Parcopa Prescribing Information
Package insert / product label
Generic name: carbidopa and levodopa
Dosage form: tablet, orally disintegrating
Drug class: Dopaminergic antiparkinsonism agents
Medically reviewed by Drugs.com. Last updated on Mar 25, 2024.
On This Page
Parcopa Description
PARCOPA® (carbidopa and levodopa orally disintegrating tablets) is a combination of carbidopa and levodopa for the treatment of Parkinson’s disease and syndrome. PARCOPA® is an orally administered formulation of carbidopa and levodopa which rapidly disintegrates on the tongue and does not require water to aid dissolution or swallowing.
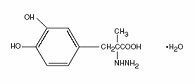
Carbidopa, an inhibitor of aromatic amino acid decarboxylation, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 244.24. It is designated chemically as (–)-L-α-hydrazino-α-methyl-ß-(3,4-dihydroxybenzene) propanoic acid monohydrate. Its empirical formula is C10H14N2O4∙H2O, and its structural formula is:
Tablet content is expressed in terms of anhydrous carbidopa which has a molecular weight of 226.23.
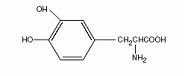
Levodopa, an aromatic amino acid, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 197.2. It is designated chemically as (–)-L-α-amino-ß-(3,4-dihydroxybenzene) propanoic acid. Its empirical formula is C9H11NO4, and its structural formula is:
PARCOPA® is supplied as tablets in three strengths:
PARCOPA® 25/100, containing 25 mg of carbidopa and 100 mg of levodopa.
PARCOPA® 10/100, containing 10 mg of carbidopa and 100 mg of levodopa.
PARCOPA® 25/250, containing 25 mg of carbidopa and 250 mg of levodopa.
Inactive ingredients are aspartame, citric acid, crospovidone, magnesium stearate, mannitol, microcrystalline cellulose, natural and artificial mint flavor and sodium bicarbonate. PARCOPA® 10/100 and 25/250 also contain FD&C blue #2 HT aluminum lake. PARCOPA® 25/100 also contains yellow 10 iron oxide.
Parcopa - Clinical Pharmacology
Parkinson’s disease is a progressive, neurodegenerative disorder of the extrapyramidal nervous system affecting the mobility and control of the skeletal muscular system. Its characteristic features include resting tremor, rigidity, and bradykinetic movements. Symptomatic treatments, such as levodopa therapies, may permit the patient better mobility.
Mechanism of Action
Current evidence indicates that symptoms of Parkinson’s disease are related to depletion of dopamine in the corpus striatum. Administration of dopamine is ineffective in the treatment of Parkinson’s disease apparently because it does not cross the blood-brain barrier. However, levodopa, the metabolic precursor of dopamine, does cross the blood-brain barrier, and presumably is converted to dopamine in the brain. This is thought to be the mechanism whereby levodopa relieves symptoms of Parkinson’s disease.
Pharmacodynamics
When levodopa is administered orally it is rapidly decarboxylated to dopamine in extracerebral tissues so that only a small portion of a given dose is transported unchanged to the central nervous system. For this reason, large doses of levodopa are required for adequate therapeutic effect and these may often be accompanied by nausea and other adverse reactions, some of which are attributable to dopamine formed in extracerebral tissues.
Since levodopa competes with certain amino acids for transport across the gut wall, the absorption of levodopa may be impaired in some patients on a high protein diet.
Carbidopa inhibits decarboxylation of peripheral levodopa. It does not cross the blood-brain barrier and does not affect the metabolism of levodopa within the central nervous system.
The incidence of levodopa-induced nausea and vomiting is less with carbidopa and levodopa than with levodopa. In many patients, this reduction in nausea and vomiting will permit more rapid dosage titration.
Since its decarboxylase inhibiting activity is limited to extracerebral tissues, administration of carbidopa with levodopa makes more levodopa available for transport to the brain.
