Dipentocaine Cream Prescribing Information
Package insert / product label
Generic name: diclofenac sodium, gabapentin, and lidocaine
Dosage form: cream, compounding kit
Dipentocaine Cream Description
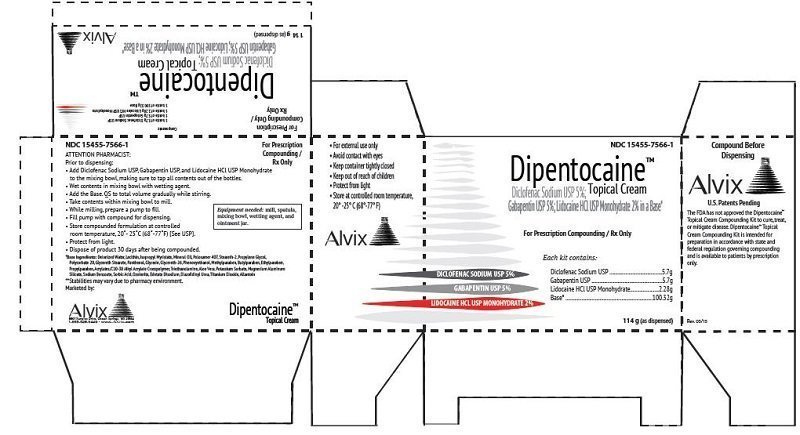
Diclofenac 5%, Gabapentin 5%, Lidocaine HCl 2%
For Pharmacy Prescription Compounding Only / Rx Only
For Topical Use Only
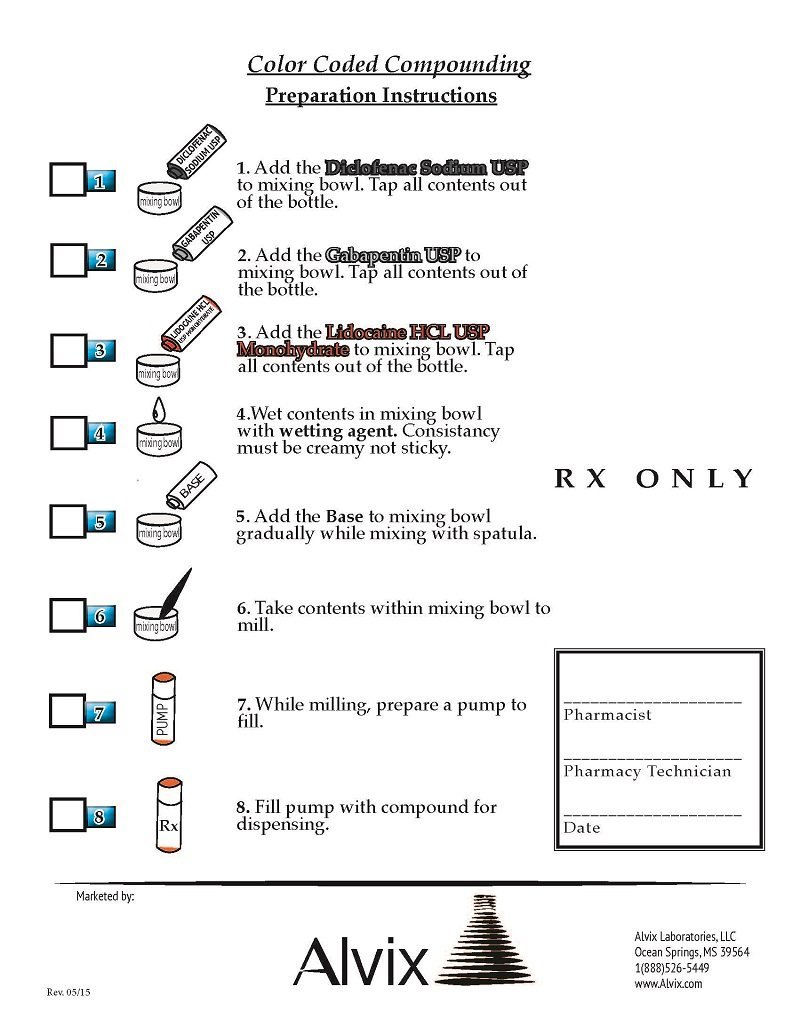
Each Dipentocaine™ Topical Cream Pharmacy Compounding Kit provides 5.7 grams of Diclofenac Sodium USP, 5.7 grams of Gabapentin USP, 2.28 grams of Lidocaine HCL USP, and 100.32 grams of Base.
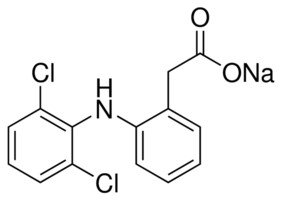
Diclofenac Sodium:
CAS Number 15307-79-6
2-[(2,6-Dichlorophenyl)amino]benzeneacetic acid sodium salt
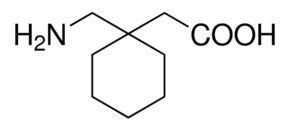
Gabapentin:
CAS Number 60142-96-3
1-(Aminomethyl)-cyclohexaneacetic acid, Neurontin
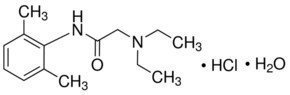
Licocaine Hydrochloride Anhydrous:
CAS Number 6108-05-0
2-Diethylamino-N-(2,6-dimethylphenyl)acetamide hydrochloride monohydrate, Lignocaine hydrochloride monohydrate, Xylocaine hydrochloride monohydrate
Warnings
Diclofenac jSodium:
Pictogram



