide levothyroxine (T4) as a substrate for the physiologic deiodination to liothyronine (T3). Administration of levothyroxine sodium alone will result in complete physiologic thyroid replacement. Canine hypothyroidism is usually primary, i.e., due to atrophy of the thyroid gland. In the majority of cases the atrophy is associated with lymphocytic thyroiditis and in the remainder it is noninflammatory and as of yet unknown etiology. Less than 10 percent of cases of hypothyroidism are secondary, i.e., due to deficiency of thyroid stimulating hormone (TSH). TSH deficiency may occur as a component of congenital hypopituitarism or as an acquired disorder in adult dogs, in which case it is invariably due to the growth of a pituitary tumor.
Hypothyroidism in the Dog:
Hypothyroidism usually occurs in middle-aged and older dogs although the condition will sometimes be seen in younger dogs of the larger breeds. Neutered animals of either sex are also frequently affected, regardless of age. The following are clinical signs of hypothyroidism in dogs: Lethargy, lack of endurance, increased sleeping; Reduced interest, alertness and excitability; Slow heart rate, weak apex beat and pulse, low voltage on ECG; Preference for warmth, low body temperature, cool skin; Increased body weight; Stiff and slow movements, dragging of front feet; Head tilt, disturbed balance, unilateral facial paralysis; Atrophy of epidermis, thickening of dermis; Surface and follicular hyperkeratosis, pigmentation; Puffy face, blepharoptosis, tragic expression; Dry, coarse, sparse coat, slow regrowth after clipping; Retarded turnover of hair (carpet coat of boxers); Shortening or absence of estrus, lack of libido; Dry feces, occasional diarrhea. Hypercholesterolemia. Normochromic, normocytic anemia; Elevated serum creatinine phosphokinase.
Contraindications
Levothyroxine sodium therapy is contraindicated in thyrotoxicosis, acute myocardial infarction and uncorrected adrenal insufficiency. Use in pregnant bitches has not been evaluated.
Precautions
The effects of levothyroxine sodium therapy are slow in being manifested. Overdosage of any thyroid drug may produce the signs and symptoms of thyrotoxicosis including, but not limited to: polydipsia, polyuria, polyphagia, reduced heat tolerance and hyperactivity or personality change. Administer with caution to animals with clinically significant heart disease, hypertension or other complications for which a sharply increased metabolic rate might prove hazardous.
Adverse Reactions
There are no particular adverse reactions associated with levothyroxine sodium therapy at the recommended dosage levels. Overdosage will result in the signs of thyrotoxicosis listed above under precautions.
Dosage:
The initial recommended dose is 0.1mg/10lb (4.5kg) body weight twice daily. Dosage is then adjusted by monitoring the thyroid blood levels of the dog every four weeks until an adequate maintenance dose is established. The usual maintenance dose is 0.1mg/10lb (4.5kg) once daily.
Administration:
Levothyroxine sodium tablets may be administered orally or placed in the food.
Dosage forms available:
Levothyroxine sodium tablets are available as round, color-coded, potency marked tablets in bottles of 180 or 1000 as follows: 0.1mg - yellow, 0.2mg - pink, 0.3mg - green, 0.4mg - maroon, 0.5mg - white, 0.6mg - purple, 0.7mg - orange, 0.8mg - blue.
Storage
Store between 20°C-25°C (68°F-77°F) with excursions between 15°C-30°C (59°F-86°F)
References
1. Evinger, J.V., and Nelson, R.W. JAVMA 314:185, 314-316, 1984.
2. Richard Nelson, DVM. Current Veterinary Therapy X. Edition by R.W. Kirk, W.B. Saunders, Co., Philadelphia, PA, page 994, 1989.
3. Edward Feldman, DVM and Richard Nelson, DVM. Canine and Feline Endocrinology and Reproduction, W.B. Saunders, page 82, 1987.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN
Manufactured by LLOYD, Inc., Shenandoah, Iowa 51601
™ Trademark of Butler Animal Health Supply, LLC
Distributed Exclusively by: Butler Animal Health Supply, Dublin, OH 43017
Rev-00
NAC No.: 1082179.3
400 METRO PLACE NORTH, DUBLIN, OH, 43017-7545
| Telephone: | 614-761-9095 | |
| Toll-Free: | 1-855-724-3461 | |
| Website: | www.henryscheinvet.com |
 |
Every effort has been made to ensure the accuracy of the Thyroxine L Tablets information published above. However, it remains the responsibility of the readers to familiarize themselves with the product information contained on the US product label or package insert. |
Copyright © 2024 Animalytix LLC. Updated: 2024-02-27
