Lessina Prescribing Information
Package insert / product label
Generic name: levonorgestrel and ethinyl estradiol
Dosage form: tablets
Drug class: Contraceptives
Medically reviewed by Drugs.com. Last updated on Dec 25, 2023.
On This Page
Cigarette smoking increases the risk of serious cardiovascular side effects from oral contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes per day) and is quite marked in women over 35 years of age. Women who use oral contraceptives should be strongly advised not to smoke.
Lessina Description
Each cycle of Lessina® 28 (levonorgestrel and ethinyl estradiol tablets USP) consists of 21 pink active tablets each containing 0.1 mg levonorgestrel, USP and 0.02 mg ethinyl estradiol, USP; and seven white inert tablets. The inactive ingredients include anhydrous lactose, FD&C red no. 40 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, corn starch, and titanium dioxide. Each white tablet contains only inert ingredients as follows: anhydrous lactose, hypromellose, microcrystalline cellulose, and magnesium stearate.
The structural formulas are as follows:
Levonorgestrel, USP
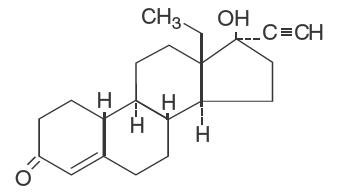
C21H28O2 Molecular Weight: 312.4
Ethinyl Estradiol, USP
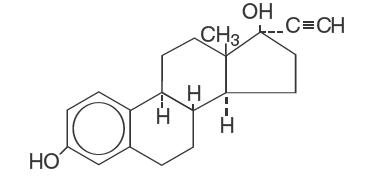
C20H24O2 Molecular Weight: 296.4
Lessina - Clinical Pharmacology
Combination oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which reduce the likelihood of implantation).
PHARMACOKINETICS
Absorption
No specific investigation of the absolute bioavailability of levonorgestrel and ethinyl estradiol tablets in humans has been conducted. However, literature indicates that levonorgestrel is rapidly and completely absorbed after oral administration and is not subject to first-pass metabolism. Ethinyl estradiol is rapidly and almost completely absorbed from the gastrointestinal tract but, due to first-pass metabolism in gut mucosa and liver, the absolute bioavailability of ethinyl estradiol is about 40%. After a single dose of three levonorgestrel and ethinyl estradiol tablets to 17 women under fasting conditions, the extents of absorption of levonorgestrel and ethinyl estradiol were 98.6% and 99%, respectively, relative to the same dose of the 2 drugs when given as a microcrystalline suspension in water. The effect of food on the bioavailability of levonorgestrel and ethinyl estradiol tablets following oral administration has not been evaluated.
The pharmacokinetics of levonorgestrel and ethinyl estradiol following daily administration of levonorgestrel and ethinyl estradiol tablets for 21 days per cycle for three cycles, were determined in 18 women. Estimates of the pharmacokinetic parameters of levonorgestrel and ethinyl estradiol following single and multiple dose administration of levonorgestrel and ethinyl estradiol tablets are summarized in Table I. Mean levonorgestrel and ethinyl estradiol levels after a single dose and on day 21 at steady state are shown in Figure I.
The pharmacokinetics of total levonorgestrel are non-linear due to an increase in binding to SHBG, which is attributed to increased SHBG levels that are induced by the daily administration of ethinyl estradiol. Increased binding of levonorgestrel to SHBG leads to decreased clearance of levonorgestrel. Observed maximum levonorgestrel concentrations increased from day 1 to day 21 of the 1st and 3rd cycles by 66% and 83%, respectively.
Figure I
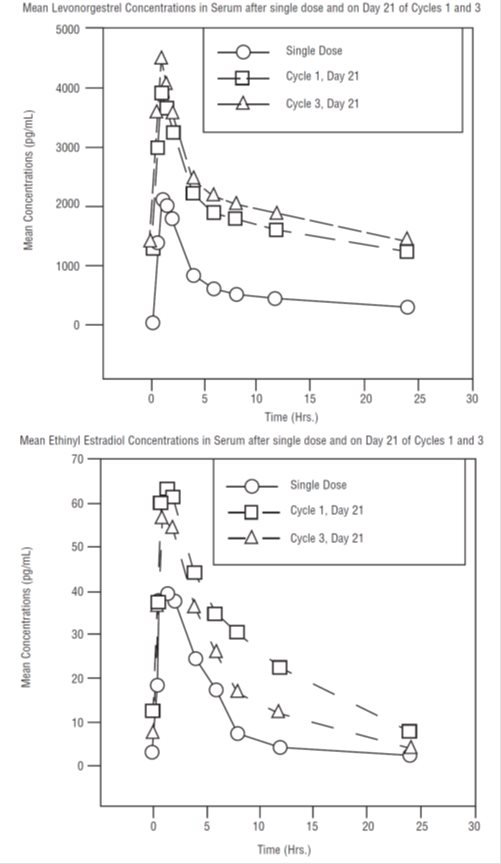
In calculating the mean concentration for ethinyl estradiol, any individual subject value below the quantifiable limit (i.e., 20 pg/mL) was converted to 0; and the 0 values were included for calculation of the mean concentration.
Table I provides a summary of levonorgestrel and ethinyl estradiol pharmacokinetic parameters.
TABLE I MEAN (SD) PHARMACOKINETIC PARAMETERS OF LEVONORGESTREL AND ETHINYL ESTRADIOL TABLETS AFTER SINGLE DOSE AND AFTER MULTIPLE DOSING FOR 3 CYCLES
Levonorgestrel
| Day (cycle) | Cmax ng/mL | tmax h | AUC ng·h/mL | CL/F mL/min/kg | Vz L | SHBG nmol/L |
| 1 | 2.36(0.79) | 1.3(0.4) | 29.2(10) | 1(0.3) | 129(46) | 64.5(22) |
| AUC(0 to 24h) ng·h/mL | ||||||
| 21(1) | 4.04(2.08) | 1(0.3) | 43.8(22.4) | 0.73(0.34) | 106(42) | 94.7(37.4) |
| 21(3) | 4.53(1.94) | 1(0.3) | 49.5(24.5) | 0.65(0.33) | 96(35) | 107.4(45.8) |
Ethinyl Estradiol
| Day (cycle) | Cmax
| tmax
| AUC(0 to 24h) |
| 1 | 49.5(13.4) | 1.5(0.4) | 298(215) |
| 21(1) | 66.2(29.5) | 1.4(0.4) | 596(494) |
| 21(3) | 58.1(19.3) | 1.4(0.3) | 417(289) |
Cmax = maximum concentration
tmax = time to maximum concentration
AUC = area under the drug concentration curve from time 0 to infinity
CL/F = oral clearance
Vz = volume of distribution
SHBG = sex hormone binding globulin
AUC(0 to 24) = area under the drug concentration time curve from time 0 to 24 hours; this represents the area for one dosing interval at steady state.
Distribution
Levonorgestrel in serum is primarily bound to SHBG. Protein binding values for levonorgestrel are provided in Table II. Ethinyl estradiol is about 97% bound to plasma albumin. Ethinyl estradiol does not bind to SHBG, but induces SHBG synthesis.
TABLE II.
Protein binding (mean ± SD) of levonorgestrel in pools of serum samples collected from 18 women after a single dose of levonorgestrel and ethinyl estradiol tablets, and following administration (once daily) over 3x21 days.
| Parameter | Single Dose | Cycle 2 | Cycle 4 |
| % free | 1.11(0.27) | 0.79(0.22) | 0.80(0.23) |
| % SHBG-bound | 64.5(8.54) | 75.6(6.59) | 74.7(7.89) |
| % albumin-bound | 34.4(8.28) | 23.6(6.41) | 24.5(7.67) |
Metabolism
Levonorgestrel
The most important metabolic pathway occurs in the reduction of the Δ4-3-oxo group and hydroxylation at positions 2α, 1ß, and 16ß, followed by conjugation. Most of the metabolites that circulate in the blood are sulfates of 3α, 5ß-tetrahydro-levonorgestrel, while excretion occurs predominantly in the form of glucuronides. Some of the parent levonorgestrel also circulates as 17ß-sulfate. Metabolic clearance rates may differ among individuals by several-fold, and this may account in part for the wide variation in levonorgestrel concentrations among users.
Ethinyl estradiol
Cytochrome P450 enzymes (CYP3A4) in the liver are responsible for the 2-hydroxylation that is the major oxidative reaction. The 2-hydroxy metabolite is further transformed by methylation and glucuronidation prior to urinary and fecal excretion. Levels of Cytochrome P450 (CYP3A) vary widely among individuals and can explain the variation in the rates of ethinyl estradiol 2-hydroxylation. Ethinyl estradiol is excreted in the urine and feces as glucuronide and sulfate conjugates and undergoes enterohepatic circulation.
Excretion
The elimination half-life for levonorgestrel after a single dose of levonorgestrel and ethinyl estradiol tablets is 25.4 ± 9.7 hours. Levonorgestrel and its metabolites are primarily excreted in the urine. The elimination half-life of ethinyl estradiol has been reported to be between 15 and 25 hours.
SPECIAL POPULATIONS
Hepatic Insufficiency
No formal studies have evaluated the effect of hepatic disease on the disposition of levonorgestrel and ethinyl estradiol tablets. However, steroid hormones may be poorly metabolized in patients with impaired liver function.
Renal Insufficiency
No formal studies have evaluated the effect of renal disease on the disposition of levonorgestrel and ethinyl estradiol tablets.
Drug-Drug Interactions
Interactions between ethinyl estradiol and other drugs have been reported in the literature.
- Interactions with Absorption: Diarrhea may increase gastrointestinal motility and reduce hormone absorption. Similarly, any drug which reduces gut transit time may reduce hormone concentrations in the blood.
