The originating document has been archived. We cannot confirm the completeness, accuracy, or currency of the content.
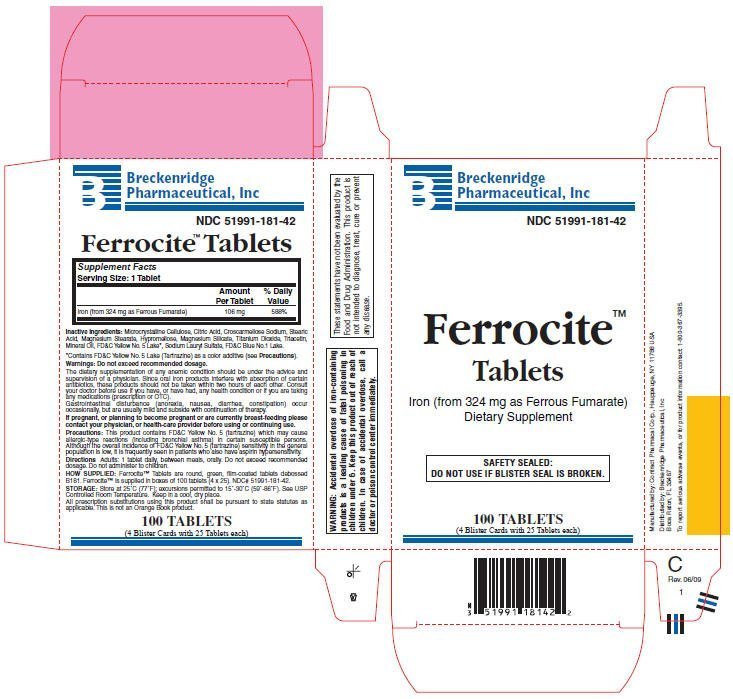
Ferrocite
Dosage form: tablet, film coated
Ingredients: Ferrous Fumarate 106mg
Labeler: Breckenridge Pharmaceutical, Inc.
NDC code: 51991-181
| Supplement Facts
Serving Size: 1 Tablet |
||
|---|---|---|
| Amount Per Tablet | % Daily Value | |
| Iron (from 324 mg as Ferrous Fumarate) | 106 mg | 588% |
Microcrystalline Cellulose, Citric Acid, Croscarmellose Sodium, Stearic Acid, Magnesium Stearate, Hypromellose, Magnesium Silicate, Titanium Dioxide, Triacetin, Mineral Oil, FD&C Yellow No. 5 Lake, Sodium Lauryl Sulfate, FD&C Blue No.1 Lake.
- Contains FD&C Yellow No. 5 Lake (Tartrazine) as a color additive (see Precautions).
Do not exceed recommended dosage.
The dietary supplementation of any anemic condition should be under the advice and supervision of a physician. Since oral iron products interfere with absorption of certain antibiotics, these products should not be taken within two hours of each other. Consult your doctor before use if you have, or have had, any health condition or if you are taking any medications (prescription or OTC).
Gastrointestinal disturbance (anorexia, nausea, diarrhea, constipation) occur occasionally, but are usually mild and subside with continuation of therapy.
If pregnant, or planning to become pregnant or are currently breast-feeding please contact your physician, or health-care provider before using or continuing use.
This product contains FD&C Yellow No. 5 (tartrazine) which may cause allergic-type reactions (including bronchial asthma) in certain susceptible persons. Although the overall incidence of FD&C Yellow No. 5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity.
1 tablet daily, between meals, orally. Do not exceed recommended dosage. Do not administer to children.
Ferrocite™ Tablets are round, green, film-coated tablets debossed B181. Ferrocite™ is supplied in boxes of 100 tablets (4 × 25). NDC# 51991-181-42.
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F). See USP Controlled Room Temperature. Keep in a cool, dry place.
All prescription substitutions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product.
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. Keep this product out of reach of children. In case of accidental overdose, call a doctor or poison control center immediately. |
| These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease. |
Manufactured by: Contract Pharmacal Corp., Hauppauge, NY 11788 USA
Distributed by: Breckenridge Pharmaceutical, Inc
Boca Raton, FL 33487
To report serious adverse events, or for product information contact: 1-800-367-3395.
Rev. 06/09
| FERROCITE
ferrous fumarate tablet, film coated |
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||
| Labeler - Breckenridge Pharmaceutical, Inc. (150554335) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Contract Pharmacal Corporation | 057795122 | MANUFACTURE | |
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.