Vitrasert Prescribing Information
Package insert / product label
Generic name: ganciclovir
Dosage form: implant
Drug class: Ophthalmic anti-infectives
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
The Vitrasert brand name has been discontinued in the U.S. If generic versions of this product have been approved by the FDA, there may be generic equivalents available.
On This Page
Vitrasert Description
The Vitrasert Implant contains the antiviral drug ganciclovir. Each Vitrasert Implant contains a minimum of 4.5 mg ganciclovir.
Each Vitrasert Implant contains a ganciclovir tablet which contains the inactive ingredient, magnesium stearate (0.25%). Each tablet is coated with polyvinyl alcohol and ethylene vinyl acetate polymers.
The chemical name of ganciclovir is 9-[[2-hydroxy -1- (hydroxymethyl)ethoxy] methyl]guanine, and has the following structure:
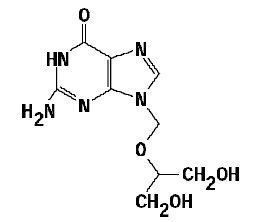
Ganciclovir is a white to off-white crystalline powder with a molecular formula of C 9 H 13 N 5 O 4 and molecular weight of 255.23. Ganciclovir has a solubility of 4.3 mg/mL in water at 25°C.
Vitrasert - Clinical Pharmacology
Virology
Ganciclovir is a synthetic nucleoside analogue of 2'-deoxyguanosine that inhibits replication of herpes viruses both in vitro and in vivo. Sensitive human viruses include cytomegalovirus (CMV), herpes simplex virus-1 and -2 (HSV-1, HSV-2), Epstein-Barr virus (EBV) and varicella zoster virus (VZV). Clinical studies have been limited to assessment of efficacy in patients with CMV infection.
Median effective inhibitory doses (ED 50 ) of ganciclovir for human CMV isolates tested in vitro in several cell lines ranged from 0.2 to 3.0 µg/mL. The relationship between in vitro sensitivity of CMV to ganciclovir and clinical response has not been established. Ganciclovir inhibits mammalian cell proliferation in vitro at higher concentrations (10 to 60 µg/mL) with bone marrow colony forming cells being the most sensitive (ID 50 >/=10 µg/mL) of those cell types tested.
Emergence of viral resistance has been reported based on in vitro sensitivity testing of CMV isolates from patients receiving intravenous ganciclovir treatment. The prevalence of resistant isolates is unknown, and there is a possibility that some patients may be infected with strains of CMV resistant to ganciclovir. Therefore, the possibility of viral resistance should be considered in patients who show poor clinical response.
Pharmacokinetics
In a clinical trial of Vitrasert Implants, 26 patients (30 eyes) received a total of 39 primary implants and 12 exchange implants (performed 32 weeks after the implant was inserted or earlier if progression of CMV retinitis occurred). Because most of the exchanged implants were empty, the time the implant actually ran out of drug was unknown, and a precise in-vivo release rate could not be calculated. However, approximate in-vivo release rates could be determined for the exchanged implants, which ranged from 1.00 µg/h to more than 1.62 µg/h.
In 14 implants (3 exchanged, 11 autopsy) in which the in-vivo release rate could accurately be calculated, the mean release rate was 1.40 µg/h, with a range from 0.5 to 2.88 µg/h. The mean vitreous drug levels in eight eyes (4 collected at the time of retinal detachment surgery; 2 collected from autopsy eyes within 6 hours of death and prior to fixation; 2 collected from implant exchanges) was 4.1 µg/mL.
Indications and Usage for Vitrasert
The Vitrasert Implant is indicated for the treatment of CMV retinitis in patients with acquired immunodeficiency syndrome (AIDS).
The diagnosis of CMV retinitis is opthalmologic and should be made by indirect ophthalmoscopy. Other conditions in the differential diagnosis of CMV retinitis include candidiasis, toxoplasmosis, histoplasmosis, retinal scars, and cotton wool spots, any of which may produce a retinal appearance similar to CMV. For this reason, it is essential that the diagnosis of CMV be established by a physician familiar with the retinal presentation of these conditions.
The Vitrasert Implant is for intravitreal implantation only.
Clinical Trials
In a randomized, controlled parallel group trial conducted between May 1993 and December 1994, treatment with the Vitrasert Implant was compared to treatment with intravenous ganciclovir (Cytovene-IV; Roche) in 188 patients with AIDS and newly diagnosed CMV retinitis. Patients randomized to the Cytovene-IV treatment group received Cytovene-IV solution at induction doses (5 mg/kg twice daily) for 14 days, followed by maintenance dosing (5 mg/kg once daily). Based on masked assessment of fundus photographs, the median time to progression was approximately 210 days for the Vitrasert Implant treatment group compared to approximately 120 days for the intravenous ganciclovir treatment group.