- Interactions with Metabolism: Gastrointestinal Wall: Sulfation of ethinyl estradiol has been shown to occur in the gastrointestinal wall. Therefore, drugs which act as competitive inhibitors for sulfation in the gastrointestinal wall may increase ethinyl estradiol bioavailability.
- Hepatic metabolism: Interactions can occur with drugs that induce microsomal enzymes which can decrease ethinyl estradiol concentrations (e.g., rifampin, barbiturates, phenylbutazone, phenytoin, griseofulvin).
- Interference with Enterohepatic Circulation: Some clinical reports suggest that enterohepatic circulation of estrogens may decrease when certain antibiotic agents are given, which may reduce ethinyl estradiol concentrations (e.g., ampicillin, tetracycline).
- Interference in the Metabolism of Other Drugs: Ethinyl estradiol may interfere with the metabolism of other drugs by inhibiting hepatic microsomal enzymes or by inducing hepatic drug conjugation, particularly glucuronidation. Accordingly, plasma and tissue concentrations may either be increased or decreased, respectively (e.g., cyclosporin, theophylline).
Indications and Usage for Lessina
Oral contraceptives are indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception. Oral contraceptives are highly effective. Table III lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization, depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
| % of Women Experiencing | % of Women Continuing Use at One Year3 |
||
| Method (1) | Typical Use1 (2) | Perfect Use2 (3) |
|
| Chance4 | 85 | 85 | |
| Spermicides5 | 26 | 6 | 40 |
| Periodic abstinence | 25 | 63 |
|
| Calendar | 9 | ||
| Ovulation method | 3 | ||
| Sympto-thermal6 | 2 | ||
| Post ovulation | 1 | ||
| Withdrawal | 19 | 4 | |
| Cap7 | |||
| Parous women | 40 | 26 | 42 |
| Nulliparous women | 20 | 9 | 56 |
| Sponge | |||
| Parous women | 40 | 20 | 42 |
| Nulliparous women | 20 | 9 | 56 |
| Diaphragm7 | 20 | 6 | 56 |
| Condom8 | |||
| Female (Reality) | 21 | 5 | 56 |
| Male | 14 | 3 | 61 |
| Pill | 5 | 71 |
|
| Progestin only | 0.5 | ||
| Combined | 0.1 | ||
| IUD | |||
| Progesterone T | 2 | 1.5 | 81 |
| Copper T 380A | 0.8 | 0.6 | 78 |
| Lng 20 | 0.1 | 0.1 | 81 |
| Depo Provera | 0.3 | 0.3 | 70 |
| Norplant and Norplant-2 | 0.05 | 0.05 | 88 |
| Female sterilization | 0.5 | 0.5 | 100 |
| Male sterilization | 0.15 | 0.1 | 100 |
|
Source: Trussell J, Contraceptive efficacy. In Hatcher RA, Trussell J, Stewart F, Cates W, Stewart GK, Kowal D, Guest F, Contraceptive Technology: Seventeenth Revised Edition. New York NY: Irvington Publishers, 1998, in press. |
|||
1. Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
2. Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
3. Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year.
4. The percentages becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percentage who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether.
5.Foams, creams, gels, vaginal suppositories, vaginal film.
6. Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the postovulatory phases.
7. With spermicidal cream or jelly.
8. Without spermicides.
Contraindications
Lessina is contraindicated in females who are known to have or develop the following conditions:
- Thrombophlebitis or thromboembolic disorders
- A past history of deep-vein thrombophlebitis or thromboembolic disorders
- Cerebral-vascular or coronary-artery disease
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive
- Carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia
- Undiagnosed abnormal genital bleeding
- Cholestatic jaundice of pregnancy or jaundice with prior pill use
- Hepatic adenomas or carcinomas
- Known or suspected pregnancy
- Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see WARNINGS, Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment).
Warnings
The use of oral contraceptives is associated with increased risks of several serious conditions including myocardial infarction, thromboembolism, stroke, hepatic neoplasia, gallbladder disease, and hypertension, although the risk of serious morbidity or mortality is very small in healthy women without underlying risk factors. The risk of morbidity and mortality increases significantly in the presence of other underlying risk factors such as hypertension, hyperlipidemias, obesity and diabetes.
Practitioners prescribing oral contraceptives should be familiar with the following information relating to these risks.
The information contained in this package insert is based principally on studies carried out in patients who used oral contraceptives with higher formulations of estrogens and progestogens than those in common use today. The effect of long-term use of the oral contraceptives with lower formulations of both estrogens and progestogens remains to be determined.
Throughout this labeling, epidemiologic studies reported are of two types: retrospective or case control studies and prospective or cohort studies. Case control studies provide a measure of the relative risk of a disease, namely, a ratio of the incidence of a disease among oral contraceptive users to that among nonusers. The relative risk does not provide information on the actual clinical occurrence of a disease. Cohort studies provide a measure of attributable risk, which is the difference in the incidence of disease between oral contraceptive users and nonusers. The attributable risk does provide information about the actual occurrence of a disease in the population. For further information, the reader is referred to a text on epidemiologic methods.
1. Thromboembolic disorders and other vascular problems
a. Myocardial Infarction
An increased risk of myocardial infarction has been attributed to oral contraceptive use. This risk is primarily in smokers or women with other underlying risk factors for coronary-artery disease such as hypertension, hypercholesterolemia, morbid obesity, and diabetes. The relative risk of heart attack for current oral contraceptive users has been estimated to be two to six. The risk is very low under the age of 30. Smoking in combination with oral contraceptive use has been shown to contribute substantially to the incidence of myocardial infarctions in women in their mid-thirties or older with smoking accounting for the majority of excess cases. Mortality rates associated with circulatory disease have been shown to increase substantially in smokers over the age of 35 and nonsmokers over the age of 40 (Table IV) among women who use oral contraceptives.
| AGE | EVER-USERS NON- SMOKERS | EVER-USERS SMOKERS | CONTROLS NON- SMOKERS | CONTROL SMOKERS |
| 15 to 24 | 0 | 10.5 | 0 | 0 |
| 25 to 34 | 4.4 | 14.2 | 2.7 | 4.2 |
| 35 to 44 | 21.5 | 63.4 | 6.4 | 15.2 |
| 45+ | 52.4 | 206.7 | 11.4 | 27.9 |
Oral contraceptives may compound the effects of well-known risk factors, such as hypertension, diabetes, hyperlipidemias, age and obesity. In particular, some progestogens are known to decrease HDL cholesterol and cause glucose intolerance, while estrogens may create a state of hyperinsulinism. Oral contraceptives have been shown to increase blood pressure among users (see section 10 in WARNINGS). Similar effects on risk factors have been associated with an increased risk of heart disease. Oral contraceptives must be used with caution in women with cardiovascular disease risk factors.
b. Thromboembolism
An increased risk of thromboembolic and thrombotic disease associated with the use of oral contraceptives is well established. Case control studies have found the relative risk of users compared to nonusers to be 3 for the first episode of superficial venous thrombosis, 4 to 11 for deep vein thrombosis or pulmonary embolism, and 1.5 to 6 for women with predisposing conditions for venous thromboembolic disease. Cohort studies have shown the relative risk to be somewhat lower, about 3 for new cases and about 4.5 for new cases requiring hospitalization. The risk of thromboembolic disease due to oral contraceptives is not related to length of use and disappears after pill use is stopped.
A two- to four-fold increase in the relative risk of post-operative thromboembolic complications has been reported with the use of oral contraceptives. The relative risk of venous thrombosis in women who have predisposing conditions is twice that of women without such medical conditions. If feasible, oral contraceptives should be discontinued from at least four weeks prior to and for two weeks after elective surgery of a type associated with an increase in risk of thromboembolism and during and following prolonged immobilization. Since the immediate postpartum period is also associated with an increased risk of thromboembolism, oral contraceptives should be started no earlier than four to six weeks after delivery in women who elect not to breast-feed.
c. Cerebrovascular diseases
Oral contraceptives have been shown to increase both the relative and attributable risks of cerebrovascular events (thrombotic and hemorrhagic strokes), although, in general, the risk is greatest among older (>35 years), hypertensive women who also smoke. Hypertension was found to be a risk factor, for both users and nonusers, for both types of strokes, while smoking interacted to increase the risk for hemorrhagic strokes.
In a large study, the relative risk of thrombotic strokes has been shown to range from 3 for normotensive users to 14 for users with severe hypertension. The relative risk of hemorrhagic stroke is reported to be 1.2 for nonsmokers who used oral contraceptives, 2.6 for smokers who did not use oral contraceptives, 7.6 for smokers who used oral contraceptives, 1.8 for normotensive users and 25.7 for users with severe hypertension. The attributable risk is also greater in older women.
d. Dose-related risk of vascular disease from oral contraceptives
A positive association has been observed between the amount of estrogen and progestogen in oral contraceptives and the risk of vascular disease. A decline in serum high-density lipoproteins (HDL) has been reported with many progestational agents. A decline in serum high-density lipoproteins has been associated with an increased incidence of ischemic heart disease. Because estrogens increase HDL cholesterol, the net effect of an oral contraceptive depends on a balance achieved between doses of estrogen and progestogen and the nature and absolute amount of progestogen used in the contraceptive. The amount of both hormones should be considered in the choice of an oral contraceptive.