Pharmacokinetics
Carbidopa reduces the amount of levodopa required to produce a given response by about 75 percent and, when administered with levodopa, increases both plasma levels and the plasma half-life of levodopa, and decreases plasma and urinary dopamine and homovanillic acid.
The plasma half-life of levodopa is about 50 minutes, without carbidopa. When carbidopa and levodopa are administered together, the half-life of levodopa is increased to about 1.5 hours. At steady state, the bioavailability of carbidopa from carbidopa and levodopa tablets is approximately 99% relative to the concomitant administration of carbidopa and levodopa.
In clinical pharmacologic studies, simultaneous administration of carbidopa and levodopa produced greater urinary excretion of levodopa in proportion to the excretion of dopamine than administration of the two drugs at separate times.
Pyridoxine hydrochloride (vitamin B6), in oral doses of 10 mg to 25 mg, may reverse the effects of levodopa by increasing the rate of aromatic amino acid decarboxylation. Carbidopa inhibits this action of pyridoxine; therefore, PARCOPA® can be given to patients receiving supplemental pyridoxine (vitamin B6).
Indications and Usage for Parcopa
PARCOPA® is indicated in the treatment of the symptoms of idiopathic Parkinson’s disease (paralysis agitans), postencephalitic parkinsonism, and symptomatic parkinsonism which may follow injury to the nervous system by carbon monoxide intoxication and/or manganese intoxication. PARCOPA® is indicated in these conditions to permit the administration of lower doses of levodopa with reduced nausea and vomiting, with more rapid dosage titration, with a somewhat smoother response, and with supplemental pyridoxine (vitamin B6).
In some patients, a somewhat smoother antiparkinsonian effect results from therapy with carbidopa and levodopa than with levodopa. However, patients with markedly irregular (“on-off”) responses to levodopa have not been shown to benefit from carbidopa and levodopa therapy.
Although the administration of carbidopa permits control of parkinsonism and Parkinson’s disease with much lower doses of levodopa, there is no conclusive evidence at present that this is beneficial other than in reducing nausea and vomiting, permitting more rapid titration, and providing a somewhat smoother response to levodopa.
Certain patients who responded poorly to levodopa have improved when carbidopa and levodopa was substituted. This is most likely due to decreased peripheral decarboxylation of levodopa which results from administration of carbidopa rather than to a primary effect of carbidopa on the nervous system. Carbidopa has not been shown to enhance the intrinsic efficacy of levodopa in parkinsonian syndromes.
In considering whether to give PARCOPA® to patients already on levodopa who have nausea and/or vomiting, the practitioner should be aware that, while many patients may be expected to improve, some do not. Since one cannot predict which patients are likely to improve, this can only be determined by a trial of therapy. It should be further noted that in controlled trials comparing carbidopa and levodopa with levodopa, about half of the patients with nausea and/or vomiting on levodopa improved spontaneously despite being retained on the same dose of levodopa during the controlled portion of the trial.
Contraindications
Nonselective monoamine oxidase (MAO) inhibitors are contraindicated for use with PARCOPA®. These inhibitors must be discontinued at least two weeks prior to initiating therapy with PARCOPA®. PARCOPA® may be administered concomitantly with the manufacturer’s recommended dose of an MAO inhibitor with selectivity for MAO type B (e.g., selegiline HCl) (See PRECAUTIONS, Drug Interactions).
PARCOPA® is contraindicated in patients with known hypersensitivity to any component of this drug, and in patients with narrow-angle glaucoma.
Because levodopa may activate a malignant melanoma, PARCOPA® should not be used in patients with suspicious, undiagnosed skin lesions or a history of melanoma.
Warnings
When PARCOPA® (carbidopa and levodopa orally disintegrating tablets) is to be given to patients who are being treated with levodopa, levodopa must be discontinued at least twelve hours before therapy with PARCOPA® (carbidopa and levodopa orally disintegrating tablets) is started. In order to reduce adverse reactions, it is necessary to individualize therapy. See DOSAGE AND ADMINISTRATION section before initiating therapy.
