The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
Bronchi-Shield III (Canada)
This page contains information on Bronchi-Shield III for veterinary use.The information provided typically includes the following:
- Bronchi-Shield III Indications
- Warnings and cautions for Bronchi-Shield III
- Direction and dosage information for Bronchi-Shield III
Bronchi-Shield III
This treatment applies to the following species:Canine Adenovirus Type 2-Parainfluenza-Bordetella Bronchiseptica Vaccine, Modified Live Virus & Avirulent Live Culture
Veterinary Use Only
Bronchi-Shield III Indications
For the vaccination of healthy dogs and puppies eight weeks of age or older as an aid in prevention of diseases caused by Bordetella bronchiseptica, canine parainfluenza virus and canine adenovirus type 2.
Preparation of the Vaccine: Rehydrate with the accompanying sterile diluent. Shake well and draw back into the syringe the required amount. Remove needle from syringe and attach enclosed applicator tip. Use immediately.
Administration and Dosage: Instill 0.5 mL of rehydrated vaccine into each nostril using a syringe with applicator tip. Annual revaccination is recommended.
The PEEL-OFF LABEL I.D. SYSTEM provides a simple and effective method of recording pertinent information on the vaccines administered to animals in a veterinary practice.
For vaccines requiring reconstitution, remove label from both vials and affix both labels to the animal’s medical chart.
Using the System:
|
1. |
Grasp the vial label at the corner marked with an indicator arrow, between your thumb and forefinger (figure 1). |
 |
|
2. |
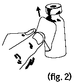
Pull steadily at a slight angle until the label is separated from the vial (figure 2). |
 |
|
3. |
Place the label on the animal’s medical chart. Press down on the label to ensure adhesion. |
|
Precautions
This product is designed for intranasal use only. Do not vaccinate dogs parenterally. A very small percentage of animals may show sneezing, coughing or nasal discharge following vaccination. These signs are usually transient. Store in the dark at 2-7°C. Do not freeze. Shake well after rehydration. Burn container and all unused contents. In case of anaphylactoid reaction, administer epinephrine.Bronchi-Shield® is a registered trademark of Boehringer Ingelheim Vetmedica, Inc. Used under license.
Manufactured by: Boehringer Ingelheim Vetmedica, Inc., St. Joseph, Missouri 64506 U.S.A.
U.S. Veterinary License No. 124
Manufactured for: Boehringer Ingelheim (Canada) Ltd., Burlington, Ontario L7L 5H4
|
25 Doses |
This package contains 25 x 1 mL vials of vaccine plus 25 x 1 mL vials of diluent. |
150206-00 |
CPN: 12300701
5180 SOUTH SERVICE ROAD, BURLINGTON, ON, L7L 5H4
| Customer Care No.: | 1-800-567-1885 | |
| Technical Services No.: | 1-877-565-5501 | |
| Website: | www.boehringer-ingelheim.ca |
 |
This service and data are provided "AS IS". DVMetrics and Drugs.com assumes no liability, and each user assumes full risk, responsibility, and liability, related to its use of the DVMetrics and Drugs.com services and data. See the terms of use for further details. |

Copyright © 2024 Animalytix LLC. Updated: 2024-02-27