Minimizing exposure to estrogen and progestogen is in keeping with good principles of therapeutics. For any particular estrogen/progestogen combination, the dosage regimen prescribed should be one which contains the least amount of estrogen and progestogen that is compatible with a low failure rate and the needs of the individual patient. New acceptors of oral-contraceptive agents should be started on preparations containing the lowest estrogen content which provides satisfactory results in the individual.
e. Persistence of risk of vascular disease
There are two studies which have shown persistence of risk of vascular disease for ever-users of oral contraceptives. In a study in the United States, the risk of developing myocardial infarction after discontinuing oral contraceptives persists for at least 9 years for women aged 40 to 49 years who had used oral contraceptives for five or more years, but this increased risk was not demonstrated in other age groups. In another study in Great Britain, the risk of developing cerebrovascular disease persisted for at least 6 years after discontinuation of oral contraceptives, although excess risk was very small. However, both studies were performed with oral contraceptive formulations containing 50 micrograms or higher of estrogens.
2. Estimates of mortality from contraceptive use
One study gathered data from a variety of sources which have estimated the mortality rate associated with different methods of contraception at different ages (Table V). These estimates include the combined risk of death associated with contraceptive methods plus the risk attributable to pregnancy in the event of method failure. Each method of contraception has its specific benefits and risks. The study concluded that with the exception of oral contraceptive users 35 and older who smoke and 40 and older who do not smoke, mortality associated with all methods of birth control is less than that associated with childbirth.
The observation of a possible increase in risk of mortality with age for oral contraceptive users is based on data gathered in the 1970’s-but not reported until 1983. However, current clinical practice involves the use of lower estrogen dose formulations combined with careful restriction of oral contraceptive use to women who do not have the various risk factors listed in this labeling.
Because of these changes in practice and, also, because of some limited new data which suggest that the risk of cardiovascular disease with the use of oral contraceptives may now be less than previously observed, the Fertility and Maternal Health Drugs Advisory Committee was asked to review the topic in 1989. The Committee concluded that although cardiovascular disease risks may be increased with oral contraceptive use after age 40 in healthy nonsmoking women (even with the newer low-dose formulations), there are greater potential health risks associated with pregnancy in older women and with the alternative surgical and medical procedures which may be necessary if such women do not have access to effective and acceptable means of contraception.
Therefore, the Committee recommended that the benefits of oral contraceptive use by healthy nonsmoking women over 40 may outweigh the possible risks. Of course, older women, as all women who take oral contraceptives, should take the lowest possible dose formulation that is effective.
| Method of Control and Outcome | 15 to 19 | 20 to 24 | 25 to 29 | 30 to 34 | 35 to 39 | 40 to 44 |
| No fertility | 7 | 7.4 | 9.1 | 14.8 | 25.7 | 28.2 |
| Oral contraceptives | 0.3 | 0.5 | 0.9 | 1.9 | 13.8 | 31.6 |
| Oral contraceptives | 2.2 | 3.4 | 6.6 | 13.5 | 51.1 | 117.2 |
| IUD2 | 0.8 | 0.8 | 1 | 1 | 1.4 | 1.4 |
| Condom1 | 1.1 | 1.6 | 0.7 | 0.2 | 0.3 | 0.4 |
| Diaphragm/ | 1.9 | 1.2 | 1.2 | 1.3 | 2.2 | 2.8 |
| Periodic | 2.5 | 1.6 | 1.6 | 1.7 | 2.9 | 3.6 |
|
Adapted from H.W. Ory, Family Planning Perspectives 15:57-63, 1983. |
||||||
1. Deaths are method related
2. Deaths are birth related
3. Malignant Neoplasms
Breast Cancer
Lessina is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive (see CONTRAINDICATIONS).
Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use (see POSTMARKETING EXPERIENCE).
Cervical Cancer
Some studies suggest that oral contraceptive use has been associated with an increase in the risk of cervical intraepithelial neoplasia in some populations of women. However, there continues to be controversy about the extent to which such findings may be due to differences in sexual behavior and other factors.
In spite of many studies of the relationship between oral contraceptive use and breast and cervical cancers, a cause-and-effect relationship has not been established.
4. Hepatic neoplasia
Benign hepatic adenomas are associated with oral contraceptive use, although the incidence of benign tumors is rare in the United States. Indirect calculations have estimated the attributable risk to be in the range of 3.3 cases/100,000 for users, a risk that increases after four or more years of use. Rupture of rare, benign, hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies from Britain have shown an increased risk of developing hepatocellular carcinoma in long-term (>8 years) oral contraceptive users. However, these cancers are extremely rare in the U.S. and the attributable risk (the excess incidence) of liver cancers in oral contraceptive users approaches less than one per million users.
5. Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications such as COCs. Discontinue Lessina prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir (see CONTRAINDICATIONS). Lessina can be restarted approximately 2 weeks following completion of treatment with the combination drug regimen.
6. Ocular lesions
There have been clinical case reports of retinal thrombosis associated with the use of oral contraceptives. Oral contraceptives should be discontinued if there is unexplained partial or complete loss of vision; onset of proptosis or diplopia; papilledema; or retinal vascular lesions. Appropriate diagnostic and therapeutic measures should be undertaken immediately.
7. Oral contraceptive use before or during early pregnancy
Extensive epidemiological studies have revealed no increased risk of birth defects in women who have used oral contraceptives prior to pregnancy. Studies also do not suggest a teratogenic effect, particularly insofar as cardiac anomalies and limb-reduction defects are concerned, when taken inadvertently during early pregnancy.
The administration of oral contraceptives to induce withdrawal bleeding should not be used as a test for pregnancy. Oral contraceptives should not be used during pregnancy to treat threatened or habitual abortion. It is recommended that for any patient who has missed two consecutive periods, pregnancy should be ruled out before continuing oral contraceptive use. If the patient has not adhered to the prescribed schedule, the possibility of pregnancy should be considered at the time of the first missed period. Oral contraceptive use should be discontinued if pregnancy is confirmed.
8. Gallbladder disease
Earlier studies have reported an increased lifetime relative risk of gallbladder surgery in users of oral contraceptives and estrogens. More recent studies, however, have shown that the relative risk of developing gallbladder disease among oral contraceptive users may be minimal. The recent findings of minimal risk may be related to the use of oral contraceptive formulations containing lower hormonal doses of estrogens and progestogens.
9. Carbohydrate and lipid metabolic effects
Oral contraceptives have been shown to cause glucose intolerance in a significant percentage of users. Oral contraceptives containing greater than 75 micrograms of estrogens cause hyperinsulinism, while lower doses of estrogen cause less glucose intolerance. Progestogens increase insulin secretion and create insulin resistance, this effect varying with different progestational agents. However, in the nondiabetic woman, oral contraceptives appear to have no effect on fasting blood glucose. Because of these demonstrated effects, prediabetic and diabetic women should be carefully observed while taking oral contraceptives.
A small proportion of women will have persistent hypertriglyceridemia while on the pill. As discussed earlier (See WARNINGS, 1a. and 1d.), changes in serum triglycerides and lipoprotein levels have been reported in oral contraceptive users.
10. Elevated blood pressure
An increase in blood pressure has been reported in women taking oral contraceptives and this increase is more likely in older oral contraceptive users and with continued use. Data from the Royal College of General Practitioners and subsequent randomized trials have shown that the incidence of hypertension increases with increasing quantities of progestogens.
Women with a history of hypertension or hypertension-related diseases, or renal disease should be encouraged to use another method of contraception. If women with hypertension elect to use oral contraceptives, they should be monitored closely, and if significant elevation of blood pressure occurs, oral contraceptives should be discontinued. For most women, elevated blood pressure will return to normal after stopping oral contraceptives, and there is no difference in the occurrence of hypertension among ever- and never-users.
11. Headache
The onset or exacerbation of migraine or development of headache with a new pattern which is recurrent, persistent or severe requires discontinuation of oral contraceptives and evaluation of the cause.
12. Bleeding irregularities
Breakthrough bleeding and spotting are sometimes encountered in patients on oral contraceptives, especially during the first three months of use. Nonhormonal causes should be considered and adequate diagnostic measures taken to rule out malignancy or pregnancy in the event of breakthrough bleeding, as in the case of any abnormal vaginal bleeding. If pathology has been excluded, time or a change to another formulation may solve the problem. In the event of amenorrhea, pregnancy should be ruled out.
Some women may encounter post-pill amenorrhea or oligomenorrhea, especially when such a condition was pre-existent.
Precautions
1. General
Patients should be counseled that this product does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
2. Physical examination and follow-up
It is good medical practice for all women to have annual history and physical examinations, including women using oral contraceptives. The physical examination, however, may be deferred until after initiation of oral contraceptives if requested by the woman and judged appropriate by the clinician. The physical examination should include special reference to blood pressure, breasts, abdomen and pelvic organs, including cervical cytology and relevant laboratory tests. In case of undiagnosed, persistent or recurrent abnormal vaginal bleeding, appropriate measures should be conducted to rule out malignancy. Women with a strong family history of breast cancer or who have breast nodules should be monitored with particular care.
3. Lipid disorders
Women who are being treated for hyperlipidemias should be followed closely if they elect to use oral contraceptives. Some progestogens may elevate LDL levels and may render the control of hyperlipidemias more difficult.
4. Liver function
If jaundice develops in any woman receiving such drugs, the medication should be discontinued. Steroid hormones may be poorly metabolized in patients with impaired liver function.
5. Fluid retention
Oral contraceptives may cause some degree of fluid retention. They should be prescribed with caution, and only with careful monitoring, in patients with conditions which might be aggravated by fluid retention.
6. Emotional disorders
Women with a history of depression should be carefully observed and the drug discontinued if depression recurs to a serious degree.
7. Contact lenses
Contact-lens wearers who develop visual changes or changes in lens tolerance should be assessed by an ophthalmologist.