The addition of carbidopa with levodopa in the form of PARCOPA® reduces the peripheral effects (nausea, vomiting) due to decarboxylation of levodopa; however, carbidopa does not decrease the adverse reactions due to the central effects of levodopa. Because carbidopa permits more levodopa to reach the brain and more dopamine to be formed, certain adverse CNS effects, e.g., dyskinesias (involuntary movements), may occur at lower dosages and sooner with PARCOPA® than with levodopa alone.
Levodopa alone, as well as PARCOPA®, is associated with dyskinesias. The occurrence of dyskinesias may require dosage reduction.
As with levodopa, PARCOPA® may cause mental disturbances. These reactions are thought to be due to increased brain dopamine following administration of levodopa. All patients should be observed carefully for the development of depression with concomitant suicidal tendencies. Patients with past or current psychoses should be treated with caution.
PARCOPA® should be administered cautiously to patients with severe cardiovascular or pulmonary disease, bronchial asthma, renal, hepatic or endocrine disease.
As with levodopa, care should be exercised in administering PARCOPA® to patients with a history of myocardial infarction who have residual atrial, nodal, or ventricular arrhythmias. In such patients, cardiac function should be monitored with particular care during the period of initial dosage adjustment, in a facility with provisions for intensive cardiac care.
As with levodopa, treatment with PARCOPA® may increase the possibility of upper gastrointestinal hemorrhage in patients with a history of peptic ulcer.
Neuroleptic Malignant Syndrome (NMS)
Sporadic cases of a symptom complex resembling NMS have been reported in association with dose reductions or withdrawal of therapy with carbidopa and levodopa. Therefore, patients should be observed carefully when the dosage of PARCOPA® is reduced abruptly or discontinued, especially if the patient is receiving neuroleptics.
NMS is an uncommon but life-threatening syndrome characterized by fever or hyperthermia. Neurological findings, including muscle rigidity, involuntary movements, altered consciousness, mental status changes; other disturbances, such as autonomic dysfunction, tachycardia, tachypnea, sweating, hyper- or hypotension; laboratory findings, such as creatine phosphokinase elevation, leukocytosis, myoglobinuria, and increased serum myoglobin have been reported.
The early diagnosis of this condition is important for the appropriate management of these patients. Considering NMS as a possible diagnosis and ruling out other acute illnesses (e.g., pneumonia, systemic infection, etc.) is essential. This may be especially complex if the clinical presentation includes both serious medical illness and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system (CNS) pathology.
The management of NMS should include: 1) intensive symptomatic treatment and medical monitoring and 2) treatment of any concomitant serious medical problems for which specific treatments are available. Dopamine agonists, such as bromocriptine, and muscle relaxants, such as dantrolene, are often used in the treatment of NMS, however, their effectiveness has not been demonstrated in controlled studies.
Precautions
General
As with levodopa, periodic evaluations of hepatic, hematopoietic, cardiovascular, and renal function are recommended during extended therapy.
Patients with chronic wide-angle glaucoma may be treated cautiously with PARCOPA® provided the intraocular pressure is well controlled and the patient is monitored carefully for changes in intraocular pressure during therapy.
Dopaminergic agents, including levodopa, may be associated with somnolence and very rarely episodes of sudden onset of sleep. In some cases, these episodes may occur without awareness or warning during daily activities. Patients must be informed of this and advised to exercise caution while driving or operating machines while being treated with dopaminergic agents, including levodopa. Patients who have experienced somnolence and/or an episode of sudden sleep onset must refrain from driving or operating machines (see Information for Patients).
Melanoma
Epidemiological studies have shown that patients with Parkinson’s disease have a higher risk (2- to approximately 6-fold higher) of developing melanoma than the general population. Whether the increased risk observed was due to Parkinson’s disease or other factors, such as drugs used to treat Parkinson’s disease, is unclear.
For the reasons stated above, patients and providers are advised to monitor for melanomas frequently and on a regular basis when using PARCOPA® for any indication. Ideally, periodic skin examinations should be performed by appropriately qualified individuals (e.g., dermatologists).
