The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Ovaban Tablets (20 mg) (Canada)
This page contains information on Ovaban Tablets (20 mg) for veterinary use.The information provided typically includes the following:
- Ovaban Tablets (20 mg) Indications
- Warnings and cautions for Ovaban Tablets (20 mg)
- Direction and dosage information for Ovaban Tablets (20 mg)
Ovaban Tablets (20 Mg)
This treatment applies to the following species: Manufacturer: Schering-Plough
Manufacturer: Schering-Plough
DIN 00460818 (20 mg), 00460796 (5 mg)
Veterinary Use Only
For Use In Dogs Only
Pharmacological Classification
Oral contraceptive
Structural Formula And Chemistry
Megestrol acetate USP
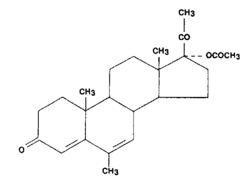
Mol. Formula
C24H32O4
Mol. Weight
384.5
Chemical Formula
17α-acetoxy-6-methylpregna-4, 6-diene-3,20-dione.
It is a tasteless, odourless, crystalline powder with a melting point of about 217°C, and is almost insoluble in water; soluble in 55 parts of alcohol (95 percent); in 0.8 parts of chloroform, and in 130 parts of solvent ether; slightly soluble in fixed oils.
Action
It is a potent oral progestogen with marked anti-estrogenic properties and more active in the Clauberg assay than progesterone given subcutaneously.1)
Megestrol acetate is rapidly metabolized. In dogs given the recommended oral dose for eight days the drug (as metabolites) was recovered from feces and urine. An average of 50% of the total dose was excreted by the last treatment day and 90% within an additional fifteen days.2)
The exact mechanism of action of megestrol acetate is not known. It is theorized that progestogens may act by one or more of the following methods:
1) inhibition of follicle stimulation hormone (FSH) release,
2) suppression of the growth of ovarian follicles and estrogen production,
3) inhibition of luteinizing hormone (LH) release and prevention of ovulation,
4) possession of a local anti-estrogenic effect which prevents vulvar swelling and vaginal discharge and,
5) affecting the corpus luteum by inhibition of the release of prolactin or Luteotrophic factors.
Current knowledge suggests that megestrol acetate may be most influential in the control of the LH surge.
Ovaban Tablets (20 mg) Indications And Clinical Uses
Ovaban is recommended for the postponement of estrus and the alleviation of false pregnancy in the dog.
Postponement Of Estrus
Treatment regimens for the postponement of estrus depends on the stage of the estrus cycle when therapy is initiated.Proestrus Treatment
An eight day course of Ovaban is recommended for dogs in proestrus. Vaginal bleeding and vulvar swelling will usually disappear in 3-8 days. The time until the next estrus occurs is dependent on timing of the dose and will vary with individual dogs. In clinical studies return to estrus ranged from 2 to 9 months with the majority recurring in 4 to 6 months.Treatment should be initiated during the first three days of proestrus (i.e., within three days of the onset of both vulvar swelling and vaginal discharge) for the best results.
Once therapy is started owners should confine their dogs for a few days, since dogs in proestrus may accept a male. Once bleeding stops, male dogs should not be attracted. The occasional bitch on Ovaban therapy that accepts a male will not become pregnant, and therapy should be continued to completion of the regimen.
Anestrus Treatment
A thirty-two day course of Ovaban therapy is recommended for dogs in anestrus. The dog will remain in anestrus as long as therapy is continued. The next estrus may occur any time after cessation of treatment. In clinical studies there was a range of 2-9 months though most dogs returned to estrus in 4-6 months.Alleviation Of False Pregnancy
Ovaban can be administered to alleviate the physical and nervous signs of false pregnancy. Remission of signs will usually occur in 3 to 8 days. For the best results treatment should be initiated at the first signs of false pregnancy.Contraindications
Ovaban must not be used in dogs when there is evidence of disease of reproductive organs.
Ovaban is less effective if used in dogs during their first estrus cycle and should not be used.
Ovaban must not be used in pregnant dogs. It should be established that there was not exposure to a male dog during that previous heat period.
Ovaban should not be used in diabetic dogs.
Ovaban must not be used in dogs with mammary tumors. Growth of some mammary tumors may be stimulated by exogenous progestogens.
Precautions
There are no data on conception of bitches mated less than 2 months after anestrus treatment; therefore, breeding of such animals is not recommended.