8. Drug interactions
Reduced efficacy and increased incidence of breakthrough bleeding and menstrual irregularities have been associated with concomitant use of rifampin. A similar association, though less marked, has been suggested with barbiturates, phenylbutazone, phenytoin sodium, and possibly with griseofulvin, ampicillin and tetracyclines.
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not co-administer Lessina with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations (see WARNINGS, Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment).
9. Interactions with laboratory tests
Certain endocrine- and liver-function tests and blood components may be affected by oral contraceptives:
- Increased prothrombin and factors VII, VIII, IX and X; decreased antithrombin 3; increased norepinephrine-induced platelet aggregability.
- Increased thyroid-binding globulin (TBG) leading to increased circulating total thyroid hormone, as measured by protein-bound iodine (PBI), T4 by column or by radioimmunoassay. Free T3 resin uptake is decreased, reflecting the elevated TBG, free T4 concentration is unaltered.
- Other binding proteins may be elevated in serum.
- Sex-binding globulins are increased and result in elevated levels of total circulating sex steroids and corticoids; however, free or biologically active levels remain unchanged.
- Triglycerides may be increased.
- Glucose tolerance may be decreased.
- Serum folate levels may be depressed by oral contraceptive therapy. This may be of clinical significance if a woman becomes pregnant shortly after discontinuing oral contraceptives.
12. Nursing mothers
Small amounts of oral contraceptive steroids have been identified in the milk of nursing mothers, and a few adverse effects on the child have been reported, including jaundice and breast enlargement. In addition, oral contraceptives given in the postpartum period may interfere with lactation by decreasing the quantity and quality of breast milk. If possible, the nursing mother should be advised not to use oral contraceptives but to use other forms of contraception until she has completely weaned her child.
13. Pediatric use
Safety and efficacy of levonorgestrel and ethinyl estradiol tablets have been established in women of reproductive age. Safety and efficacy are expected to be the same for post-pubertal adolescents under the age of 16 and for users 16 years and older. Use of this product before menarche is not indicated.
Adverse Reactions/Side Effects
Post Marketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure II).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure II). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure II Risk of Breast Cancer with Combined Oral Contraceptive Use
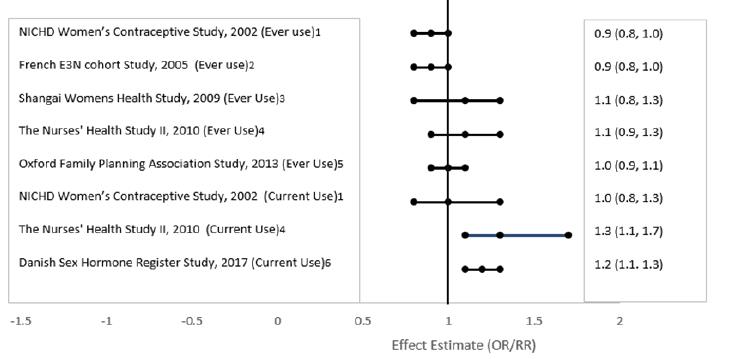
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
An increased risk of the following serious adverse reactions has been associated with the use of oral contraceptives (see WARNINGS section).
- Thrombophlebitis
- Arterial thromboembolism
- Pulmonary embolism
- Myocardial infarction
- Cerebral hemorrhage
- Cerebral thrombosis
- Hypertension
- Gallbladder disease
- Hepatic adenomas or benign liver tumors
There is evidence of an association between the following conditions and the use of oral contraceptives, although additional confirmatory studies are needed:
- Mesenteric thrombosis
- Retinal thrombosis
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug related:
- Nausea
- Vomiting
- Gastrointestinal symptoms, (such as abdominal cramps and bloating)
- Breakthrough bleeding
- Spotting
- Change in menstrual flow
- Amenorrhea
- Temporary infertility after discontinuation of treatment
- Edema
- Melasma which may persist
- Breast changes: tenderness, enlargement, secretion
- Change in weight (increase or decrease)
- Change in cervical erosion and secretion
- Diminution in lactation when given immediately postpartum
- Cholestatic jaundice
- Migraine
- Rash (allergic)
- Mental depression
- Reduced tolerance to carbohydrates
- Vaginal candidiasis
- Change in corneal curvature (steepening)
- Intolerance to contact lenses
The following adverse reactions have been reported in users of oral contraceptives and the association has been neither confirmed nor refuted:
- Pre-menstrual syndrome
- Cataracts
- Optic neuritis
- Changes in appetite
- Cystitis-like syndrome
- Headache
- Nervousness
- Dizziness
- Hirsutism
- Loss of scalp hair
- Erythema multiforme
- Erythema nodosum
- Hemorrhagic eruption
- Vaginitis
- Porphyria
- Impaired renal function
- Hemolytic uremic syndrome
- Budd-Chiari syndrome
- Acne
- Changes in libido
- Colitis
Overdosage
Serious ill effects have not been reported following acute ingestion of large doses of oral contraceptives by young children. Overdosage may cause nausea, and withdrawal bleeding may occur in females.
NON-CONTRACEPTIVE HEALTH BENEFITS
The following non-contraceptive health benefits related to the use of oral contraceptives are supported by epidemiological studies which largely utilized oral contraceptive formulations containing doses exceeding 0.035 mg of ethinyl estradiol or 0.05 mg mestranol.
Effects on menses:
- increased menstrual cycle regularity
- decreased blood loss and decreased incidence of iron-deficiency anemia
- decreased incidence of dysmenorrhea
Effects related to inhibition of ovulation:
- decreased incidence of functional ovarian cysts
- decreased incidence of ectopic pregnancies
Effects from long-term use:
- decreased incidence of fibroadenomas and fibrocystic disease of the breast
- decreased incidence of acute pelvic inflammatory disease
- decreased incidence of endometrial cancer
- decreased incidence of ovarian cancer
Lessina Dosage and Administration
To achieve maximum contraceptive effectiveness, Lessina® (levonorgestrel and ethinyl estradiol tablets USP) must be taken exactly as directed at intervals not exceeding 24-hours.
Lessina Tablets are a monophasic preparation plus 7 inert tablets. The dosage of Lessina Tablets is one tablet daily for 21 consecutive days per menstrual cycle plus 7 white inert tablets according to the prescribed schedule. It is recommended that Lessina Tablets be taken at the same time each day, preferably after the evening meal or at bedtime. During the first cycle of medication, the patient should be instructed to take one pink Lessina Tablet daily and then 7 white inert tablets for twenty-eight (28) consecutive days, beginning on day one (1) of her menstrual cycle. (The first day of menstruation is day one.) Withdrawal bleeding usually occurs within 3 days following the last pink tablet. (If Lessina Tablets are first taken later than the first day of the first menstrual cycle of medication or postpartum, contraceptive reliance should not be placed on Lessina Tablets until after the first 7 consecutive days of administration. The possibility of ovulation and conception prior to initiation of medication should be considered.)
When switching from another oral contraceptive, Lessina Tablets should be started on the first day of bleeding following the last active tablet taken of the previous oral contraceptive.
The patient begins her next and all subsequent 28-day courses of Lessina Tablets on the same day of the week that she began her first course, following the same schedule. She begins taking her pink tablets on the next day after ingestion of the last white tablet, regardless of whether or not a menstrual period has occurred or is still in progress. Anytime a subsequent cycle of Lessina Tablets is started later than the next day, the patient should be protected by another means of contraception until she has taken a tablet daily for seven consecutive days.
If spotting or breakthrough bleeding occurs, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance, however, if the bleeding is persistent or prolonged, the patient is advised to consult her physician. Although the occurrence of pregnancy is highly unlikely if Lessina Tablets are taken according to directions, if withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (missed one or more active tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered at the time of the first missed period and appropriate diagnostic measures taken before the medication is resumed. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing the contraceptive regimen.
The risk of pregnancy increases with each active (pink) tablet missed. For additional patient instructions regarding missed pills, see the “WHAT TO DO IF YOU MISS PILLS” section in the DETAILED PATIENT LABELING below. If breakthrough bleeding occurs following missed tablets, it will usually be transient and of no consequence. If the patient misses one or more white tablets, she is still protected against pregnancy provided she begins taking pink tablets again on the proper day.
In the nonlactating mother, Lessina Tablets may be initiated postpartum, for contraception. When the tablets are administered in the postpartum period, the increased risk of thromboembolic disease associated with the postpartum period must be considered. (See CONTRAINDICATIONS, WARNINGS, and PRECAUTIONS concerning thromboembolic disease.) It is to be noted that early resumption of ovulation may occur if bromocriptine mesylate has been used for the prevention of lactation.
How is Lessina supplied
Lessina® (levonorgestrel and ethinyl estradiol tablets USP 0.1 mg/ 0.02 mg) are packaged in blister cards. Each card contains 21 pink, round, film-coated, biconvex, unscored tablets, debossed stylized b on one side and 965 on the other side, and 7 white, round, biconvex, unscored tablets, debossed stylized b on one side and 208 on the other side.
Available in cartons of 3 blister cards (NDC: 0555-9014-67).
Store at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature].
REFERENCES FURNISHED UPON REQUEST
TEVA PHARMACEUTICALS USA, INC.
North Wales, PA 19454
Rev. C 12/2021
BRIEF SUMMARY PATIENT PACKAGE INSERT
This product (like all oral contraceptives) is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
Oral contraceptives, also known as “birth-control pills” or “the pill”, are taken to prevent pregnancy, and when taken correctly, have a failure rate of less than 1% per year when used without missing any pills. The typical failure rate of large numbers of pill users is less than 3% per year when women who miss pills are included. For most women oral contraceptives are also free of serious or unpleasant side effects. However, forgetting to take pills considerably increases the chances of pregnancy.