Information for Patients
Phenylketonurics
Phenylketonuric patients should be informed that PARCOPA® contains phenylalanine 3.4 mg per 25/100 orally disintegrating tablet, 3.4 mg per 10/100 orally disintegrating tablet, and 8.4 mg per 25/250 orally disintegrating tablet.
Patients should be instructed not to remove PARCOPA® Tablets from the bottle until just prior to dosing. With dry hands, the tablet should be gently removed and immediately placed on the tongue to dissolve and be swallowed with the saliva.
The patient should be informed that PARCOPA® is an immediate-release formulation of carbidopa and levodopa that is designed to begin release of ingredients within 30 minutes. It is important that PARCOPA® be taken at regular intervals according to the schedule outlined by the physician. The patient should be cautioned not to change the prescribed dosage regimen and not to add any additional antiparkinson medications, including other carbidopa and levodopa preparations, without first consulting the physician.
Patients should be advised that sometimes a “wearing-off” effect may occur at the end of the dosing interval. The physician should be notified if such response poses a problem to lifestyle.
Patients should be advised that occasionally, dark color (red, brown, or black) may appear in saliva, urine, or sweat after ingestion of PARCOPA®. Although the color appears to be clinically insignificant, garments may become discolored.
The patient should be advised that a change in diet to foods that are high in protein may delay the absorption of levodopa and may reduce the amount taken up in the circulation. Excessive acidity also delays stomach emptying, thus delaying the absorption of levodopa. Iron salts (such as in multi-vitamin tablets) may also reduce the amount of levodopa available to the body. The above factors may reduce the clinical effectiveness of the levodopa or carbidopa and levodopa therapy.
Patients should be alerted to the possibility of sudden onset of sleep during daily activities, in some cases without awareness or warning signs, when they are taking dopaminergic agents, including levodopa. Patients should be advised to exercise caution while driving or operating machinery and that if they have experienced somnolence and/or sudden sleep onset, they must refrain from these activities. (See PRECAUTIONS: General.)
There have been reports of patients experiencing intense urges to gamble, increased sexual urges, and other intense urges, and the inability to control these urges while taking one or more of the medications that increase central dopaminergic tone and that are generally used for the treatment of Parkinson’s disease, including PARCOPA®. Although it is not proven that the medications caused these events, these urges were reported to have stopped in some cases when the dose was reduced or the medication was stopped. Prescribers should ask patients about the development of new or increased gambling urges, sexual urges or other urges while being treated with PARCOPA®. Patients should inform their physician if they experience new or increased gambling urges, increased sexual urges, or other intense urges while taking PARCOPA®. Physicians should consider dose reduction or stopping the medication if a patient develops such urges while taking PARCOPA®.
Laboratory Tests
Abnormalities in laboratory tests may include elevations of liver function tests such as alkaline phosphatase, SGOT (AST), SGPT (ALT), lactic dehydrogenase, and bilirubin. Abnormalities in blood urea nitrogen and positive Coombs test have also been reported. Commonly, levels of blood urea nitrogen, creatinine, and uric acid are lower during administration of carbidopa and levodopa than with levodopa.
Carbidopa and levodopa may cause a false-positive reaction for urinary ketone bodies when a test tape is used for determination of ketonuria. This reaction will not be altered by boiling the urine specimen. False-negative tests may result with the use of glucose-oxidase methods of testing for glucosuria.
Cases of falsely diagnosed pheochromocytoma in patients on carbidopa and levodopa therapy have been reported very rarely. Caution should be exercised when interpreting the plasma and urine levels of catecholamines and their metabolites in patients on levodopa or carbidopa and levodopa therapy.
Drug Interactions
Caution should be exercised when the following drugs are administered concomitantly with PARCOPA® (carbidopa and levodopa orally disintegrating tablets).
Symptomatic postural hypotension has occurred when carbidopa and levodopa was added to the treatment of a patient receiving antihypertensive drugs. Therefore, when therapy with PARCOPA® is started, dosage adjustment of the antihypertensive drug may be required.
