Revalor-200
This page contains information on Revalor-200 for veterinary use.The information provided typically includes the following:
- Revalor-200 Indications
- Warnings and cautions for Revalor-200
- Direction and dosage information for Revalor-200
Revalor-200
This treatment applies to the following species:(trenbolone acetate and estradiol implants)
For Growing Beef Steers and Heifers Fed In Confinement For Slaughter
Description
Revalor®-200 is an implant containing 200 mg of trenbolone acetate and 20 mg estradiol. Each implant consists of 10 small yellow pellets. Ten implants are provided in a cartridge.
Manufactured by a non-sterilizing process.
Revalor-200 Indications For Use
For increased rate of weight gain and improved feed efficiency in growing beef steers and heifers fed in confinement for slaughter.
Not approved for repeated implantation (re-implantation) with this or any other cattle ear implant in growing beef steers and heifers fed in confinement for slaughter. Safety and effectiveness following re-implantation have not been evaluated.
Do not use in beef calves less than 2 months of age, dairy calves, and veal calves because effectiveness and safety have not been established.
Do not use in animals intended for subsequent breeding, or in dairy cows.
NOTE:
Revalor®-200 is not better for increased average daily gain or improved feed efficiency than Revalor®-H (140 mg trenbolone acetate/14 mg estradiol) in heifers fed in confinement for slaughter.
Studies have demonstrated that the administration of Revalor®-200 can result in decreased marbling scores when compared to nonimplanted steers and heifers.
Directions
The cartridge is designed to be used with a special implanting tool. The special implanting tool is available from Intervet Inc. Ten doses (implants) are in each cartridge. The implant is placed under the skin on the posterior aspect of the ear (see SITE OF IMPLANTATION and METHOD OF USE sections).
Route Of Administration
The implant is placed under the skin on the posterior aspect of the ear by means of a special implanter available from Intervet Inc.
With the animal suitably restrained, the skin on the outer surface of the ear should be cleaned. The implant is then administered by the method shown in the diagram below.
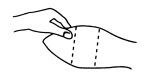
Fig. 1 - Ear of Bovine Ready for Implantation
Site Of Implantation
After appropriately restraining the animal to allow access to the ear, cleanse the skin at the implant needle puncture site. It is subcutaneous between the skin and cartilage on the back side of the ear and below the midline of the ear. The implant must not be placed closer to the head than the edge of the cartilage ring farthest from the head. The location of insertion of the needle is a point toward the tip of the ear and at least a needle length away from the intended deposition site. Care should be taken to avoid injuring the major blood vessels or cartilage of the ear.
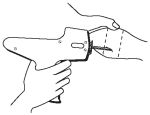
Fig. 2 - Rear View of the Bovine Ear Showing the Site for Insertion of the Implanter Needle.
METHOD OF USE
1. Do not remove the cap of the cartridge containing the implants.
2. Place the cartridge (D) (with the capped end to the front) into slot at the top of the implanter magazine (marked A on the diagram).
3. Gently push the cartridge into the slot until it clicks into place.
4. The implanter is then ready for use.
5. Take the ear of the animal firmly with the free hand (in the manner shown in Fig. 1). Then insert the needle into the subcutaneous tissue at the point indicated (in Fig. 2).
6. After inserting the needle to its full extent, squeeze the trigger (E) gradually. Allow the pellets of the implant to be deposited in a single row.
7. Withdraw the implanter. This will advance the cartridge one groove in the magazine and the next implant is now ready for use.
8. When all the implants have been administered, the cartridge will fall out of the bottom of the magazine and may be replaced by a new one.
9. To change the needle, loosen the needle locking nut (labeled F in Fig. 3) and replace the needle. Tighten the nut finger tight and the implanter is ready for use.
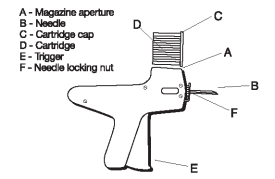
Fig. 3 - Diagram of the Implanter and Cartridge
 |
WITHDRAWAL PERIODS AND RESIDUE WARNINGS: No withdrawal period is required when used according to labeling. Do not use in beef calves less than 2 months of age, dairy calves, and veal calves. A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in dairy cows or in animals intended for subsequent breeding. Use in these cattle may cause drug residues in milk and/or calves born to these cows. Implant pellets subcutaneously in ear only. Any other location is a violation of Federal law. Do not attempt salvage of implanted site for human or animal food. |
 |
User Safety Warnings
Not for use in humans. Keep out of reach of children.
Storage Conditions
Store unopened product at or below 25°C (77°F). Avoid excessive heat and humidity. Use product before the expiration date printed on the carton labeling and on the cartridge pouch. Opened cartridges may be stored in the foil pouch protected from light in the refrigerator (2-8°C/36-47°F) for up to 6 months.
How Supplied
Box of 10 x 10 cartridges, each containing 10 implants per cartridge (100 implants total).
QUESTIONS/COMMENTS? For a copy of the Safety Data Sheet or to report side effects, contact Merck Animal Health Technical Services at 1-800-211-3573. For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae
Made in Austria by:
Intervet GesmbH
Distributed by:
Intervet Inc (d/b/a Merck Animal Health), Madison, NJ 07940
Copyright© Merck & Co., Inc., Rahway, NJ, USA and its affiliates. All rights reserved.
Restricted Drug (California) - use only as directed.
Approved by FDA under NADA # 140-992
Rev. 03/2023
167363 R4
CPN: 1047415.3
Intervet Inc.
126 E. LINCOLN AVENUE, PO BOX 2000, Rahway, NJ, 07065
| Customer Service: | 800-521-5767 | |
| Technical Service (Companion Animal): | 800-224-5318 | |
| Technical Service (Livestock): | 800-211-3573 | |
| Website: | www.merck-animal-health-usa.com |
 |
THIS SERVICE AND DATA ARE PROVIDED "AS IS". DVMetrics assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics service and data. See the Terms of Use for further details. |
Copyright © 2024 Animalytix LLC. Updated: 2024-02-27