For the majority of women, oral contraceptives can be taken safely. But there are some women who are at high risk of developing certain serious diseases that can be life-threatening or may cause temporary or permanent disability or death. The risks associated with taking oral contraceptives increase significantly if you:
- smoke
- have high blood pressure, diabetes, high cholesterol
- have or have had clotting disorders, heart attack, stroke, angina pectoris, cancer of the breast or sex organs, jaundice, or malignant or benign liver tumors.
You should not take the pill if you suspect you are pregnant or have unexplained vaginal bleeding.
Cigarette smoking increases the risk of serious adverse effects on the heart and blood vessels from oral contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes per day) and is quite marked in women over 35 years of age. Women who use oral contraceptives should not smoke.
Most side effects of the pill are not serious. The most common such effects are nausea, vomiting, bleeding between menstrual periods, weight gain, breast tenderness, and difficulty wearing contact lenses. These side effects, especially nausea and vomiting may subside within the first three months of use.
The serious side effects of the pill occur very infrequently, especially if you are in good health and are young. However, you should know that the following medical conditions have been associated with or made worse by the pill:
- Blood clots in the legs (thrombophlebitis), lungs (pulmonary embolism), stoppage or rupture of a blood vessel in the brain (stroke), blockage of blood vessels in the heart (heart attack and angina pectoris) or other organs of the body. As mentioned above, smoking increases the risk of heart attacks and strokes and subsequent serious medical consequences.
- Liver tumors, which may rupture and cause severe bleeding. A possible but not definite association has been found with the pill and liver cancer. However, liver cancers are extremely rare. The chance of developing liver cancer from using the pill is thus even rarer.
- High blood pressure, although blood pressure usually returns to normal when the pill is stopped.
The symptoms associated with these serious side effects are discussed in the detailed leaflet given to you with your supply of pills. Notify your doctor or healthcare provider if you notice any unusual physical disturbances while taking the pill. In addition, drugs such as rifampin, as well as some anticonvulsants and some antibiotics, may decrease oral contraceptive effectiveness.
There may be slight increases in the risk of breast cancer among current users of hormonal birth control pills with longer duration of use of 8 years or more.
Taking the pill provides some important non-contraceptive benefits. These include less painful menstruation, less menstrual blood loss and anemia, fewer pelvic infections, and fewer cancers of the ovary and the lining of the uterus.
Be sure to discuss any medical condition you may have with your healthcare provider. Your healthcare provider will take a medical and family history before prescribing oral contraceptives and will examine you. The physical examination may be delayed to another time if you request it and the healthcare provider believes that it is appropriate to postpone it. You should be reexamined at least once a year while taking oral contraceptives. The detailed patient information booklet gives you further information which you should read and discuss with your healthcare provider.
This product (like all oral contraceptives) is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
INSTRUCTIONS TO PATIENTS
HOW TO TAKE THE PILL
IMPORTANT POINTS TO REMEMBER
BEFORE YOU START TAKING YOUR PILLS:
- BE SURE TO READ THESE DIRECTIONS:
Before you start taking your pills.
Anytime you are not sure what to do. - THE RIGHT WAY TO TAKE THE PILL IS TO TAKE ONE PILL EVERY DAY AT THE SAME TIME.
If you miss pills you could get pregnant. This includes starting the pack late. The more pills you miss, the more likely you are to get pregnant. - MANY WOMEN HAVE SPOTTING OR LIGHT BLEEDING, OR MAY FEEL SICK TO THEIR STOMACH DURING THE FIRST 1 to 3 PACKS OF PILLS.
If you do feel sick to your stomach, do not stop taking the pill. The problem will usually go away. If it does not go away, check with your doctor or clinic. - MISSING PILLS CAN ALSO CAUSE SPOTTING OR LIGHT BLEEDING, even when you make up these missed pills.
On the days you take two pills, to make up for missed pills, you could also feel a little sick to your stomach. - IF YOU HAVE VOMITING OR DIARRHEA, for any reason, or IF YOU TAKE SOME MEDICINES, including some antibiotics, your pills may not work as well.
Use a back-up method (such as condoms, foam, or sponge) until you check with your doctor or clinic. - IF YOU HAVE TROUBLE REMEMBERING TO TAKE THE PILL, talk to your doctor or clinic about how to make pill-taking easier or about using another method of birth control.
- IF YOU HAVE ANY QUESTIONS OR ARE UNSURE ABOUT THE INFORMATION IN THIS LEAFLET, call your doctor or clinic.
BEFORE YOU START TAKING YOUR PILLS
- DECIDE WHAT TIME OF DAY YOU WANT TO TAKE YOUR PILL.
It is important to take it at about the same time every day. - LOOK AT YOUR PILL PACK TO SEE IF IT HAS 28 PILLS:
The pill pack has 21 (pink) “active” pills (with hormones) to take for three weeks, followed by 1 week of reminder pills (white) (without hormones).
Refer to the sample of the blister card below:
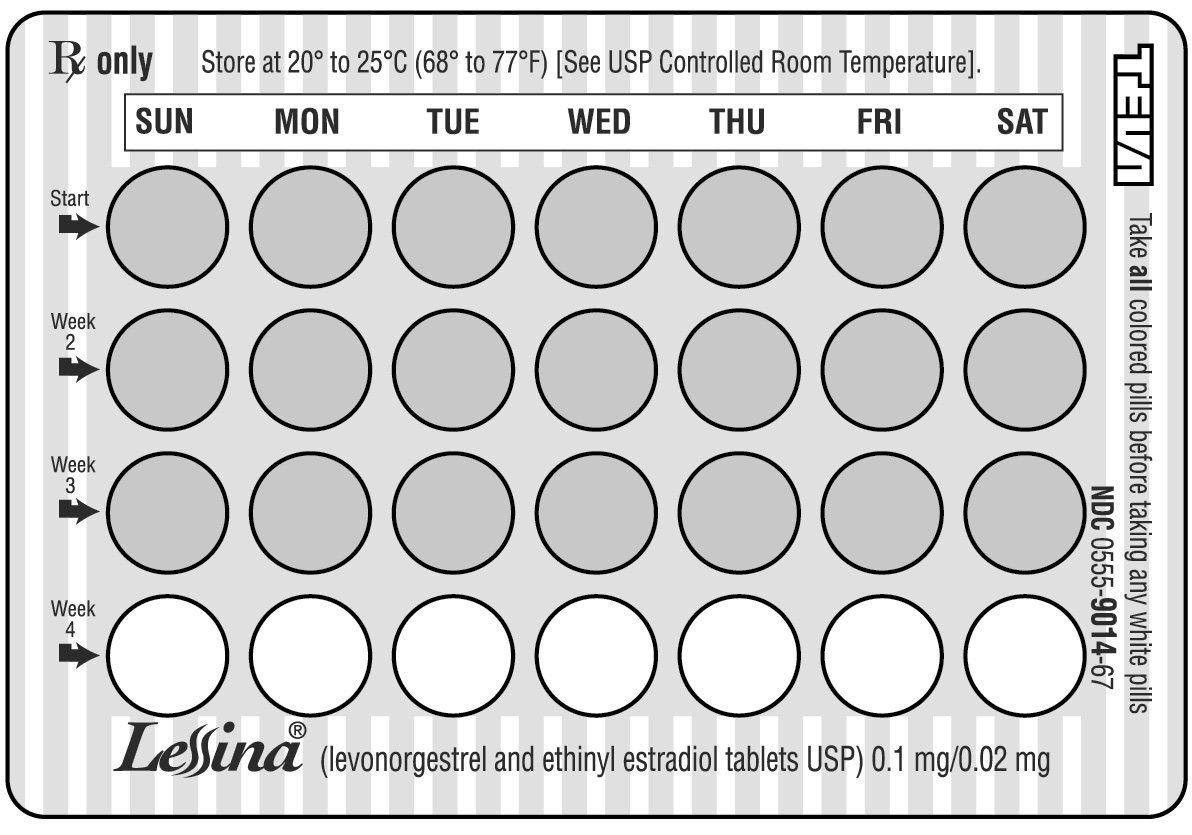
- ALSO FIND:
1) where on the pack to start taking pills,
2) in what order to take the pills - BE SURE YOU HAVE READY AT ALL TIMES:
ANOTHER KIND OF BIRTH CONTROL (such as condoms, foam or sponge) to use as a back-up in case you miss pills.
AN EXTRA, FULL PILL PACK
WHEN TO START THE FIRST PACK OF PILLS
You have a choice for which day to start taking your first pack of pills. Decide with your doctor or clinic which is the best day for you. Pick a time of day which will be easy to remember.
DAY 1 START:
- Take the first (pink) “active” pill of the first pack during the first 24 hours of your period.
- You will not need to use a back-up method of birth control, since you are starting the pill at the beginning of your period.
SUNDAY START:
- Take the first (pink) “active” pill of the first pack on the Sunday after your period starts, even if you are still bleeding. If your period begins on Sunday, start the pack that same day.
- Use another method of birth control as a back-up method if you have sex anytime from the Sunday you start your first pack until the next Sunday (7 days). Condoms, foam, or the sponge are good back-up methods of birth control.
WHAT TO DO DURING THE MONTH
-
TAKE ONE PILL AT THE SAME TIME EVERY DAY UNTIL THE PACK IS EMPTY
Do not skip pills even if you are spotting or bleeding between monthly periods or feel sick to your stomach (nausea).
Do not skip pills even if you do not have sex very often. -
WHEN YOU FINISH A PACK OR SWITCH YOUR BRAND OF PILLS:
Start the next pack on the day after your last (white) “reminder” pill. Do not wait any days between packs.