Toxicology
Megestrol acetate at 0.1 to 0.25 mg/kg/day orally in dogs for 36 months did not produce any gross abnormalities. Histologically, cystic endometrial hyperplasia was observed at 36 months. These uterine changes were reversed when dosing ceased and the dogs were observed after an additional 15 months.
No uterine abnormalities were observed in one dog receiving 0.5 mg/kg/day for 5 months; pyometra was produced after an additional 4 months dosing. A second dog showed hyperplasia after 5 months dosing; however, after 2 months without treatment, the uterus returned to normal.
Three dogs were treated with 2 mg/kg/day and uterine biopsies were taken after 32 and 64 days of continuous treatment. The uteri were all normal at 32 days; at 64 days, there were 2 normal uteri, and the third showed early signs of endometritis. Other abnormalities were not observed.
Cox3) compared several progestogens in dogs and determined megestrol acetate did not produce cystic endometrial hyperplasia at the recommended doses.
Burke4) reported studies in dogs that did not show any effects on conception, litter size, mortality, or sex ratios following megestrol acetate at the recommended dosage for 32 days.
Megestrol acetate at 0.25 mg/lb for 32 days did not affect the dam or litter when administered during the first half of pregnancy; however, reduced litter size and livability resulted when the dose was given during the last half of pregnancy.
Ovaban Tablets (20 mg) Dosage And Administration
Ovaban Tablets are palatable and may be given intact or crushed and mixed in the food.
Postponement Of Estrus
Proestrus Treatment
Administer 1 mg per pound of body weight per day for eight days.Anestrus Treatment
Administer 0.25 mg per pound of body weight per day for thirty-two days.Alleviation Of False Pregnancy
Administer 1 mg per pound of body weight per day for eight days.Side Effects
Oral doses of megestrol acetate are well tolerated. Occasionally, the following transient progestational side effects were noted in clinical studies: mammary enlargement, lactation, listlessness, increased appetite, and temperament change. In clinical studies, confirmed cases of pyometra occurred in 0.6% of the cases.
The incidence of polyuria/polydipsia and post-treatment false pregnancy were occasionally reported.
How Supplied
Ovaban Tablets, 5 mg and 20 mg, are available in bottles of 200 tablets each.
Bibliography
1. David, A., et al: Anti-ovulatory and other biological properties of megestrol acetate. Jour. Reproductive Fertility (1963) 5, p. 331-346.
2. Evans, J.M., et al: The excretion of me estrol acetate by beagle bitches. Vet. Rec., March 7, 1970.
3. Cox, J.E., Progestogens in bitches: a review, Jour. Small Animal Pract. (1970) 11, p. 759-778.
4. Burke, T.J., et al: Megestrol Acetate for Estrus Postponement in the Bitch. Journal of the American Veterinary Medical Association (1975) Vol. 167, No. 4, Pages 285-287.
5. Bigbee, H.G., DVM, Hennessey, P.W., BS, Megestrol Acetate for Postponing Estrus in FirstHeat Bitches, Veterinary Medicine/Small Animal Clinician, Nov. 1977 Pages 1727-30.
© Copyright Schering Canada Inc. Assignment of original copyright by Schering Corporation.
Schering-Plough Animal Health, A division of SCHERING CANADA INC., Pointe Claire, Québec
H9R 1B4
* Reg. T.M. of Schering Canada Inc.
71-465737 E
Nac No.
12080740Division of Schering Canada Inc.
3535 TRANS CANADA HWY., POINTE CLAIRE, QC, H9R 1B4
| Telephone: | 800-605-2584 | |
| Telephone Orders: | ||
| Quebec: | FRENCH: 800-361-2431 or 514-426-7340 | |
| ENGLISH: 800-361-6550 or 514-426-7344 | ||
| Atlantic Provinces & Ontario: | 800-361-6550 | |
| Fax Orders: | 888-428-7400 or 514-428-7400 | |
| Telephone Orders: | ||
| Manitoba/Saskatchewan/Alberta/British Columbia: | 800-661-3134 or 403-236-7363 | |
| Fax Orders: | 403-236-5196 |
 |
Every effort has been made to ensure the accuracy of the Ovaban Tablets (20 mg) information published above. However, it remains the responsibility of the readers to familiarize themselves with the product information contained on the Ovaban Tablets (20 mg) product label or package insert. |

Copyright © 2024 Animalytix LLC. Updated: 2024-08-26