For patients receiving MAO inhibitors (Type A or B), see CONTRAINDICATIONS. Concomitant therapy with selegiline and carbidopa and levodopa may be associated with severe orthostatic hypotension not attributable to carbidopa and levodopa alone (see CONTRAINDICATIONS).
There have been rare reports of adverse reactions, including hypertension and dyskinesia, resulting from the concomitant use of tricyclic antidepressants and carbidopa and levodopa.
Dopamine D2 receptor antagonists (e.g., phenothiazines, butyrophenones, risperidone) and isoniazid may reduce the therapeutic effects of levodopa. In addition, the beneficial effects of levodopa in Parkinson’s disease have been reported to be reversed by phenytoin and papaverine. Patients taking these drugs with PARCOPA® should be carefully observed for loss of therapeutic response.
Iron salts may reduce the bioavailability of levodopa and carbidopa. The clinical relevance is unclear.
Although metoclopramide may increase the bioavailability of levodopa by increasing gastric emptying, metoclopramide may also adversely affect disease control by its dopamine receptor antagonistic properties.
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a two-year bioassay of carbidopa and levodopa, no evidence of carcinogenicity was found in rats receiving doses of approximately two times the maximum daily human dose of carbidopa and four times the maximum daily human dose of levodopa.
In reproduction studies with carbidopa and levodopa, no effects on fertility were found in rats receiving doses of approximately two times the maximum daily human dose of carbidopa and four times the maximum daily human dose of levodopa.
Pregnancy
Pregnancy Category C
No teratogenic effects were observed in a study in mice receiving up to 20 times the maximum recommended human dose of carbidopa and levodopa. There was a decrease in the number of live pups delivered by rats receiving approximately two times the maximum recommended human dose of carbidopa and approximately five times the maximum recommended human dose of levodopa during organogenesis. Carbidopa and levodopa caused both visceral and skeletal malformations in rabbits at all doses and ratios of carbidopa/levodopa tested, which ranged from 10 times/5 times the maximum recommended human dose of carbidopa/levodopa to 20 times/10 times the maximum recommended human dose of carbidopa/levodopa.
There are no adequate or well-controlled studies in pregnant women. It has been reported from individual cases that levodopa crosses the human placental barrier, enters the fetus, and is metabolized. Carbidopa concentrations in fetal tissue appeared to be minimal. Use of PARCOPA® in women of childbearing potential requires that the anticipated benefits of the drug be weighed against possible hazards to mother and child.
Adverse Reactions/Side Effects
The most common adverse reactions reported with carbidopa and levodopa therapy have included dyskinesias, such as choreiform, dystonic, and other involuntary movements and nausea.
The following other adverse reactions have been reported with carbidopa and levodopa:
Body as a Whole: chest pain, asthenia.
Cardiovascular: cardiac irregularities, hypotension, orthostatic effects including orthostatic hypotension, hypertension, syncope, phlebitis, palpitation.
Gastrointestinal: dark saliva, gastrointestinal bleeding, development of duodenal ulcer, anorexia, vomiting, diarrhea, constipation, dyspepsia, dry mouth, taste alterations.
Hematologic: agranulocytosis, hemolytic and nonhemolytic anemia, thrombocytopenia, leukopenia.
Hypersensitivity: angioedema, urticaria, pruritus, Henoch-Schonlein purpura, bullous lesions (including pemphigus-like reactions).
Musculoskeletal: back pain, shoulder pain, muscle cramps.
Nervous System/Psychiatric: psychotic episodes including delusions, hallucinations, and paranoid ideation, neuroleptic malignant syndrome (see WARNINGS), bradykinetic episodes (“on-off” phenomenon), confusion, agitation, dizziness, somnolence, dream abnormalities including nightmares, insomnia, paresthesia, headache, depression with or without development of suicidal tendencies, dementia, pathological gambling, increased libido including hypersexuality, impulse control symptoms. Convulsions also have occurred; however, a causal relationship with carbidopa and levodopa has not been established.