WHAT TO DO IF YOU MISS PILLS
If you MISS 1 (pink) “active” pill:
- Take it as soon as you remember. Take the next pill at your regular time. This means you may take two pills in one day.
- You do not need to use a back-up birth control method if you have sex.
If you MISS 2 (pink) “active” pills in a row in WEEK 1 OR WEEK 2 of your pack:
- Take two pills on the day you remember and two pills the next day.
- Then take one pill a day until you finish the pack.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as a back-up for those 7 days.
If you MISS 2 (pink) “active” pills in a row in THE 3rd WEEK:
-
If you are a Day 1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking one pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day. - You may not have your period this month but this is expected. However, if you miss your period two months in a row, call your doctor or clinic because you might be pregnant.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as a back-up for those 7 days.
If you MISS 3 OR MORE (pink) “active” pills in a row (during the first 3 weeks):
-
If you are a Day 1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking one pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day. - You may not have your period this month but this is expected. However, if you miss your period two months in a row, call your doctor or clinic because you might be pregnant.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as a back-up for those 7 days.
A REMINDER FOR THOSE ON 28-DAY PACKS:
If you forget any of the 7 (white) “reminder” pills in Week 4:
THROW AWAY the pills you missed.
Keep taking one pill each day until the pack is empty.
You do not need a back-up method.
FINALLY, IF YOU ARE STILL NOT SURE WHAT TO DO ABOUT THE PILLS YOU HAVE MISSED:
Use a BACK-UP METHOD anytime you have sex.
KEEP TAKING ONE ACTIVE PILL EACH DAY until you can reach your doctor or clinic.
Store at 20° to 25°C (68° to 77°F).
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
DETAILED PATIENT PACKAGE INSERT
This product (like all oral contraceptives) is intended to prevent pregnancy. It does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
INTRODUCTION
Any woman who considers using oral contraceptives (the “birth-control pill” or “the pill”) should understand the benefits and risks of using this form of birth control. This leaflet will give you much of the information you will need to make this decision and will also help you determine if you are at risk of developing any of the serious side effects of the pill. It will tell you how to use the pill properly so that it will be as effective as possible. However, this leaflet is not a replacement for a careful discussion between you and your healthcare provider. You should discuss the information provided in this leaflet with him or her, both when you first start taking the pill and during your revisits. You should also follow your healthcare provider’s advice with regard to regular check-ups while you are on the pill.
EFFECTIVENESS OF ORAL CONTRACEPTIVES
Oral contraceptives or “birth-control pills” or “the pill” are used to prevent pregnancy and are more effective than other nonsurgical methods of birth control. When they are taken correctly, the chance of becoming pregnant is less than 1% when used perfectly, without missing any pills. Typical failure rates are less than 3% per year. The chance of becoming pregnant increases with each missed pill during a menstrual cycle.
In comparison, typical failure rates for other nonsurgical methods of birth control during the first year of use are as follows:
|
Percentage of women experiencing an unintended pregnancy during the first year of typical use and first year of perfect use of contraception and the percentage continuing use at the end of the first year. United States. |
|||
|
% of Women Experiencing |
% of Women Continuing Use at One Year3 |
||
|
Method (1) |
Typical Use1 (2) |
Perfect Use2 (3) |
|
|
Chance4 |
85 |
85 | |
|
Spermicides5 |
26 |
6 |
40 |
|
Periodic abstinence |
25 |
63 |
|
|
Calendar |
9 | ||
|
Ovulation method |
3 | ||
|
Sympto-thermal6 |
2 | ||
|
Post ovulation |
1 | ||
|
Withdrawal |
19 |
4 | |
|
Cap7 | |||
|
Parous women |
40 |
26 |
42 |
|
Nulliparous women |
20 |
9 |
56 |
|
Sponge | |||
|
Parous women |
40 |
20 |
42 |
|
Nulliparous women |
20 |
9 |
56 |
|
Diaphragm7 |
20 |
6 |
56 |
|
Condom8 | |||
|
Female (Reality) |
21 |
5 |
56 |
|
Male |
14 |
3 |
61 |
|
Pill |
5 |
71 |
|
|
Progestin only |
0.5 | ||
|
Combined |
0.1 | ||
|
IUD | |||
|
Progesterone T |
2 |
1.5 |
81 |
|
Copper T 380A |
0.8 |
0.6 |
78 |
|
Lng 20 |
0.1 |
0.1 |
81 |
|
Depo Provera |
0.3 |
0.3 |
70 |
|
Norplant and Norplant-2 |
0.05 |
0.05 |
88 |
|
Female sterilization |
0.5 |
0.5 |
100 |
|
Male sterilization |
0.15 |
0.1 |
100 |
|
Source: Trussell J, Contraceptive efficacy. In Hatcher RA, Trussell J, Stewart F, Cates W, Stewart GK, Kowal D, Guest F, Contraceptive Technology: Seventeenth Revised Edition. New York NY: Irvington Publishers, 1998, in press. |
|||
1. Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
2. Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
3. Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year.
4. The percentages becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percentage who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether.
5. Foams, creams, gels, vaginal suppositories, vaginal film.
6. Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the postovulatory phases.
7. With spermicidal cream or jelly.
8. Without spermicides.
WHO SHOULD NOT TAKE ORAL CONTRACEPTIVES
Cigarette smoking increases the risk of serious adverse effects on the heart and blood vessels from oral contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes per day) and is quite marked in women over 35 years of age. Women who use oral contraceptives should not smoke.
Some women should not use the pill. For example, you should not take the pill if you are pregnant or think you may be pregnant. You should also not use the pill if you have had any of the following conditions:
- A history of heart attack or stroke
- Blood clots in the legs (thrombophlebitis), lungs (pulmonary embolism), or eyes
- A history of blood clots in the deep veins of your legs
- Chest pain (angina pectoris)
- Known or suspected breast cancer or cancer of the lining of the uterus, cervix or vagina
- Unexplained vaginal bleeding (until a diagnosis is reached by your doctor)
- Yellowing of the whites of the eyes or of the skin (jaundice) during pregnancy or during previous use of the pill
- Liver tumor (benign or cancerous)
- Take any Hepatitis C drug combination containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir. This may increase levels of the liver enzyme “alanine aminotransferase” (ALT) in the blood.
- Known or suspected pregnancy
Tell your healthcare provider if you have ever had any of these conditions. Your healthcare provider can recommend another method of birth control.
OTHER CONSIDERATIONS BEFORE TAKING ORAL CONTRACEPTIVES
Tell your healthcare provider if you or any family member has ever had:
- Breast nodules, fibrocystic disease of the breast, an abnormal breast X-ray or mammogram
- Diabetes
- Elevated cholesterol or triglycerides
- High blood pressure
- Migraine or other headaches or epilepsy
- Mental depression
- Gallbladder, heart or kidney disease
- History of scanty or irregular menstrual periods
Women with any of these conditions should be checked often by their healthcare provider if they choose to use oral contraceptives.
Also, be sure to inform your doctor or healthcare provider if you smoke or are on any medications.
RISKS OF TAKING ORAL CONTRACEPTIVES
-
Risk of developing blood clots
Blood clots and blockage of blood vessels are the most serious side effects of taking oral contraceptives and can be fatal. In particular, a clot in the legs can cause thrombophlebitis and a clot that travels to the lungs can cause sudden blocking of the vessel carrying blood to the lungs. Rarely, clots occur in the blood vessels of the eye and may cause blindness, double vision, or impaired vision.
If you take oral contraceptives and need elective surgery, need to stay in bed for a prolonged illness or have recently delivered a baby, you may be at risk of developing blood clots. You should consult your doctor about stopping oral contraceptives three to four weeks before surgery and not taking oral contraceptives for two weeks after surgery or during bed rest. You should also not take oral contraceptives soon after delivery of a baby or a mid-trimester pregnancy termination. It is advisable to wait for at least four weeks after delivery if you are not breast-feeding. If you are breast-feeding, you should wait until you have weaned your child before using the pill. (See also the section on breast-feeding in “GENERAL PRECAUTIONS”.) -
Heart attacks and strokes
Oral contraceptives may increase the tendency to develop strokes (stoppage or rupture of blood vessels in the brain) and angina pectoris and heart attacks (blockage of blood vessels in the heart). Any of these conditions can cause death or serious disability.
Smoking greatly increases the possibility of suffering heart attacks and strokes. Furthermore, smoking and the use of oral contraceptives greatly increase the chances of developing and dying of heart disease. -
Gallbladder disease
Oral contraceptive users probably have a greater risk than nonusers of having gallbladder disease, although this risk may be related to pills containing high doses of estrogens. -
Liver tumors
In rare cases, oral contraceptives can cause benign but dangerous liver tumors. These benign liver tumors can rupture and cause fatal internal bleeding. In addition, a possible but not definite association has been found with the pill and liver cancers in two studies, in which a few women who developed these very rare cancers were found to have used oral contraceptives for long periods. However, liver cancers are extremely rare. The chance of developing liver cancer from using the pill is thus even rarer. -
Risk of Cancer
It is not known if hormonal birth control pills causes breast cancer. Some studies, but not all, suggest that there could be a slight increase in the risk of breast cancer among current users with longer duration of use.If you have breast cancer now, or have had it in the past, do not use hormonal birth control because some breast cancers are sensitive to hormones.