Signal word: Danger
Hazard statement(s):
H301 Toxic if swallowed.
Precautionary statement(s):
P264 Wash skin thoroughly after handling.
P270 Do not eat, drink or smoke when using this product.
P301 + P310 IF SWALLOWED: Immediately call a POISON CENTER or doctor/ physician.
P321 Specific treatment (see supplemental first aid instructions on this label).
P330 Rinse mouth. P405 Store locked up.
P501 Dispose of contents/ container to an approved waste disposal plant.
Gabapentin:
Pictograms:


Signal word: Danger
Hazard statement(s):
H315 Causes skin irritation.
H319 Causes serious eye irritation.
H335 May cause respiratory irritation.
H360 May damage fertility or the unborn child.
Precautionary statement(s):
P201 Obtain special instructions before use.
P202 Do not handle until all safety precautions have been read and understood.
P261 Avoid breathing dust/ fume/ gas/ mist/ vapours/ spray.
P264 Wash skin thoroughly after handling.
P271 Use only outdoors or in a well-ventilated area.
P280 Wear protective gloves/ eye protection/ face protection.
P302 + P352 IF ON SKIN: Wash with plenty of soap and water.
P304 + P340 IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
P305 + P351 + P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P308 + P313 IF exposed or concerned: Get medical advice/ attention.
P321 Specific treatment (see supplemental first aid instructions on this label).
P332 + P313 If skin irritation occurs: Get medical advice/ attention.
P337 + P313 If eye irritation persists: Get medical advice/ attention.
P362 Take off contaminated clothing and wash before reuse.
P403 + P233 Store in a well-ventilated place. Keep container tightly closed. P405 Store locked up. P501 Dispose of contents/ container to an approved waste disposal plant.
Lidocaine Hydrochloride Anhydrous:
Pictogram

Signal word: Danger
Hazard statement(s):
H301 Toxic if swallowed.
Precautionary statement(s):
P264 Wash skin thoroughly after handling.
P270 Do not eat, drink or smoke when using this product.
P301 + P310 IF SWALLOWED: Immediately call a POISON CENTER or doctor/ physician.
P321 Specific treatment (see supplemental first aid instructions on this label). P330 Rinse mouth.
P405 Store locked up.
P501 Dispose of contents/ container to an approved waste disposal plant.
How is Dipentocaine Cream supplied
Kit Components: 5.7 g of Diclofenac Sodium, 5.7 g of Gabapentin, 2.28 g of Lidocaine Hydrochloride Anhydrous, and 100.32 g TDC Max Cream Base.
Equipment needed: mill, spatula, mixing bowl, and Topi-Pump™.
| SIZE | 114 grams |
| NDC # | 15455-7566-1 |
| Dichlofenac Sodium, USP | 5.7 grams |
| Gabapentin, USP | 5.7 grams |
| Lidocaine HCL, USP | 2.28 grams |
| Base | 100.32 grams |
For external use only. Avoid contact with eyes. Keep container tightly closed. Keep out of reach of children. Protect from light. Dispose of product after 30 days of being compounded.
The FDA has not approved Alvix-Dipentocaine™ Topical Cream Compounding Kit to cure, treat, or mitigate disease. Alvix-Dipentocaine™ Topical Cream intended for preparation in accordance with state and federal regulation governing compounding and is available to patients by prescription only.
Certificate of analysis on file
Rx ONLY
Marketed by:
Alvix Laboratories, LLC
1(888) 526-5449
Ocean Springs, MS 39564

Storage and Handling
Prior to compounding, store Alvix- Dipentocaine™ Topical Cream Compounding Kit at room temperature between 20 - 25 degrees C (68 - 77 degrees F). Protect from light.
| DIPENTOCAINE TOPICAL CREAM COMPOUNDING KIT
diclofenac sodium, gabapentin, and lidocaine hydrochloride kit |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Alvix Laboratories, LLC (962445925) |