Contraindications
The Vitrasert Implant is contraindicated in patients with hypersensitivity to ganciclovir or acyclovir, and in patients with any contraindications for intraocular surgery, such as external infection or severe thrombocytopenia.
Warnings
CMV retinitis may be associated with CMV disease elsewhere in the body. The Vitrasert Implant provides localized therapy limited to the implanted eye. The Vitrasert Implant does not provide treatment for systemic CMV disease. Patients should be monitored for extraocular CMV disease.
As with any surgical procedure, there is risk involved. Potential complications accompanying intraocular surgery to place the Vitrasert Implant into the vitreous cavity may include, but are not limited to, the following: vitreous loss, vitreous hemorrhage, cataract formation, retinal detachment, uveitis, endophthalmitis, and decrease in visual acuity.
Following implantation of the Vitrasert Implant, nearly all patients will experience an immediate and temporary decrease in visual acuity in the implanted eye which lasts for approximately two to four weeks post-operatively. This decrease in visual acuity is likely a result of the surgical implant procedure.
Precautions
General
As with all intraocular surgery, sterility of the surgical field and the Vitrasert Implant should be rigorously maintained. The Vitrasert Implant should be handled only by the suture tab in order to avoid damaging the polymer coatings since this could affect release rate of ganciclovir inside the eye. The Vitrasert Implant should not be resterilized by any method.
A high level of surgical skill is required for implantation of the Vitrasert Implant. A surgeon should have observed or assisted in surgical implantation of the Vitrasert Implant prior to attempting the procedure.
Information for patients
The Vitrasert Implant is not a cure for CMV retinitis, and some immunocompromised patients may continue to experience progression of retinitis with the Vitrasert Implant. Patients should be advised to have ophthalmologic follow-up examinations of both eyes at appropriate intervals following implantation of the Vitrasert Implant.
As with any surgical procedure, there is risk involved. Potential complications accompanying intraocular surgery to place the Vitrasert Implant into the vitreous cavity may include, but are not limited to, the following: intraocular infection or inflammation, detachment of the retina, and formation of cataract in the natural crystalline lens.
Following implantation of the Vitrasert Implant, nearly all patients will experience an immediate and temporary decrease in visual acuity in the implanted eye which lasts for approximately two to four weeks post-operatively. This decrease in visual acuity is likely a result of the surgical implant procedure.
The Vitrasert Implant only treats eyes in which it has been implanted. Additionally, because CMV is a systemic disease, patients should be monitored for extraocular CMV infections (e.g., pneumonitis, colitis) in the body.
Patients should be advised that ganciclovir has caused decreased sperm production in animals and may cause infertility in humans. Women of childbearing potential should be advised that ganciclovir causes birth defects in animals and should not be used during pregnancy.
Patients should be advised that ganciclovir has caused tumors in animals. Although there is no information from human studies, ganciclovir should be considered a potential carcinogen.
Drug interactions
No drug interactions have been observed with the Vitrasert Implant. There is limited experience with use of retinal tamponades in conjunction with the Vitrasert Implant.
Carcinogenesis, mutagenesis, impairment of fertility
Ganciclovir was carcinogenic in the mouse at oral doses of 20 and 1000 mg/kg/day. At the dose of 1000 mg/kg/day there was a significant increase in the incidence of tumors of the preputial gland in males, forestomach (nonglandular mucosa) in males and females, and reproductive tissues (ovaries, uterus, mammary gland, clitoral gland, and vagina) and liver in females. At the dose of 20 mg/kg/day, a slightly increased incidence of tumors was noted in the preputial and harderian glands in males, forestomach in males and females, and liver in females. Except for histiocytic sarcoma of the liver, ganciclovir-induced tumors were generally of epithelial or vascular origin. Although the preputial and clitoral glands, forestomach, and harderian glands of mice do not have human counterparts, ganciclovir should be considered a potential carcinogen in humans.
Ganciclovir increased mutations in mouse lymphoma cells and DNA damage in human lymphocytes in vitro at concentrations between 50-500 and 250-2000 µg/mL, respectively. In the mouse micronucleus assay, ganciclovir was clastogenic at doses of 150 and 500 mg/kg (IV) (2.8 - 10x human exposure based on AUC) but not 50 mg/kg (exposure approximately comparable to the human based on AUC). Ganciclovir was not mutagenic in the Ames Salmonella assay at concentrations of 500-5000 µg/mL.
Ganciclovir caused decreased mating behavior, decreased fertility, and an increased incidence of embryolethality in female mice following intravenous doses of 90 mg/kg/day. Ganciclovir caused decreased fertility in male mice and hypospermatogenesis in mice and dogs following daily oral or intravenous administration of doses ranging from 0.2-10 mg/kg.