Some studies have found an increase in the incidence of cancer of the cervix in women who use oral contraceptives. However, this finding may be related to factors other than the use of oral contraceptives.
|
Adapted from H.W. Ory, Family Planning Perspectives 15:57-63, 1983. 1. Deaths are birth related 2. Deaths are method related |
||||||
| Method of Control and Outcome | 15 to 19 | 20 to 24 | 25 to 29 | 30 to 34 | 35 to 39 | 40 to 44 |
| No fertility | 7 | 7.4 | 9.1 | 14.8 | 25.7 | 28.2 |
| Oral contraceptives | 0.3 | 0.5 | 0.9 | 1.9 | 13.8 | 31.6 |
| Oral contraceptives | 2.2 | 3.4 | 6.6 | 13.5 | 51.1 | 117.2 |
| IUD2 | 0.8 | 0.8 | 1 | 1 | 1.4 | 1.4 |
| Condom1 | 1.1 | 1.6 | 0.7 | 0.2 | 0.3 | 0.4 |
| Diaphragm/ | 1.9 | 1.2 | 1.2 | 1.3 | 2.2 | 2.8 |
| Periodic | 2.5 | 1.6 | 1.6 | 1.7 | 2.9 | 3.6 |
In the above table, the risk of death from any birth-control method is less than the risk of childbirth, except for oral contraceptive users over the age of 35 who smoke and pill users over the age of 40 even if they do not smoke. It can be seen in the table that for women aged 15 to 39, the risk of death was highest with pregnancy (7-26 deaths per 100,000 women, depending on age). Among pill users who do not smoke, the risk of death was always lower than that associated with pregnancy for any age group, except for those women over the age of 40, when the risk increases to 32 deaths per 100,000 women, compared to 28 associated with pregnancy at that age. However, for pill users who smoke and are over the age of 35, the estimated number of deaths exceeds those for other methods of birth control. If a woman is over the age of 40 and smokes, her estimated risk of death is four times higher (117/100,000 women) than the estimated risk associated with pregnancy (28/100,000 women) in that age group.
The suggestion that women over 40 who do not smoke should not take oral contraceptives is based on information from older high-dose pills and on less-selective use of pills than is practiced today. An Advisory Committee of the FDA discussed this issue in 1989 and recommended that the benefits of oral contraceptive use by healthy, non-smoking women over 40 years of age may outweigh the possible risks. However, all women, especially older women, are cautioned to use the lowest-dose pill that is effective.
WARNING SIGNALS
If any of these adverse effects occur while you are taking oral contraceptives, call your doctor immediately:
- Sharp chest pain, coughing of blood, or sudden shortness of breath (indicating a possible clot in the lung)
- Pain in the calf (indicating a possible clot in the leg)
- Crushing chest pain or heaviness in the chest (indicating a possible heart attack)
- Sudden severe headache or vomiting, dizziness or fainting, disturbances of vision or speech, weakness, or numbness in an arm or leg (indicating a possible stroke)
- Sudden partial or complete loss of vision (indicating a possible clot in the eye)
- Breast lumps (indicating possible breast cancer or fibrocystic disease of the breast; ask your doctor or healthcare provider to show you how to examine your breasts)
- Severe pain or tenderness in the stomach area (indicating a possibly ruptured liver tumor)
- Difficulty in sleeping, weakness, lack of energy, fatigue, or change in mood (possibly indicating severe depression)
- Jaundice or a yellowing of the skin or eyeballs, accompanied frequently by fever, fatigue, loss of appetite, dark-colored urine, or light-colored bowel movements (indicating possible liver problems)
SIDE EFFECTS OF ORAL CONTRACEPTIVES
-
Vaginal bleeding
Irregular vaginal bleeding or spotting may occur while you are taking the pills. Irregular bleeding may vary from slight staining between menstrual periods to breakthrough bleeding, which is a flow much like a regular period. Irregular bleeding occurs most often during the first few months of oral contraceptive use, but may also occur after you have been taking the pill for some time. Such bleeding may be temporary and usually does not indicate any serious problems. It is important to continue taking your pills on schedule. If the bleeding occurs in more than one cycle or lasts for more than a few days, talk to your doctor or healthcare provider. -
Contact lenses
If you wear contact lenses and notice a change in vision or an inability to wear your lenses, contact your doctor or healthcare provider. -
Fluid retention
Oral contraceptives may cause edema (fluid retention) with swelling of the fingers or ankles and may raise your blood pressure. If you experience fluid retention, contact your doctor or healthcare provider. -
Melasma
A spotty darkening of the skin is possible, particularly of the face. -
Other side effects
Other side effects may include change in appetite, headache, nervousness, depression, dizziness, loss of scalp hair, rash, and vaginal infections.
If any of these side effects bother you, call your doctor or healthcare provider.
GENERAL PRECAUTIONS
-
Missed periods and use of oral contraceptives before or during early pregnancy
There may be times when you may not menstruate regularly after you have completed taking a cycle of pills. If you have taken your pills regularly and miss one menstrual period, continue taking your pills for the next cycle but be sure to inform your healthcare provider before doing so. If you have not taken the pills daily as instructed and missed a menstrual period, or if you missed two consecutive menstrual periods, you may be pregnant. Check with your healthcare provider immediately to determine whether you are pregnant. Do not continue to take oral contraceptives until you are sure you are not pregnant, but continue to use another method of contraception.
There is no conclusive evidence that oral contraceptive use is associated with an increase in birth defects when taken inadvertently during early pregnancy. Previously, a few studies had reported that oral contraceptives might be associated with birth defects, but these studies have not been confirmed. Nevertheless, oral contraceptives or any other drugs should not be used during pregnancy unless clearly necessary and prescribed by your doctor. You should check with your doctor about risks to your unborn child of any medication taken during pregnancy.
-
While breast-feeding
If you are breast-feeding, consult your doctor before starting oral contraceptives. Some of the drug will be passed on to the child in the milk. A few adverse effects on the child have been reported, including yellowing of the skin (jaundice) and breast enlargement. In addition, oral contraceptives may decrease the amount and quality of your milk. If possible, do not use oral contraceptives while breast-feeding. You should use another method of contraception since breast-feeding provides only partial protection from becoming pregnant, and this partial protection decreases significantly as you breast-feed for longer periods of time. You should consider starting oral contraceptives only after you have weaned your child completely.
-
Laboratory tests
If you are scheduled for any laboratory tests, tell your doctor you are taking birth-control pills. Certain blood tests may be affected by birth-control pills.
-
Drug Interactions
Certain drugs may interact with birth-control pills to make them less effective in preventing pregnancy or cause an increase in breakthrough bleeding. Such drugs include rifampin, drugs used for epilepsy such as barbiturates (for example, phenobarbital) and phenytoin (Dilantin is one brand of this drug), phenylbutazone (Butazolidin is one brand) and possibly certain antibiotics. You may need to use an additional method of contraception during any cycle in which you take drugs that can make oral contraceptives less effective.
-
Sexually transmitted diseases
This product (like all oral contraceptives) is intended to prevent pregnancy. It does not protect against transmission of HIV (AIDS) and other sexually transmitted diseases such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
HOW TO TAKE THE PILL
IMPORTANT POINTS TO REMEMBER
BEFORE YOU START TAKING YOUR PILLS:
- BE SURE TO READ THESE DIRECTIONS:
Before you start taking your pills.
Anytime you are not sure what to do. - THE RIGHT WAY TO TAKE THE PILL IS TO TAKE ONE PILL EVERY DAY AT THE SAME TIME.
If you miss pills you could get pregnant. This includes starting the pack late. The more pills you miss, the more likely you are to get pregnant. - MANY WOMEN HAVE SPOTTING OR LIGHT BLEEDING, OR MAY FEEL SICK TO THEIR STOMACH DURING THE FIRST 1 to 3 PACKS OF PILLS.
If you do feel sick to your stomach, do not stop taking the pill. The problem will usually go away. If it does not go away, check with your doctor or clinic. - MISSING PILLS CAN ALSO CAUSE SPOTTING OR LIGHT BLEEDING, even when you make up these missed pills.
On the days you take two pills, to make up for missed pills, you could also feel a little sick to your stomach. - IF YOU HAVE VOMITING OR DIARRHEA, for any reason, or IF YOU TAKE SOME MEDICINES, including some antibiotics, your pills may not work as well.
Use a back-up method (such as condoms, foam, or sponge) until you check with your doctor or clinic. - IF YOU HAVE TROUBLE REMEMBERING TO TAKE THE PILL, talk to your doctor or clinic about how to make pill-taking easier or about using another method of birth control.
- IF YOU HAVE ANY QUESTIONS OR ARE UNSURE ABOUT THE INFORMATION IN THIS LEAFLET, call your doctor or clinic.
BEFORE YOU START TAKING YOUR PILLS
- DECIDE WHAT TIME OF DAY YOU WANT TO TAKE YOUR PILL.
It is important to take it at about the same time every day. - LOOK AT YOUR PILL PACK TO SEE IF IT HAS 28 PILLS:
The pill pack has 21 (pink) “active” pills (with hormones) to take for three weeks, followed by 1 week of reminder pills (white) (without hormones).