Respiratory: dyspnea, upper respiratory infection.
Skin: rash, increased sweating, alopecia, dark sweat.
Urogenital: urinary tract infection, urinary frequency, dark urine.
Laboratory Tests: decreased hemoglobin and hematocrit; abnormalities in alkaline phosphatase, SGOT (AST), SGPT (ALT), lactic dehydrogenase, bilirubin, blood urea nitrogen (BUN), Coombs test; elevated serum glucose; white blood cells, bacteria, and blood in the urine.
Other adverse reactions that have been reported with levodopa alone and with various carbidopa and levodopa formulations, and may occur with PARCOPA® are:
Body as a Whole: abdominal pain and distress, fatigue.
Cardiovascular: myocardial infarction.
Gastrointestinal: gastrointestinal pain, dysphagia, sialorrhea, flatulence, bruxism, burning sensation of the tongue, heartburn, hiccups.
Metabolic: edema, weight gain, weight loss.
Musculoskeletal: leg pain.
Nervous System/Psychiatric: ataxia, extrapyramidal disorder, falling, anxiety, gait abnormalities, nervousness, decreased mental acuity, memory impairment, disorientation, euphoria, blepharospasm (which may be taken as an early sign of excess dosage; consideration of dosage reduction may be made at this time), trismus, increased tremor, numbness, muscle twitching, activation of latent Horner’s syndrome, peripheral neuropathy.
Respiratory: pharyngeal pain, cough.
Skin: malignant melanoma (see also CONTRAINDICATIONS), flushing.
Special Senses: oculogyric crises, diplopia, blurred vision, dilated pupils.
Urogenital: urinary retention, urinary incontinence, priapism.
Miscellaneous: bizarre breathing patterns, faintness, hoarseness, malaise, hot flashes, sense of stimulation.
Laboratory Tests: decreased white blood cell count and serum potassium; increased serum creatinine and uric acid; protein and glucose in urine.
Overdosage
Management of acute overdosage with PARCOPA® is the same as management of acute overdosage with levodopa. Pyridoxine is not effective in reversing the actions of PARCOPA®.
General supportive measures should be employed, along with immediate gastric lavage. Intravenous fluids should be administered judiciously and an adequate airway maintained. Electrocardiographic monitoring should be instituted and the patient carefully observed for the development of arrhythmias; if required, appropriate anti-arrhythmic therapy should be given. The possibility that the patient may have taken other drugs as well as PARCOPA® should be taken into consideration. To date, no experience has been reported with dialysis; hence, its value in overdosage is not known.
Based on studies in which high doses of levodopa and/or carbidopa were administered, a significant proportion of rats and mice given single oral doses of levodopa of approximately 1500-2000 mg/kg are expected to die. A significant proportion of infant rats of both sexes are expected to die at a dose of 800 mg/kg. A significant proportion of rats are expected to die after treatment with similar doses of carbidopa. The addition of carbidopa in a 1:10 ratio with levodopa increases the dose at which a significant proportion of mice are expected to die to 3360 mg/kg.
Parcopa Dosage and Administration
Instructions for Use/Handling PARCOPA® Tablets
Just prior to administration, GENTLY remove the tablet from the bottle with dry hands. IMMEDIATELY place the PARCOPA® Tablet on top of the tongue where it will dissolve in seconds, then swallow with saliva. Administration with liquid is not necessary.
The optimum daily dosage of PARCOPA® must be determined by careful titration in each patient. PARCOPA® is available in a 1:4 ratio of carbidopa to levodopa (PARCOPA® 25/100) as well as 1:10 ratio (PARCOPA® 25/250 and PARCOPA® 10/100). Tablets of the two ratios may be given separately or combined as needed to provide the optimum dosage.
Studies show that peripheral dopa decarboxylase is saturated by carbidopa at approximately 70 to 100 mg a day. Patients receiving less than this amount of carbidopa are more likely to experience nausea and vomiting.