Pregnancy
Teratogenic effects
Pregnancy Category C. Ganciclovir has been shown to be embryotoxic in rabbits and mice following intravenous administration and teratogenic in rabbits. Fetal resorptions were present in at least 85% of rabbits and mice administered 60 mg/kg/day and 108 mg/kg/day, respectively. Effects observed in rabbits included: fetal growth retardation, embryolethality, teratogenicity, and/or maternal toxicity. Teratogenic changes included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly, and brachygnathia. In mice, effects observed were maternal/fetal toxicity and embryolethality.
Daily intravenous doses of 90 mg/kg administered to female mice prior to mating, during gestation, and during lactation caused hypoplasia of the testes and seminal vesicles in the month-old male offspring, as well as pathologic changes in the nonglandular region of the stomach (see Carcinogenesis, Mutagenesis subsection).
Although each Vitrasert Implant contains from 4.5 to 6.4 mg of ganciclovir, which is released locally in the vitreous, there are no adequate and well-controlled studies in pregnant women on the effects of the Vitrasert Implant. Therefore, the Vitrasert Implant should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing mothers
It is not known whether ganciclovir from the Vitrasert Implant is excreted in human milk. Daily intravenous doses of 90 mg/kg administered to female mice prior to mating, during gestation, and during lactation caused hypoplasia of the testes and seminal vesicles in the month-old male offspring, as well as pathologic changes in the non-glandular region of the stomach. Because many drugs are excreted in human milk and, because carcinogenicity and teratogenicity effects occurred in animals treated with ganciclovir, mothers should be instructed to discontinue nursing if they have a Vitrasert Implant.
Adverse Reactions/Side Effects
During clinical trials, the most frequent adverse events seen in patients treated with the Vitrasert Implant involved the eye.
During the first two months following implantation, visual acuity loss of 3 lines or more, vitreous hemorrhage, and retinal detachments occurred in approximately 10-20% of patients. Cataract formation/lens opacities, macular abnormalities, intraocular pressure spikes, optic disk/nerve changes, hyphemas and uveitis occurred in approximately 1-5%.
Adverse events with an incidence of less than 1% were: retinopathy, anterior chamber cell and flare, synechia, hemorrhage (other than vitreous), cotton wool spots, keratopathy, astigmatism, endophthalmitis, microangiopathy, sclerosis, choroiditis, chemosis, phthisis bulbi, angle closure glaucoma with anterior chamber shallowing, vitreous detachment, vitreous traction, hypotony, severe post-operative inflammation, retinal tear, retinal hole, corneal dellen, choroidal folds, pellet extrusion from scleral wound, and gliosis.
Vitrasert Dosage and Administration
Each Vitrasert Implant contains a minimum of 4.5 mg of ganciclovir, and is designed to release the drug over a 5 to 8 month period of time. Following depletion of ganciclovir from the Vitrasert Implant, as evidenced by progression of retinitis, the Vitrasert Implant may be removed and replaced.
Handling and Disposal
Caution should be exercised in handling of the Vitrasert Implant in order to avoid damage to the polymer coating on the implant, which may result in an increased rate of drug release from the implant. Thus, the Vitrasert Implant should be handled only by the suture tab. Aseptic technique should be maintained at all times prior to and during the surgical implantation procedure.
Because the Vitrasert Implant contains ganciclovir, which shares some of the properties of anti-tumor agents (i.e., carcinogenicity and mutagenicity), consideration should be given to handling and disposal of the Vitrasert Implant according to guidelines issued for antineoplastic drugs.
How is Vitrasert supplied
The Vitrasert Implant is supplied in individual unit boxes in a sterile Tyvek package (NDC 24208-412-01).
Storage
Store at room temperature, 15-30°C (59-86°F). Protect from freezing, excessive heat and light.
FOR INTRAVITREAL IMPLANTATION ONLY
Rx Only
Manufacturer Information
Revised August 2005
Manufactured for:
Bausch & Lomb, Incorporated
Rochester, NY 14609 USA
800/338-2020
Manufactured by:
AMP, Inc.
Irvine, CA 92618 USA
Vitrasert is a registered trademark of Bausch & Lomb Incorporated.
©Copyright 2003 Bausch & Lomb Incorporated. All rights reserved.
U.S. Patent #5,378,475. Foreign Patents pending.
U.S. Patent #4,355,032, #4,507,305 (ganciclovir compound).
Cytovene is a registered trademark of Roche Laboratories. A member of the Roche Group.
| VITRASERT
ganciclovir implant |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Bausch & Lomb Incorporated (196603781) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Alliance Medical Products | 102688657 | MANUFACTURE | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Roche Colorado | 076470525 | API MANUFACTURE, ANALYSIS | |
More about Vitrasert (ganciclovir ophthalmic)
- Compare alternatives
- Side effects
- Dosage information
- During pregnancy
- Drug class: ophthalmic anti-infectives
- Breastfeeding