Refer to the sample of the blister card below:
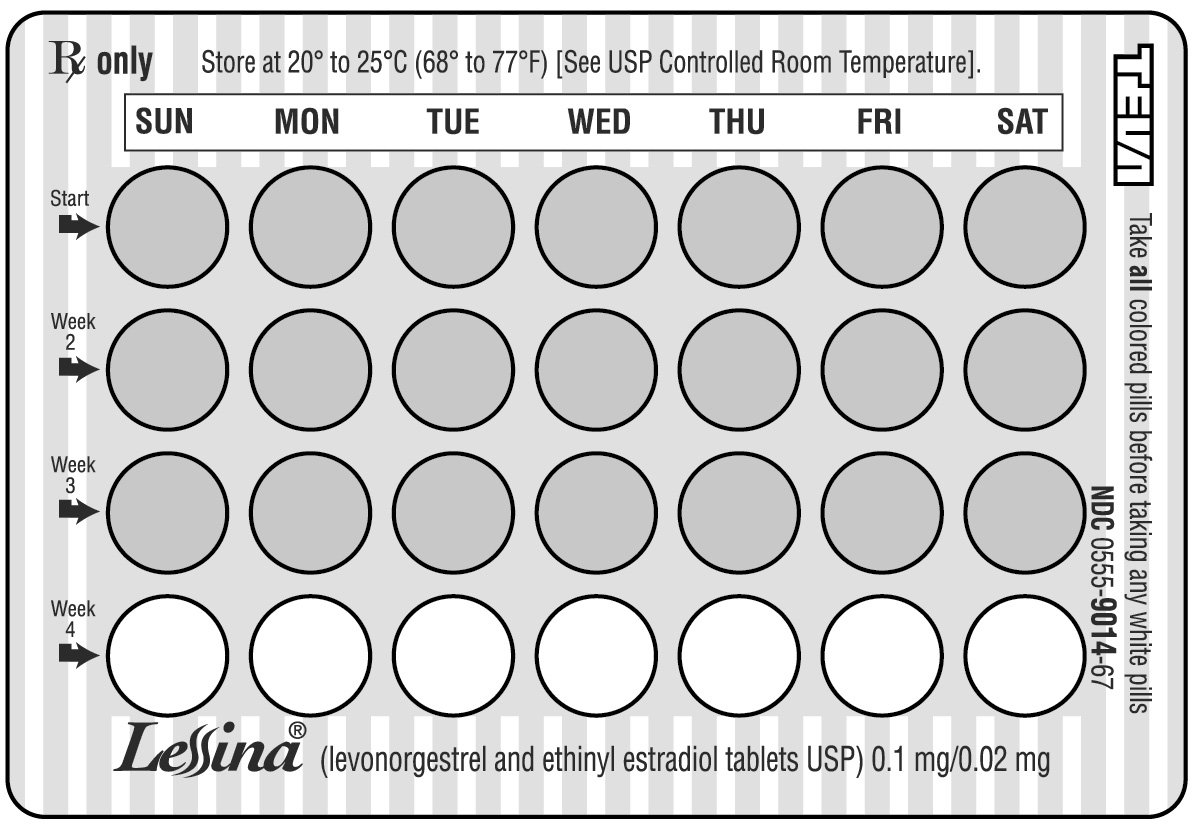
- ALSO FIND:
1) where on the pack to start taking pills,
2) in what order to take the pills - BE SURE YOU HAVE READY AT ALL TIMES:
ANOTHER KIND OF BIRTH CONTROL (such as condoms, foam or sponge) to use as a back-up in case you miss pills.
AN EXTRA, FULL PILL PACK
WHEN TO START THE FIRST PACK OF PILLS
You have a choice for which day to start taking your first pack of pills. Decide with your doctor or clinic which is the best day for you. Pick a time of day which will be easy to remember.
DAY 1 START:
- Take the first (pink) “active” pill of the first pack during the first 24 hours of your period.
- You will not need to use a back-up method of birth control, since you are starting the pill at the beginning of your period.
SUNDAY START:
- Take the first (pink) “active” pill of the first pack on the Sunday after your period starts, even if you are still bleeding. If your period begins on Sunday, start the pack that same day.
- Use another method of birth control as a back-up method if you have sex anytime from the Sunday you start your first pack until the next Sunday (7 days). Condoms, foam, or the sponge are good back-up methods of birth control.
WHAT TO DO DURING THE MONTH
-
TAKE ONE PILL AT THE SAME TIME EVERY DAY UNTIL THE PACK IS EMPTY
Do not skip pills if you are spotting or bleeding between monthly periods or feel sick to your stomach (nausea).
Do not skip pills even if you do not have sex very often. -
WHEN YOU FINISH A PACK OR SWITCH YOUR BRAND OF PILLS:
Start the next pack on the day after your last (white) “reminder” pill. Do not wait any days between packs.
WHAT TO DO IF YOU MISS PILLS
If you MISS 1 (pink) “active” pill:
- Take it as soon as you remember. Take the next pill at your regular time. This means you may take two pills in one day.
- You do not need to use a back-up birth control method if you have sex.
If you MISS 2 (pink) “active” pills in a row in WEEK 1 OR WEEK 2 of your pack:
- Take two pills on the day you remember and two pills the next day.
- Then take one pill a day until you finish the pack.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as back-up for those 7 days.
If you MISS 2 (pink) “active” pills in a row in THE 3rd WEEK:
-
If you are a Day 1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking one pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day. - You may not have your period this month but this is expected. However, if you miss your period two months in a row, call your doctor or clinic because you might be pregnant.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as a back-up for those 7 days.
If you MISS 3 OR MORE (pink) “active” pills in a row (during the first 3 weeks):
-
If you are a Day 1 Starter:
THROW OUT the rest of the pill pack and start a new pack that same day.
If you are a Sunday Starter:
Keep taking one pill every day until Sunday. On Sunday, THROW OUT the rest of the pack and start a new pack of pills that same day. - You may not have your period this month but this is expected. However, if you miss your period two months in a row, call your doctor or clinic because you might be pregnant.
- You MAY BECOME PREGNANT if you have sex in the 7 days after you miss pills. You MUST use another birth control method (such as condoms, foam, or sponge) as a back-up for those 7 days.
A REMINDER FOR THOSE ON 28-DAY PACKS
If you forget any of the 7 (white) “reminder” pills in Week 4:
THROW AWAY the pills you missed.
Keep taking one pill each day until the pack is empty.
You do not need a back-up method.
FINALLY, IF YOU ARE STILL NOT SURE WHAT TO DO ABOUT THE PILLS YOU HAVE MISSED
Use a BACK-UP METHOD anytime you have sex.
KEEP TAKING ONE ACTIVE PILL EACH DAY until you can reach your doctor or clinic.
PREGNANCY DUE TO PILL FAILURE
The incidence of pill failure resulting in pregnancy is approximately less than 1.0% if taken every day as directed, but more typical failure rates are less than 3.0%. If failure does occur, the risk to the fetus is minimal.
PREGNANCY AFTER STOPPING THE PILL
There may be some delay in becoming pregnant after you stop using oral contraceptives, especially if you had irregular menstrual cycles before you used oral contraceptives. It may be advisable to postpone conception until you begin menstruating regularly once you have stopped taking the pill and desire pregnancy.
There does not appear to be any increase in birth defects in newborn babies when pregnancy occurs soon after stopping the pill.
OVERDOSAGE
Serious ill effects have not been reported following ingestion of large doses of oral contraceptives by young children. Overdosage may cause nausea and withdrawal bleeding in females. In case of overdosage, contact your healthcare provider or pharmacist.
OTHER INFORMATION
Your healthcare provider will take a medical and family history before prescribing oral contraceptives and will examine you. The physical examination may be delayed to another time if you request it and the healthcare provider believes that it is appropriate to postpone it. You should be re-examined at least once a year. Be sure to inform your healthcare provider if there is a family history of any of the conditions listed previously in this leaflet. Be sure to keep all appointments with your healthcare provider, because this is a time to determine if there are early signs of side effects of oral contraceptive use.
Do not use the drug for any condition other than the one for which it was prescribed. This drug has been prescribed specifically for you; do not give it to others who may want birth-control pills.
HEALTH BENEFITS FROM ORAL CONTRACEPTIVES
In addition to preventing pregnancy, use of oral contraceptives may provide certain benefits. They are:
- Menstrual cycles may become more regular
- Blood flow during menstruation may be lighter and less iron may be lost. Therefore, anemia due to iron deficiency is less likely to occur.
- Pain or other symptoms during menstruation may be encountered less frequently
- Ovarian cysts may occur less frequently
- Ectopic (tubal) pregnancy may occur less frequently
- Noncancerous cysts or lumps in the breast may occur less frequently
- Acute pelvic inflammatory disease may occur less frequently
- Oral contraceptive use may provide some protection against developing two forms of cancer: cancer of the ovaries and cancer of the lining of the uterus.
If you want more information about birth-control pills, ask your doctor or pharmacist. They have a more technical leaflet called the Prescribing Information which you may wish to read.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Store at 20° to 25°C (68° to 77°F).
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
All brand names listed are the registered trademarks of their respective owners and are not trademarks of Teva Pharmaceuticals USA.
TEVA PHARMACEUTICALS USA, INC.
North Wales, PA 19454
Rev. C 12/2021
Package/Label Display Panel
NDC 0555-9014-67
3 Blister Cards, 28 Tablets Each
Lessina®
(levonorgestrel and
ethinyl estradiol tablets USP)
0.1 mg/0.02 mg
Rx only
THIS PRODUCT (LIKE ALL ORAL CONTRACEPTIVES) IS INTENDED TO
PREVENT PREGNANCY. IT DOES NOT PROTECT AGAINST HIV
INFECTION (AIDS) AND OTHER SEXUALLY TRANSMITTED DISEASES.
Contains 3 blister cards, each containing 28 tablets, 21 pink tablets,
each containing 0.1 mg levonorgestrel, USP and 0.02 mg ethinyl
estradiol, USP; and 7 white inert tablets.
SHAPING
WOMEN’S HEALTH®


| LESSINA
levonorgestrel and ethinyl estradiol kit |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Teva Pharmaceuticals USA, Inc. (001627975) |
More about Lessina (ethinyl estradiol / levonorgestrel)
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (133)
- Drug images
- Side effects
- Dosage information
- During pregnancy
- Drug class: contraceptives
- En español
Patient resources
Professional resources
Other brands
Vienva, Aviane, Lutera, Seasonique, ... +47 more