Usual Initial Dosage
Dosage is best initiated with one tablet of PARCOPA® 25/100 three times a day. This dosage schedule provides 75 mg of carbidopa per day. Dosage may be increased by one tablet every day or every other day, as necessary, until a dosage of eight tablets of PARCOPA® 25/100 a day is reached.
If PARCOPA® 10/100 is used, dosage may be initiated with one tablet three or four times a day. However, this will not provide an adequate amount of carbidopa for many patients. Dosage may be increased by one tablet every day or every other day until a total of eight tablets (2 tablets q.i.d.) is reached.
How to Transfer Patients from Levodopa
Levodopa must be discontinued at least twelve hours before starting PARCOPA® (carbidopa and levodopa orally disintegrating tablets). A daily dosage of PARCOPA® should be chosen that will provide approximately 25 percent of the previous levodopa dosage. Patients who are taking less than 1500 mg of levodopa a day should be started on one tablet of PARCOPA® 25/100 three or four times a day. The suggested starting dosage for most patients taking more than 1500 mg of levodopa is one tablet of PARCOPA® 25/250 three or four times a day.
Maintenance
Therapy should be individualized and adjusted according to the desired therapeutic response. At least 70 to 100 mg of carbidopa per day should be provided. When a greater proportion of carbidopa is required, one tablet of PARCOPA® 25/100 may be substituted for each tablet of PARCOPA® 10/100. When more levodopa is required, PARCOPA® 25/250 should be substituted for PARCOPA® 25/100 or PARCOPA® 10/100. If necessary, the dosage of PARCOPA® 25/250 may be increased by one-half or one tablet every day or every other day to a maximum of eight tablets a day. Experience with total daily dosages of carbidopa greater than 200 mg is limited.
Because both therapeutic and adverse responses occur more rapidly with PARCOPA® than with levodopa alone, patients should be monitored closely during the dose adjustment period. Specifically, involuntary movements will occur more rapidly with PARCOPA® than with levodopa. The occurrence of involuntary movements may require dosage reduction. Blepharospasm may be a useful early sign of excess dosage in some patients.
Addition of Other Antiparkinsonian Medications
Standard drugs for Parkinson’s disease, other than levodopa without a decarboxylase inhibitor, may be used concomitantly while PARCOPA® is being administered, although dosage adjustments may be required.
Interruption of Therapy
Sporadic cases of a symptom complex resembling Neuroleptic Malignant Syndrome (NMS) have been associated with dose reductions and withdrawal of carbidopa and levodopa. Patients should be observed carefully if abrupt reduction or discontinuation of PARCOPA® is required, especially if the patient is receiving neuroleptics. (See WARNINGS.)
If general anesthesia is required, PARCOPA® may be continued as long as the patient is permitted to take fluids and medication by mouth. If therapy is interrupted temporarily, the patient should be observed for symptoms resembling NMS, and the usual daily dosage may be administered as soon as the patient is able to take oral medication.
How is Parcopa supplied
PARCOPA® (carbidopa and levodopa orally disintegrating tablets) 25/100 are yellow, round, flat-faced, mint-flavored, scored and engraved “25/100” on the unscored side and “SP” above and “342” below the score on the other side. They are supplied as follows:
Bottles of 100 NDC 18860-342-01
PARCOPA® (carbidopa and levodopa orally disintegrating tablets) 10/100 are blue, round, flat-faced, mint-flavored, scored and engraved “10/100” on the unscored side and “SP” above and “341” below the score on the other side. They are supplied as follows:
Bottles of 100 NDC 18860-341-01
PARCOPA® (carbidopa and levodopa orally disintegrating tablets) 25/250 are blue, round, flat-faced, mint-flavored, scored, and engraved “25/250” on the unscored side and “SP” above and “343” below the score on the other side. They are supplied as follows:
Bottles of 100 NDC 18860-343-01
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture and light.
Dispense in a tight, light-resistant container as defined in the USP/NF.
Distributed by:
Azur Pharma, Inc.
Philadelphia, PA 19103, USA
U.S. Patent Nos. 6,024,981 and 6,221,392.
PARCOPA® is a registered trademark of SRZ Properties, Inc.
Rev. 01/11
Principal Display Panel - 10/100 mg Bottle Label
NDC 18860-341-01
100 tablets
PARCOPA®
(carbidopa and levodopa orally disintegrating tablets)
10 mg /100 mg
Rx only
AZUR PHARMA
orally disintegrating tablets
Each mint-flavored, orally disintegrating tablet contains 10 mg carbidopa and 100 mg levodopa.
Phenylketonurics: contains phenylalanine 3.4 mg per tablet.
USUAL DOSAGE: See package insert for further information. Do not remove PARCOPA® Tablets from the bottle until immediately before use.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture and light.
Dispense in a tight, light-resistant container as defined in the USP/NF.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Azur Pharma, Inc.
Philadelphia, PA 19103, USA
U.S. Patent Nos. 6,024,981 and 6,221,392
PARCOPA® is a registered trademark of SRZ Properties, Inc.
PARCt-10-12
Rev. 01/11 2E
00017317
Principal Display Panel - 25/100 mg Bottle Label
NDC 18860-342-01
100 tablets
PARCOPA®
(carbidopa and levodopa orally disintegrating tablets)
25 mg /100 mg
Rx only
AZUR PHARMA
orally disintegrating tablets
Each mint-flavored, orally disintegrating tablet contains 25 mg carbidopa and 100 mg levodopa.
Phenylketonurics: contains phenylalanine 3.4 mg per tablet.
USUAL DOSAGE: See package insert for further information. Do not remove PARCOPA® Tablets from the bottle until immediately before use.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture and light.
Dispense in a tight, light-resistant container as defined in the USP/NF.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Azur Pharma, Inc.
Philadelphia, PA 19103, USA
U.S. Patent Nos. 6,024,981 and 6,221,392
PARCOPA® is a registered trademark of SRZ Properties, Inc.
PARCt-10-22
Rev. 01/11 2E
00017318
Principal Display Panel - 25/250 mg Bottle Label
NDC 18860-343-01
100 tablets
PARCOPA®
(carbidopa and levodopa orally disintegrating tablets)
25 mg / 250 mg
Rx only
AZUR PHARMA
orally disintegrating tablets
Each mint-flavored, orally disintegrating tablet contains 25 mg carbidopa and 250 mg levodopa.
Phenylketonurics: contains phenylalanine 8.4 mg per tablet.
USUAL DOSAGE: See package insert for further information. Do not remove PARCOPA® Tablets from the bottle until immediately before use.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture and light.
Dispense in a tight, light-resistant container as defined in the USP/NF.
KEEP OUT OF REACH OF CHILDREN.
Distributed by:
Azur Pharma, Inc.
Philadelphia, PA 19103, USA
U.S. Patent Nos. 6,024,981 and 6,221,392
PARCOPA® is a registered trademark of SRZ Properties, Inc.
PARCt-10-32
Rev. 01/11 2E
00017319
| PARCOPA
carbidopa and levodopa tablet, orally disintegrating |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PARCOPA
carbidopa and levodopa tablet, orally disintegrating |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| PARCOPA
carbidopa and levodopa tablet, orally disintegrating |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Jazz Pharmaceuticals, Inc. (135926363) |
Frequently asked questions
- How long does it take carbidopa levodopa to work?
- How often should carbidopa/levodopa be taken?
- Can carbidopa/levodopa cause high blood pressure?
- What foods should be avoided when taking levodopa?
- Is Rytary better than Sinemet?
- What is the difference between carbidopa, levodopa, and Rytary?
- How long does it take for Rytary to start working?
- How long does Rytary stay in your system?
More about Parcopa (carbidopa / levodopa)
- Check interactions
- Compare alternatives
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: dopaminergic antiparkinsonism agents
Patient resources
Professional resources
Other brands
Sinemet, Rytary, Sinemet CR, Crexont, ... +2 more